Determination of Hardness of Water by EDTA Titrimetric Method
To determine the total hardness of the given samples by EDTA titrimetric method.
Principle
Originally, the hardness of water was understood to be a measure of the capacity of water for precipitating soap. Soap is precipitated chiefly by the calcium and magnesium ions commonly present in water, but may also be precipitated by ions of other polyvalent metals, such as aluminium, iron, manganese, strontium and zinc, and by hydrogen ions. Because, all but the first two are usually present in insignificant concentrations in natural waters, hardness is defined as a characteristic of water, which represents the total concentration of just the calcium and the magnesium ions expressed as calcium carbonate. However, if present in significant amounts, other hardness producing metallic ions should be included.
1. Burette
2. Pipette
3. Erlenmeyer flask
4. Bottle etc.
Reagents (click to check the preparation of reagents)
1. Standard EDTA titrant (0.01 M)
2. Eriochrome black T indicator
3. Ammonia buffer solution
1. Dilute 25 mL of sample (V) to about 50 mL with distilled water in an Erlenmeyer flask.
2. Add 1 mL of buffer solution.
3. Add two drops of indicator solution. The solution turns wine red in colour.
4. Add the standard EDTA titrant slowly with continuous stirring until the last reddish tinge disappears from the solution. The colour of the solution at the end point is blue under normal conditions.
5. Note down the volume of EDTA added (V1).
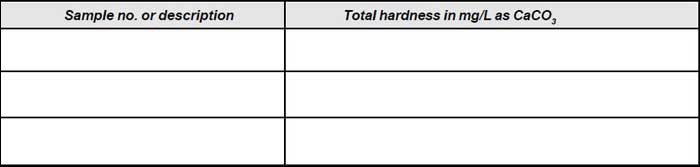
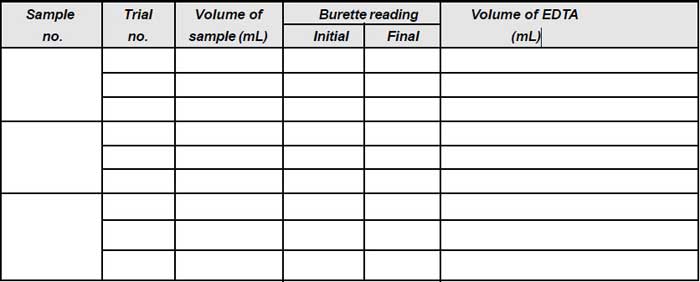
Observation
Calculation
| Hardness as CaCO3= | V1 x S x 1000 | mg / L |
| V |
S = mg CaCO3 equivalent to 1 mL of EDTA titrant
= 1 mg CaCO3
| Hardness as CaCO3= | 1000 V1 | = ..........mg / L |
| V |