Inorganic Ion Transport and Integration of Environmental Information
In the previous section, the transport of ions across the membrane was initiated by an environmental signal, namely, light. This resulted in a change in the transmembrane voltage and a subsequent influx of calcium ions into the nerve terminal of a sensory cell (Fig. 1), resulting in the release of a chemical signal. In general, these chemical signals (neurotransmitters) are released from the nerve terminals of one cell and diffuse across a gap between cells called a synapse (Fig. 1). In the membrane of the adjacent cell, about 20–40 nm removed from the nerve terminal, the neur otransmitters bind to transmembrane neurotransmitter receptors. On binding the neurotransmitter, these transmembrane proteins, transiently (a few milliseconds) open a transmembrane channel (Fig. 1). These channels are specific for inorganic cations, sodium, or potassium, and some are also specific for calcium or chloride ions, depending on the receptor. If the resulting change in the transmembrane voltage, Vm, is of appropriate sign and magnitude (~+20 mV) the voltage change will be propagated along the axon of the cell (Fig. 1), by a process in which sodium and potassium ions move across the membrane. This is described in detail at the end of this section.The major neurotransmitters are listed in Table I. Typical excitatory neurotransmitters are acetylcholine, glutamate, and serotonin. They bind to receptors that are named in the same way, for instance, the acetylcholine receptor, glutamate receptor, etc. These receptors on binding their specific
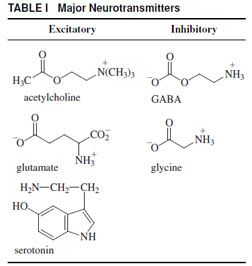 |
| TABLE I Major Neurotransmitters |
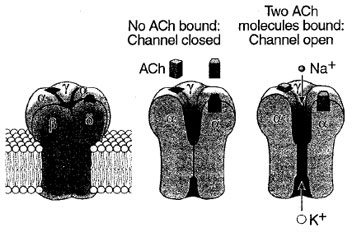 |
| Figure 6 Three-dimensional model of the nicotinic acetylcholine- gated ion channel. The structure is based on a threedimensional reconstruction of electron microscope images by Unwin and his colleagues. The receptor-channel complex consists of five subunits, all of which contribute to forming the pore. When two molecules of acetylcholine bind to portions of the α-subunits exposed to the membrane surface, the receptor-channel changes conformation. This opens a pore in the portion of the channel embedded in the lipid bilayer, and both K+ and Na+ flow through the open channel down their electrochemical gradients. (Reproduced with permission from Kandel, E. R., Schwartz, J. H., and Jessel, T. M. (2000). “Principles of Neuronal Science,” 4th edition. McGraw Hill, New York.) |
What is the relationship between the neurotransmitter concentration, the fraction of receptor-channels that open, the conductance of the channel, and the length of time the channels remain open (Fig. 2)? If we can answer these questions, and can also determine the concentration of receptor sites exposed to the neurotransmitter, we can calculate the change in transmembrane voltage (Eqs. 1–3) and predict whether or not a signal will be transmitted. The techniques for elucidating the mechanism of the reactions that allowone to determine the rate of transmembrane flux of inorganic ions and, therefore, the change in Vm have been discussed in Section I. It is important to mention that each cell is contacted by as many as 1000 different projections from other cells and each of these projections may release a different neurotransmitter from the nerve terminal. The mammalian brain contains an estimated 1012 cells; its information content is believed to exceed, by far, that of the largest supercomputers.
It appears that the techniques are now at hand to (a) investigate the kinetic mechanism of the reactions by which neurotransmitter receptors determine the rate of flow of inorganic ions across the membrane, and (b) determine whether or not a signal will be transmitted to another cell. The chemical mechanism of receptor-mediated transport of inorganic ions across the membrane of neurons (nerve cells) by a few neurotransmitter receptors has been investigated. The mechanisms by which the many receptor isoforms facilitate the flow of inorganic ions across the cell membrane are still unknown. Also virtually unknown are the effects of diseases, causing mutations of receptors, and the influence of hundreds of drugs including anticonvulsants, antidepressants, anesthetics, and abused drugs, on the mechanisms.




