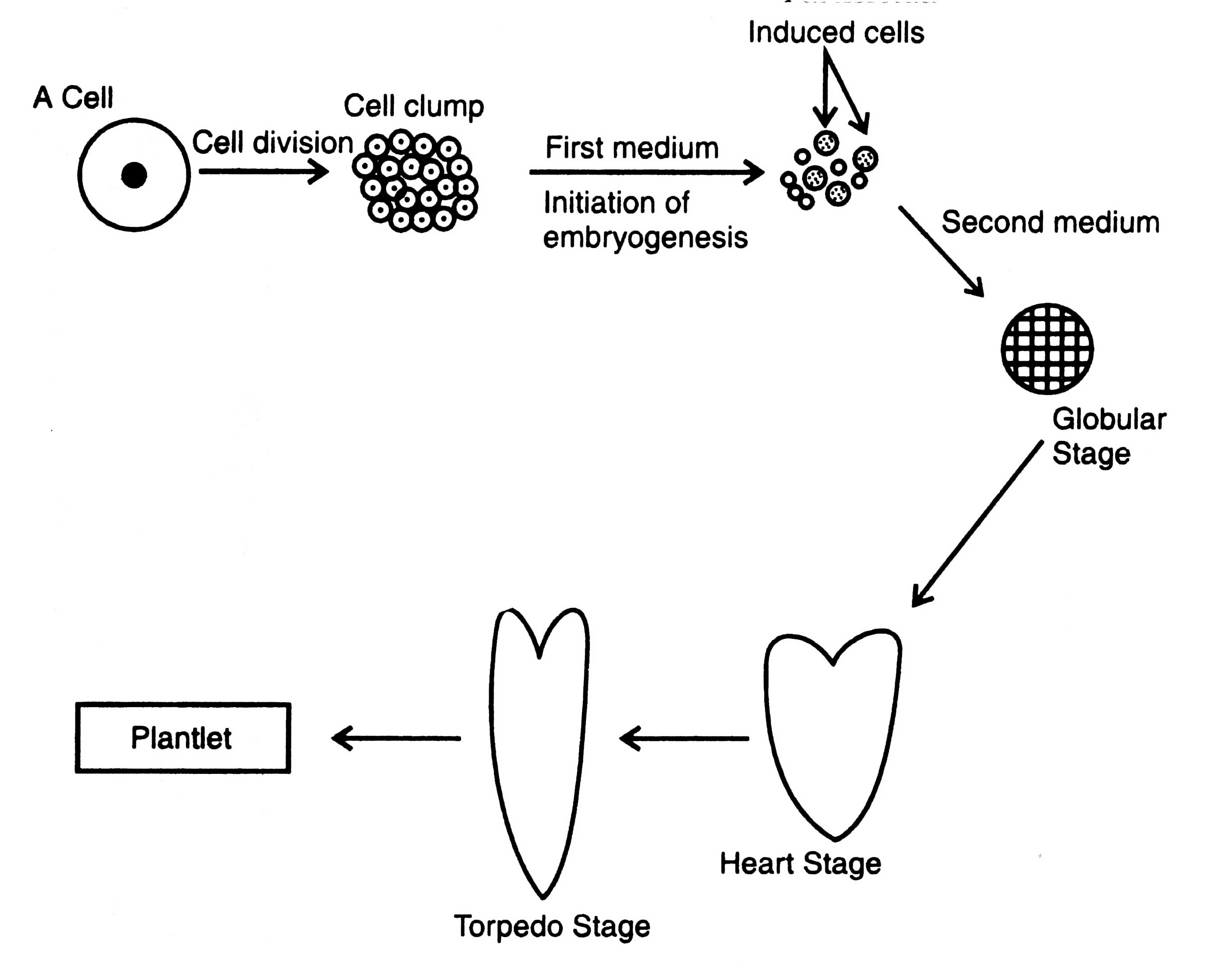
Somatic Embryogenesis
Embryo production is a characteristic feature of the flowering plants. However, such structures (embryoids) have also been artificially induced in cultured plant tissues, besides zygote. Somatic embryogenesis was first induced in suspension culture (Stewart et al. 1958) and callus culture (Reinert, 1959) of carrot. More than 30 plant families are known so far where somatic embryoids have been induced (Raghavan, 1976; Ammirato, 1983).Somatic embryogenesis can be initiated in two ways : (i) by inducing embryogenic cells within the preformed callus, and (ii) directly from preembryonic determined cell, (without callus) which are ready to differentiate into embryoids (Sharp et al 1980). In the first case, ambryoids are initiated in callus from superficial cell aggregates where cells contain a large vacuole, dense cytoplasm, large starch granules and nucleus (Me Willian et al., 1974).Two nutritional media of different composition are required to obtain embryoids. First medium contains auxin to initiate embryogenic cells. Second medium lacks auxin or reduced level of auxin is needed for subsequent development of the embryonic cells into embryoids and plantlets. In both the cases reduced amount of nitrogen is required (Ammirato, 1983). The embryogenic cells pass through 3 different stages e.g. globular, heart shaped and torpedo shaped, to form embryoids (Fig. 8.4).