Centrifugation
A centrifuge is a device for separating particles from a solution according to their size, shape, density, viscosity of, and rotor speed. In biology, the particles are usually cells, subcellular organelles, viruses, large molecules such as proteins, and nucleic acids. To simplify mathematical terminology, we will refer to all biological material as spherical particles. There are many ways to classify centrifugation.The single most important advance in the use of centrifugal force to separate biologically important substances was the combination of mechanics, optics, and mathematics by T. Svedberg and J.W. Williams in the 1920s. They initiated the mathematics and advanced the instrumentation.
Nowadays, any technique employing the quantitative application of centrifugal force is known as ultracentrifugation
Rotors
Rotors for a centrifuge are either fixed angles, swinging buckets, continuous flow, or zonal, depending upon whether the sample is held at a given angle to the rotation plane, allowed to swing out on a pivot and into the plane of rotation, designed with inlet and outlet ports for separation of large volumes, or a combination of these.
Fixed angles generally work faster; substances precipitate faster in a given rotational environment, or they have an increased relative centrifugal force for a given rotor speed and radius. These rotors are the workhorse elements of a cell laboratory, and the most common is a rotor holding 8 centrifuge tubes at an angle of 34°C from the vertical.
Swinging bucket rotors (horizontal rotors) have the advantage that there is usually a clean meniscus of minimum area. In a fixed-angle rotor, the materials are forced against the side of the centrifuge tube, and then slide down the wall of the tube. This action is the primary reason for their apparent faster separation, but also leads to abrasion of the particles along the wall of the centrifuge tube. For a swinging bucket, the materials must travel down the entire length of the centrifuge tube and always through the media within the tube. Since the media is usually a viscous substance, the swinging bucket appears to have a lower relative centrifugal force, and it takes longer to precipitate anything contained within. If, however, the point of centrifugation is to separate molecules or organelles on the basis of their movements through a viscous field, then the swinging bucket is the rotor of choice. Most common clinical centrifuges have swinging buckets.
Cell biologists employ zonal rotors for the large-scale separation of particles on density gradients. The rotors are brought up to about 3000 rpm while empty, and the density media and tissues are added through specialized ports.
Rotor Tubes
In using either a fixed-angle or swinging-bucket rotor, it is necessary to contain the sample in some type of holder. Continuous and zonal rotors are designed to be used without external tubes.
For biological work the tubes are divided into functional groups, made of regular glass, Corex glass, nitrocellulose, or polyallomer. Regular glass centrifuge tubes can be used at speeds below 3000 rpm, that is, in a standard clinical centrifuge. Above this speed, the xg forces will shatter the glass.
For work in the higher speed ranges, centrifuge tubes are made of plastic or nitrocellulose. Preparative centrifuge tubes are made of polypropylene and can withstand speeds up to 20,000 rpm.
Analytical/Preparative Centrifugation
The 2 most common types of centrifugation are analytical and preparative; the distinction is between the 2 is based on the purpose of centrifugation.
Analytical centrifugation involves measuring the physical properties of the sedimenting particles, such as sedimentation coefficient or molecular weight.
Optimal methods are used in analytical ultracentrifugation. Molecules are observed by optical system during centrifugation, to allow observation of macromolecules in solution as they move in the gravitational field.
The samples are centrifuged in cells with windows that lie parallel to the plane of rotation of the rotor head. As the rotor turns, the images of the cell (proteins) are projected by an optical system onto film or a computer. The concentration of the solution at various points in the cell is determined by absorption of a light of the appropriate wavelength. This can be accomplished either by measuring the degree of blackening of a photographic film or by the deflection of the recorder of the scanning system and fed into a computer.
The other type of centrifugation is called preparative and the objective is to isolate specific particles that can be reused. There are many type of preparative centrifugation such as rate zonal, differential, and isopycnic centrifugation.
Ultra centrifugation/Low-Speed Centrifugation
Another system of classification is the rate or speed at which the centrifuge is turning. Ultracentrifugation is carried out at speed faster than 20,000 rpm. Super speed ultracentrifugation is at speeds between 10,000 and 20,000 rpm. Low-speed centrifugation is at speeds below 10,000 rpm.
Moving boundary/Zone Centrifugation
A third method of defining centrifugation is by the way the samples are applied to the centrifuge tube. In moving boundary (differential) centrifugation, the entire tube is filled with sample and centrifuged. Through centrifugation, one obtains a separation of 2 particles, but any particle in the mixture may end up in the supernatant or the pellet, or it may be distributed in both fractions,depending upon its size, shape, density, and conditions of centrifugation. The pellet is a mixture of all of the sedimented components, and is contaminated with whatever unsedimented particles were in the bottom of the tube initially. The only component that is purified is the slowest-sedimenting one, but its yield is often very low. The 2 fractions are recovered by decanting the supernatant solution from the pellet. The supernatant can be recentrifuged at a higher speed to obtain further purification, with the formation of a new pellet and supernatant.
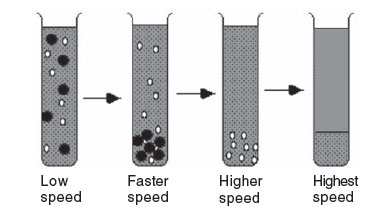 |
FIGURE 3 Differential centrifugation. |
In rate zonal centrifugation, the sample is applied in a thin zone at the top of the centrifuge tube on a density gradient. Under centrifugal force, the particles will begin sedimenting through the gradient in separate zones, according to their size, shape, and density. The run must be terminated before any of the separated particles reach the bottom of the tube.
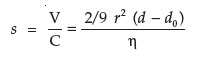
 |
Figure 4 (a) |
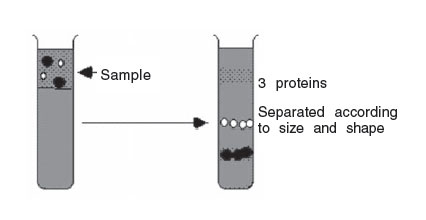 |
FIGURE 4(b) Rate zonal centrifugation. |
In isopycnic technique, the density gradient column encompasses the whole range of densities of the sample particles. The sample is uniformLy mixed with the gradient material. Each particle will sediment only to the position in the centrifuge tube at which the gradient density is equal to its own density, and it will remain there. The isopycnic technique, therefore, separates particles into zone solely on the basis of their density differences, independent of time. In many density gradient experiments, particles of both the rate zonal and isopycnic principles may enter into the final separations. For example, the gradient may be of such a density range that one component sediments to its density in the tube and remains there, while another component sediments to the bottom of the tube. The self-generating gradient technique often requires long hours of centrifugation. Isopycnically banding DNA, for example, takes 36 to 48 hours in a self-generating cesium chloride gradient. It is important to note that the run time cannot be shortened by increasing the rotor speed; this only results in changing the position of the zones in the tube, since the gradient material will redistribute farther down the tube under greater centrifugal force.
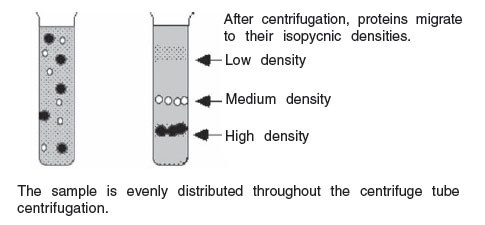 |
FIGURE 5 Isopycnic separation with a self-generating gradient. |
Basic Theory of Sedimentation
Molecules separate according to their size, shape, density, viscosity, and centrifugal force. The simplest case is a spherical molecule. If the liquid has the density of do and the molecule has a density of d, and if d > do , then the protein will sediment. In gravitational field, the motor force (Pg) equals the acceleration of gravity (g) multiplied by the difference between the mass of the molecule and the mass of a corresponding volume of medium.
Equation 1. Pg = (m – m0)g
Equation 2. Pg = 4/3 (3.14) r3 dg –4/3 (3.14) r3 do g
Equation 3. Pg = (4/3) r3 (3.14) (d – do )g
where,
Pg = force due to gravity,
g = acceleration of gravity,
do = density of liquid (or gradient)
d = density of molecule,
m = mass of the molecule,
mo = mass of equal volume of medium.
In a centrifugal field, the gravitational acceleration (g) is replaced by the centrifugal force.
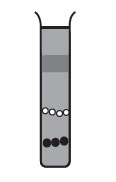 |
FIGURE 6 Sedimentation of particles by gravity. |




