Aseptic Technique and Transfer of Microorganisms
IntroductionIn natural environments, microorganisms usually exist as mixed populations. However, if we are to study, haracterize, and identify microorganisms, we must have the organisms in the form of a pure culture. A pure culture is one in which all organisms are descendants of the same organism.
In working with microorganisms, we must also have a sterile nutrientcontaining medium in which to grow the organisms. Anything in or on which we grow microorganisms is termed a medium. A sterile medium is one that is free of all life forms. It is usually sterilized by heating it to a temperature at which all contaminating microorganisms are destroyed.
Finally, in working with microorganisms, we must have a method of transferring growing organisms (called the inoculum) from a pure culture to a sterile medium without introducing any unwanted outside contaminants. This method of preventing unwanted microorganisms from gaining access is termed aseptic technique.
Aseptic Technique
The procedure for aseptically transferring microorganisms is as follows:
- Sterilize the inoculating loop. The inoculating loop is sterilized by passing it at an angle through the flame of a gas burner until the entire length of the wire becomes orange from the heat. In this way, all contaminants on the wire are incinerated. Never lay the loop down once it is sterilized or it may again become contaminated. Allow the loop to cool a few seconds to avoid killing the inoculum.
- Remove the inoculum.
- Removing inoculum from a broth culture (organisms growing in a liquid
medium):
- Hold the culture tube in one hand and in your other hand, hold the sterilized inoculating loop as if it were a pencil.
- Remove the cap of the pure culture tube with the little finger of your loop hand. Never lay the cap down or it may become contaminated.
- Very briefly hold a flame to the lip of the culture tube. This creates a convection current that forces air out of the tube and preventing airborne contaminants from entering the tube. The heat of the gas burner also causes the air around your work area to rise, and this also reduces the chance of airborne microorganisms contaminating your cultures.
- Keeping the culture tube at an angle, insert the inoculating loop and remove a loopful of inoculum.
- Again hold a flame to the lip of the culture tube.
- Replace the cap.
- Removing inoculum from a plate culture (organisms growing on an agar
surface in a petri plate):
- Sterilize the inoculating loop in the flame of a gas burner.
- Lift the lid of the culture plate slightly and stab the loop into the agar away from any growth to cool the loop.
- Scrape off a small amount of the organisms and close the lid.
- Removing inoculum from a broth culture (organisms growing in a liquid
- Transfer the inoculum to the sterile medium.
- Transferring the inoculum into a broth tube:
- Pick up the sterile broth tube and remove the cap with the little finger of your loop hand. Do not set the cap down.
- Briefly hold a flame to the lip of the broth tube.
- Place the loopful of inoculum into the broth, and withdraw the loop. Do not lay the loop down.
- Again hold a flame to the lip of the tube.
- Replace the cap.
- Resterilize the loop by placing it in the flame until it is orange. Now you may lay the loop down until it is needed again.
- Transferring the inoculum into a petri plate:
- Lift the edge of the lid just enough to insert the loop.
- Streak the loop across the surface of the agar medium. These streaking patterns allow you to obtain single isolated bacterial colonies originating from a single bacterium or arrangement of bacteria.
- In order to avoid digging into the agar, as you streak the loop over the top of the agar, you must keep the loop parallel to the agar surface. Always start streaking at the “12:00 position” of the plate and streak side-to-side as you pull the loop toward you. Each time you flame and cool the loop between sectors, rotate the plate counterclockwise so you are always working in the “12:00 position” of the plate. This keeps the inoculating loop parallel with the agar surface and helps prevent the loop from digging into the agar.
- Remove the loop and close the lid.
- Resterilize the inoculating.
- Transferring the inoculum into a broth tube:
Forms of Culture Media
- Broth tubes are tubes containing a liquid medium. A typical nutrient
containing broth medium such as rypticase soy broth contains substrates
for microbial growth such as pancreatic digest of casein, papaic digest of
soybean meal, sodium chloride, and water. After incubation, growth
(development of many cells from a few cells) may be observed as 1 or a
combination of 3 forms:
- Pellicle: A mass of organisms is floating on top of the broth.
Turbidity: The organisms appear as a general cloudiness throughout the broth.
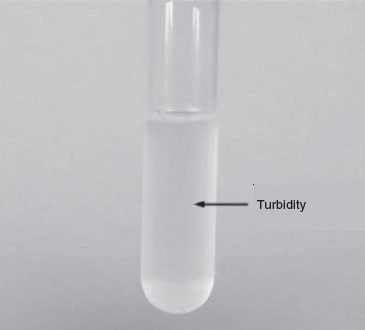
Figure 13 Broth culture showing turbidity.
- Sediment: A mass of organisms appears as a deposit at the bottom of the tube broth.

Figure 14 Culture showing sediment.
- Pellicle: A mass of organisms is floating on top of the broth.
- Slant tubes are tubes containing a nutrient medium plus a solidifying agent, agaragar. The medium has been allowed to solidify at an angle in order to get a flat inoculating surface.
- Stab tubes (deeps) are tubes of hardened agar medium that are inoculated by “stabbing” the inoculum into the agar.
- Agar plates are sterile petri plates that are aseptically filled with a melted sterile agar medium and allowed to solidify. Plates are much less confining than slants and stabs and are commonly used in the culturing, separating, and counting of microorganisms.
Oxygen Requirements for Microbial Growth
Microorganisms show a great deal of variation in their requirements for gaseous oxygen. Most can be placed in one of the following groups:
- Obligate aerobes are organisms that grow only in the presence of oxygen. They obtain energy from aerobic respiration.
- Microaerophiles are organisms that require a low concentration of oxygen for growth. They obtain energy from aerobic respiration.
- Obligate anaerobes are organisms that grow only without oxygen and, in
fact, oxygen inhibits or kills them. They obtain energy from anaerobic
respiration or fermentation. - Aerotolerant anaerobes, like obligate anaerobes, cannot use oxygen for growth, but they tolerate it fairly well. They obtain energy from fermentation.
- Facultative anaerobes are organisms that grow with or without oxygen, but
generally better with oxygen. They obtain energy from aerobic respiration,
anaerobic respiration, and fermentation. Most bacteria are facultative
anaerobes.
Temperature Requirements
Microorganisms are divided into groups on the basis of their preferred range of temperature:
- Psychrophiles are cold-loving bacteria. Their optimum growth temperature is between –5°C and 15°C. They are usually found in the Arctic and Antarctic regions and in streams fed by glaciers.
- Mesophiles are bacteria that grow best at moderate temperatures. Their optimum growth temperature is between 25°C and 45°C. Most bacteria are mesophilic and include common soil bacteria and bacteria that live in and on the body.
- Thermophiles are heat-loving bacteria. Their optimum growth temperature is between 45°C and 70°C and are commonly found in hot springs and compost heaps.
- Hyperthermophiles are bacteria that grow at very high temperatures. Their optimum growth temperature is between 70°C and 110°C. They are usually members of the Archae and are found growing near ydrothermal vents at great depths in the ocean.
Colony Morphology and Pigmentation
A colony is a visible mass of microorganisms growing on an agar surface and usually originating from a single organism or arrangement of organisms. Different microorganisms will frequently produce colonies that differ in their morphological appearance (form, elevation, margin, surface, optical characteristics, and pigmentation). Probably the most visual characteristic is pigmentation (color). Some microorganisms produce pigment during growth and are said to be chromogenic. Often, however, formation of pigment depends on environmental factors such as temperature, nutrients, pH, and moisture. For example, Serratia marcescens produces a deep red pigment at 25°C, but does not produce pigment at 37°C.
Pigments can be divided into 2 basic types: water-insoluble and watersoluble. If the pigment is water-insoluble, as in the case of most chromogenic bacteria, it does not diffuse out of the organism. As a result, the colonies are pigmented but the agar remains the normal color. If the pigment is water-soluble, as in the case of Pseudomonas aeruginosa, it will diffuse out of the organism into the surrounding medium. Both the colonies and the agar will appear pigmented.
Below is a list of several common chromogenic bacteria:
- Staphylococcus aureus - gold; water-insoluble
- Micrococcus luteus - yellow; water-insoluble
- Micrococcus roseus - pink; water-insoluble
- Mycobacterium phlei - orange; water-insoluble
- Serratia marcescens - orange/red; water-insoluble.
Media. Trypticase soy broth tubes (4), trypticase soy agar slant tubes (4), trypticase soy agar stab tubes (4), and trypticase soy agar plates (7).
Organisms. Trypticase soy broth cultures of bacillus subtilis, Escherichia coli, and Micrococcus luteus, and trypticase soy agar plate cultures of Mycobacterium phlei.
Procedure
- Aseptically inoculate one trypticase soy broth tube, one trypticase soy agar slant tube, one trypticase soy agar stab tube, and one trypticase soy agar plate with B. subtilis. Remember to label all tubes with a wax marker. When streaking the agar plates, this procedure is termed streaking for isolation and has a diluting effect. The friction of the loop against the agar causes organisms to fall off the loop. Near the end of the streaking pattern, individual organisms become separated far enough apart on the agar surface to give rise to isolated single colonies after incubation.
- Aseptically inoculate one trypticase soy broth tube, one trypticase soy agar slant tube, one trypticase soy agar stab tube, and one trypticase soy agar plate with E. coli..
- Aseptically inoculate one trypticase soy broth tube, one trypticase soy agar slant tube, one trypticase soy agar stab tube, and one trypticase soy agar plate with M. luteus.
- Aseptically inoculate one trypticase soy broth tube, one trypticase soy agar slant tube, one trypticase soy agar stab tube, and one trypticase soy agar plate with M. phlei.
- Incubate all the tubes and plates inoculated with B. subtilis, E. coli., M. luteus, and M. phlei at 37°C. Place the tubes in a plastic beaker to keep them upright. Incubate the plates upside down (lid on the bottom) to prevent condensing water from falling down on the growing colonies and causing them to run together.
- In order to illustrate that microorganisms are all around us and demonstrate
the necessity for proper aseptic technique, contaminate 3 trypticase soy
agar plates as follows:
- Remove the lid from the first agar plate and place the exposed agar
portion in or out of the building for the duration of today’s lab. Replace
the lid, label the plate “air”, and incubate it at room temperature. Do
this plate first.
- Using a wax marker, divide a second petri plate in half. You and your
partner should both moisten a sterile cotton swab in sterile water. Rub
your swab over some surface in the building or on yourself. Use this
swab to inoculate your half of the second agar plate. Label the plate
and incubate at room temperature.
- With a wax marker, divide a third petri plate in half. Rub your fingers over the surface of your half of the third agar plate. Label and incubate at 37°C. Do this plate last.
- Remove the lid from the first agar plate and place the exposed agar
portion in or out of the building for the duration of today’s lab. Replace
the lid, label the plate “air”, and incubate it at room temperature. Do
this plate first.
Results
- Draw and describe the growth seen in each of the 4 broth cultures.

- Observe the growth in the slant cultures and stab cultures for pigmentation and purity.
- Observe the results of the 3 “contamination” plates and note the differences in colony appearances.
- Observe the demonstration plates of chromogenic bacteria and note the color and water solubility of each pigment.
Performance Objectives Discussion
- Define the following terms: pure culture, sterile medium, inoculum, aseptic technique, and colony.
- Name and define the 3 types of growth that may be seen in a broth culture.
- Define the following terms: obligate aerobe, microaerophile, obligate anaerobe, aerotolerant anaerobe, and facultative anaerobe.
- Define the following terms: psychrophile, mesophile, thermophile, and hyperthermophile.
- Define the following terms: chromogenic, water-soluble pigment, and waterinsoluble pigment.
Procedure
- Using an inoculating loop, demonstrate how to aseptically remove some inoculum from either a broth tube, slant tube, stab tube, or petri plate, and inoculate a sterile broth tube, slant tube, stab tube, or petri plate without introducing outside contamination.
- Label all tubes and plates and place them on the proper shelf in the incubator.
- Dispose of all materials when the experiment is completed, being sure to remove all markings from the glassware. Place all tubes and plates in the designated areas.
Results
- Recognize and identify the following types of growth in a broth culture: pellicle, turbidity, sediment, and any combination of these.
- Name the color and water solubility of pigment seen on a plate culture of a chromogenic bacterium.




