Serology, Part 1–Direct Serologic Testing
Introduction to Serologic TestingThe immune responses refer to the ability of the body (self) to recognize specific foreign factors (nonself) that threaten its biological integrity. There are 2 major branches of the immune responses:
- Humoral immunity: Humoral immunity involves the production of antibody molecules in response to an antigen (antigen: “A substance that reacts with antibody molecules and antigen receptors on lymphocytes.” An immunogen is an antigen that is recognized by the body as nonself and stimulates an adaptive immune response) and is mediated by B-lymphocytes.
- Cell-Mediated immunity: Cell-mediated immunity involves the production of cytotoxic T-lymphocytes, activated macrophages, activated NK cells, and cytokines in response to an antigen, and is mediated by T-lymphocytes.
To understand the immune responses, we must first understand what is meant by the term antigen. Technically, an antigen is defined as a substance that reacts with antibody molecules and antigen receptors on lymphocytes. An immunogen is an antigen that is recognized by the body as nonself and stimulates an adaptive immune response. For simplicity, both antigens and immunogens are usually referred to as antigens.
Chemically, antigens are large molecular weight proteins (including conjugated proteins such as glycoproteins, lipoproteins, and nucleoproteins) and polysaccharides (including lipopolysaccharides). These protein and polysaccharide antigens are found on the surfaces of viruses and cells, including microbial cells (bacteria, fungi, protozoans) and human cells.
As mentioned above, the B-lymphocytes and T-lymphocytes are the cells that carry out the immune responses. The body recognizes an antigen as foreign when that antigen binds to the surfaces of B-lymphocytes and T-lymphocytes because antigen-specific receptors have a shape that corresponds to that of the antigen, similar to interlocking pieces of a puzzle. The antigen receptors on the surfaces of B-lymphocytes are antibody molecules called B-cell receptors or sIg; the receptors on the surfaces of T-lymphocytes are called T-cell receptors (TCRs). The actual portions or fragments of an antigen that react with receptors on B-lymphocytes and T-lymphocytes, as well as with free antibody molecules, are called epitopes, or antigenic determinants. The size of an epitope is generally thought to be equivalent to 5–15 amino acids or 3–4 sugar residues. Some antigens, such as polysaccharides, usually have many epitopes, but all of the same specificity. This is because polysaccharides may be composed of hundreds of sugars with branching sugar side chains, but usually contain only 1 or 2 different sugars. As a result, most “shapes” along the polysaccharide are the same. Other antigens such as proteins usually have many epitopes of different specificities. This is because proteins are usually hundreds of amino acids long and are composed of 20 different amino acids. Certain amino acids are able to interact with other amino acids in the protein chain, and this causes the protein to fold over upon itself and assume a complex 3-dimensional shape. As a result, there are many different “shapes” on the protein. That is why proteins are more immunogenic than polysaccharides; they are more complex chemically.
A microbe, such as a single bacterium, has many different proteins on its surface that collectively form its various structures, and each different protein may have many different epitopes. Therefore, immune responses are directed against many different parts or epitopes of the same microbe.
In terms of infectious diseases, the following may act as antigens:
- Microbial structures (cell walls, capsules, flagella, pili, viral capsids, envelope-associated glycoproteins, etc.)
- Microbial exotoxins.
- 1. Allergens (dust, pollen, hair, foods, dander, bee venom, drugs, and other agents causing allergic reactions);
- Foreign tissues and cells (from transplants and transfusions);
- The body’s own cells that the body fails to recognize as “normal self” (cancer cells, infected cells, cells involved in autoimmune diseases).
Antibodies or immunoglobulins are specific protein configurations produced by B-lymphocytes and plasma cells in response to a specific antigen, and are capable of reacting with that antigen. Antibodies are produced in the lymphoid tissue and once produced, are found mainly in the plasma portion of the blood (the liquid fraction of the blood before clotting). Serum is the liquid fraction of the blood after clotting.
There are 5 classes of human antibodies: IgG, IgM, IgA, IgD, and IgE. The simplest antibodies, such as IgG, IgD, and IgE, are “Y”-shaped macromolecules called monomers, composed of 4 glycoprotein chains. There are 2 identical heavy chains with a high molecular weight that varies with the class of antibody. In addition, there are 2 identical light chains of 1 of 2 varieties: kappa or gamma. The light chains have a lower molecular weight. The four glycoprotein chains are connected to one another by disulfide (S-S) bonds and noncovalent bonds. Additional S-S bonds fold the individual glycoprotein chains into a number of distinct globular domains. The area where the top of the “Y” joins the bottom is called the hinge. This area is flexible to enable the antibody to bind to pairs of epitopes various distances apart on an antigen.
Two classes of antibodies are more complex. IgM is a pentamer, consisting of 5 “Y”-like molecules connected at their Fc portions, and secretory IgA is a dimer consisting of 2 “Y”-like molecules.
Serology refers to using antigen-antibody reactions in the laboratory for diagnostic purposes. Its name comes from the fact that serum, the liquid portion of the blood where antibodies are found, is used in testing. Serologic testing may be used in the clinical laboratory in 2 distinct ways: to identify unknown antigens (such as microorganisms) and to detect antibodies being made against a specific antigen in the patient’s serum. There are 2 types of serologic testing: direct and indirect.
- Direct serologic testing is the use of a preparation of known antibodies, called antiserum, to identify an unknown antigen such as a microorganism.
- Indirect serologic testing is the procedure whereby antibodies in a person’s serum, made by that individual against an antigen associated with a particular disease, are detected using a known antigen.
Using Antigen-Antibody reactions in the Laboratory to Identify Unknown Antigens such as Microorganisms
This type of serologic testing employs known antiserum (serum containing specific known antibodies). The preparation of known antibodies is prepared in 1 of 2 ways: in animals or by hybridoma cells.
Preparation of known antisera in animals
Preparation of known antiserum in animals involves inoculating animals with specific known antigens, such as a specific strain of a bacterium. After the animal‘s immune responses have had time to produce antibodies against that antigen, the animal is bled and the blood is allowed to clot. The resulting liquid portion of the blood is the serum and it will contain antibodies specific for the injected antigen.
However, one of the problems of using antibodies prepared in animals (by injecting the animal with a specific antigen and collecting the serum after antibodies are produced) is that up to 90% of the antibodies in the animal’s serum may be antibodies the animal has made “on its own” against environmental antigens, rather than those made against the injected antigen. The development of monoclonal antibody technique has largely solved that problem.
Preparation of known antibodies by monoclonal antibody technique
One of the major breakthroughs in immunology occurred when monoclonal antibody technique was developed. Monoclonal antibodies are antibodies of a single specific type. In this technique, an animal is injected with the specific antigen for the antibody desired. After the appropriate time for antibody production, the animal’s spleen is removed. The spleen is rich in plasma cells, and each plasma cell produces only 1 specific type of antibody. However, plasma cells will not grow artificially in cell culture. Therefore, a plasma cell producing the desired antibody is fused with a myeloma cell, a cancer cell from bone marrow that will grow rapidly in cell culture, to produce a hybridoma cell. The hybridoma cell has the characteristics of both parent cells. It will produce specific antibodies like the plasma cell and will also grow readily in cell culture like the myeloma cell. The hybridoma cells are grown artificially in huge vats, where they produce large quantities of the specific antibody.
Monoclonal antibodies are now used routinely in medical research and diagnostic serology, and are being used experimentally in treating certain cancers and a few other diseases.
The concept and general procedure for direct serologic testing
The concept and general procedure for using antigen-antibody reactions to identify unknown antigens are as follows:
- Concept. This testing is based on the fact that antigen-antibody reactions are very specific. Antibodies usually react only with the antigen that stimulated their production in the first place, and are just as specific as an enzyme-substrate reaction. Because of this, one can use known antiserum (prepared by animal inoculation or monoclonal antibody technique as discussed above) to identify unknown antigens such as a microorganisms.
- General Procedure. A suspension of the unknown antigen to be identified is mixed with known antiserum for that antigen. One then looks for an antigen-antibody reaction.
Examples of serologic tests used to identify unknown microorganisms include the serological typing of Shigella and Salmonella, the Lancefield typing of beta streptococci, and the serological identification of meningococci. Serological tests used to identify antigens that are not microorganisms include blood typing, tissue typing, and pregnancy testing.
Detection of antigen-antibody reactions in the laboratory
Antigen-antibody reactions may be detected in the laboratory by a variety of techniques. Some of the commonly used techniques for observing in vitro antigenantibody reactions are briefly described below.
- Agglutination. Known antiserum causes bacteria or other particulate antigens to clump together or agglutinate. Molecular-sized antigens can be detected by attaching the known antibodies to larger, insoluble particles such as latex particles or red blood cells in order to make the agglutination visible to the naked eye.
- Precipitation. Known antiserum is mixed with soluble test antigen and a cloudy precipitate forms at the zone of optimum antigen-antibody proportion.
- Complement-fixation. Known antiserum is mixed with the test antigen and complement is added. Sheep red blood cells and hemolysins (antibodies that lyse the sheep red blood cells in the presence of free complement) are then added. If the complement is tied up in the first antigen-antibody reaction, it will not be available for the sheep red blood cell-hemolysin reaction and there will be no hemolysis. A negative test would result in hemolysis.
- Enzyme-linked immunosorbant assay or ELISA (also known as enzyme immunoassay, or EIA). Test antigens from specimens are passed through a tube (or a membrane) coated with the corresponding specific known antibodies and become trapped on the walls of the tube (or on the membrane). Known antibodies to which an enzyme has been chemically attached are then passed through the tube (or membrane), where they combine with the trapped antigens. Substrate for the attached enzyme is then added and the amount of antigen-antibody complex formed is proportional to the amount of enzyme-substrate reaction, as indicated by a color change.
- Radioactive binding techniques. Test antigens from specimens are passed through a tube coated with the corresponding specific known antibodies, and become trapped on the walls of the tube. Known antibodies to which a radioactive isotope has been chemically attached are then passed through the tube, where they combine with the trapped antigens. The amount of antigen-antibody complex formed is proportional to the degree of radioactivity.
- Fluorescent antibody technique. A fluorescent dye is chemically attached to the known antibodies. When the fluorescent antibody reacts with the antigen, the antigen will fluoresce when viewed with a fluorescent microscope.
Examples of a Direct Serologic Test to Identify Unknown Antigens
As stated above, this type of serologic testing uses known antiserum (antibodies) to identify unknown antigens.
Serological Typing of Shigella
There are 4 different serological subgroups of Shigella, each corresponding to a different species:
- Subgroup A = Shigella dysenteriae
- Subgroup B = Shigella flexneri
- Subgroup C = Shigella boydii
- Subgroup D = Shigella sonnei
Known antiserums are available for each of the 4 subgroups of Shigella listed above, and contain antibodies against the cell wall (“O” antigens) of Shigella. The suspected Shigella (the unknown antigen) is placed in each of 4 circles on a slide and a different known antiserum (A, B, C, or D) is then added to each circle. A positive antigen-antibody reaction appears as a clumping or agglutination of the Shigella.
Serological Typing of Streptococci
Many of the streptococci can be placed into serological groups called Lancefield groups based on carbohydrate antigens in their cell wall. Although there are 20 different Lancefield groups of streptococci, the groups A, B, C, D, F, and G are the ones usually associated with human infections. The Slidex Strepto-Kit® system is a commercial kit for typing the 6 Lancefield groups of streptococci that commonly infect humans. To make the reaction more visible, since the antigens for which one is testing are only fragments of the bacterial cell wall, the known monoclonal antibodies have been adsorbed to latex particles. This way, when the known monoclonal antibodies react with the streptococcal cell wall antigens, agglutination of the latex particles will occur and can be easily seen with the naked eye.
Serological Testing to Diagnose Pregnancy
The hormone human chorionic gonadotropin (HCG), produced by the placenta, appears in the serum and urine of pregnant females. The HCG is composed of 2 subunits — alpha and beta. The CARDS O.S.® HCG-Serum/Urine is a 1-step pregnancy test that detects measurable levels of HCG as early as 7–10 days after conception. HCG, the unknown antigen for which one is testing, is identified in the urine by using known monoclonal antibody against human HCG.
This test uses a color immunochromatographic assay to detect the antigenantibody reaction. Inside the plastic card is a membrane strip along which the urine flows and on which the reaction occurs. The urine is placed in the “add urine” well on the right side of the card and flows along the card from right to left. The membrane just to the left of the sample well is coated with red latex beads, to which known antibodies against the beta chain of human HCG have been attached. If there is HCG in the urine, the beta subunit of the HCG will react with the known anti-beta HCG antibody/red latex conjugate and this complex of HCG-antibody/red latex will become mobilized and flow with the urine towards the left side of the card. In the “read results” window of the card is a vertical line towards which known antibodies against the alpha subunit of human HCG move. As the urine containing the antibody/red latex conjugate bound to the beta subunit of HCG flows past the vertical line, the alpha subunit of the HCG binds to the immobilized antibodies located on the line, trapping the complex and causing a vertical red line to appear. The vertical red line crosses the horizontal blue line preprinted in the “read results” window to form a (+) sign.
If the woman is not pregnant and there is no HCG in the urine, there will be no antigen to react with the anti-beta HCG antibody/red latex conjugate to the left of the sample well and, likewise, no reaction with the anti-alpha HCG antibodies immobilized along the vertical line in the “read results” window. The antibody/red latex conjugate will continue to flow to the left of the slide until it reaches the “test complete” window. Since no vertical red line forms, a (–) sign appears in the “read results” window.
Identification of Microorganisms Using the Direct Fluorescent Antibody Technique
Certain fluorescent dyes can be chemically attached to the known antibody molecules in antiserum. The known fluorescent antibody is then mixed with the unknown antigen, such as a microorganism, fixed to a slide. After washing, to remove any fluorescent antibody not bound to the antigen, the slide is viewed with a fluorescent microscope.
If the fluorescent antibody reacted with the unknown antigen, the antigen will glow or fluoresce under the fluorescent microscope. If the fluorescent antibody did not react with the antigen, the antibodies will be washed off the slide and the antigen will not fluoresce.
Many bacteria, viruses, and fungi can be identified using this technique.
Procedure
Serologic Typing of Shigella
- Using a wax marker, draw 2 circles (about the size of a nickel) on 2 clean glass slides. Label the circles A, B, C, and D.
- Add 1 drop of the suspected Shigella (unknown antigen) to each circle. (The Shigella has been treated with formalin to make it noninfectious but still antigenic.)
- Now add 1 drop of known Shigella subgroup A antiserum to the “A” circle, 1 drop of known Shigella subgroup B antiserum to the “B” circle, 1 drop of known Shigella subgroup C antiserum to the “C” circle, and 1 drop of known Shigella subgroup D antiserum to the “D” circle.
- Rotate the slide carefully for 30–60 seconds. Agglutination of the bacteria indicates a positive reaction. No agglutination is negative.
- Dispose of all pipettes and slides in the disinfectant container.
Serologic Typing of Streptococci
- The cell wall antigens of the unknown Streptococcus used in this test are extracted by mixing the organism with extraction enzyme. This step has been done for you.
- Place 1 drop of the appropriate known streptococcal monoclonal antibody/ latex conjugate (groups A, B, C, D, F, and G) on the corresponding 6 circles of the slide.
- Add 1 drop of the extracted antigen from the unknown Streptococcus prepared in step 1 to each circle.
- Spread the antigen-antibody mixtures over the entire circles using separate applicator sticks for each circle.
- Rock the slide back and forth for no longer than 1 minute and look for agglutination.
Serologic Testing to Detect Pregnancy
- Fill the disposable pipette to the line with urine and dispense the urine into the “add urine” well.
- Shortly after the urine is added, a blue color will be seen moving across the “read results” window.
- The test results can be read in the “read results” window when a distinct blue line appears in the “test complete” window (approximately 5 minutes). A (+) sign indicates a positive test; a (–) sign is a negative test.
The Direct Fluorescent Antibody Technique
Observe the demonstration of a positive direct fluorescent antibody test.
Results
Serologic Typing of Shigella
Make a drawing of your results.
- Agglutination of bacteria is positive.
- No agglutination of bacteria is negative.
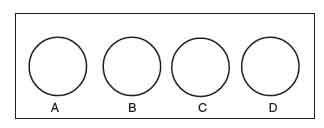 |
Figure 53 Shigella typing slide B. |
Serologic Typing of Streptococci
Make a drawing of your result.
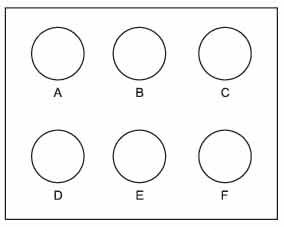 |
Figure 54 Streptococcus typing slide. |
Serologic Testing to Diagnose Pregnancy
Make a drawing of a positive pregnancy test.
 |
Figure 55 Positive pregnancy test. |
The Direct Fluorescent Antibody Technique
Make a drawing of and describe a positive direct fluorescent antibody test.
Performance Objectives
After completing this lab, the student will be able to perform the following objectives:
Introduction to Serological Testing
- Define serology.
- Define antigen and describe what might act as an antigen.
- Define antibody and explain where it is primarily found in the body.
- Define direct serologic testing and indirect serologic testing.
Using Antigen-Antibody Reactions in the Lab to Identify unknown Antigens Such as Microorganisms
- Define antiserum.
- Describe 2 ways of producing a known antiserum.
- Describe the concept and general procedure for using serologic testing to identify unknown antigens (direct serologic testing).
Examples of Serologic Tests to Identify Unknown Antigens
- Describe how to determine serologically whether an organism is a subgroup A, B, C, or D Shigella.
- Describe how to determine serologically whether an organism is a Lancefield group A, B, C, D, F, or G Streptococcus.
- Describe how to diagnose pregnancy serologically.
- Briefly describe the direct fluorescent antibody technique.
Results
Correctly interpret the results of the following serological tests:
- Serological typing of Shigella
- Serological typing of streptococci
- Serological testing for pregnancy
- A direct fluorescent antibody test.




