Ion Transport
The plasma membrane is the barrier that separates the cytoplasm of cells from the exterior medium. All cells maintain a membrane potential that is negative. There is an excess of positive charge in the external medium in comparison with that in the cytoplasm. The membrane potential in plant cells can be as high as —200 mV. Energy is required to generate and maintain the membrane potential.All cells maintain gradients in ions across the plasma membrane. The intracellular K+ concentration is higher than that of the extracellular medium, and the concentration of Na+, much lower. The free Ca2+ concentration in the cytoplasm is maintained at very low levels, 1000- fold or more below the extracellular Ca2+ concentration. Often the intracellular proton concentration can be quite different from that in the medium. The pH in the cytoplasm of plant cells is close to 7.0, whereas that in the medium is about 5.0. Energy is needed to generate and maintain these ionic disequilibria. For example, the energy cost to generate a pH gradient of two pH units is equal to RT ln([Ho+ ]/[Hi+ ]), where the subscripts o and i stand for outside and inside the cell, respectively. At 25°C, the ΔG' for a 100-fold proton activity (pH 7.0 in versus pH 5.0 out) gradient is 2.7 kcal/mol.
Plasma membranes of all higher organisms contain enzymes that are embedded in the membrane that act as ion pumps. That is, they catalyze the transport of ions against their electrochemical potential. In physiology, transport that is thermodynamically uphill is termed active transport to distinguish it from the spontaneous flow of ions down their electrochemical potential. The energy needed for the active transport of ions across the plasma membrane is provided by the hydrolysis of ATP to ADP and Pi. As much as 75% of cellular ATP may be consumed simply to generate and maintain ion gradients.
The electrogenic ion pump in the plasma membrane of animal cells is the Na+/K+-ATPase. As shown in Fig. 12, three Na+ ions are transported out of the cell and two K+ ions are pumped in for each ATP that is hydrolyzed. Since three positively charged ions are exported, but only two imported, the Na+/K+-ATPase is electrogenic. The trans plasma membrane potential is on the order of —50 mV. In addition, the pump keeps the intracellular Na+ concentration nearly 100-fold lower than that in the serum, and the intracellular concentration of K+, about 30-fold higher than in serum.
Indirectly, the Na+/K+-ATPase provides the energy for the active transport of amino acids and some carbohydrates into cells. The plasma membrane contains specific proteins that mediate the transport of these molecules in a manner that is obligatorily linked to the cotransport of Na+. Since the extracellular Na+ concentration is higher than that in the cytoplasm and the membrane potential is negative, the Na+ flows from outside to inside the cell. Assuming a membrane potential of —50 mV and a 100-fold Na+ concentration gradient, the flow of Na+ would liberate about 3.8 kcal/mol at 25°C. This exergonic flow of Na+ provides the energy needed for the active transport of the amino acid or carbohydrate. Although Na+ flux is the immediate source of energy for the active transport in Na+-linked transporters, it is important to keep in mind that the ultimate energy source is ATP hydrolysis by the Na+/K+-ATPase.
Plants, yeasts, and fungi do not contain a Na+/K+- ATPase in their plasma membranes. Instead, they contain aH+-ATPase that is the generator of the plasma membrane potential. The H+-ATPase is structurally and mechanistically related to the Na+/K+-ATPase but translocates only H+. The H+-ATPase is capable of generating large electrochemical proton gradients. The imbalance in the Na+ and K+ concentrations between the inside and the outside of the plant cell is maintained by other mechanisms that include exchange transport of Na+ for H+.
The active transport of some organic molecules across the plasma membrane of plants, yeasts, and fungi is linked to the cotransport of H+ down its eletrochemical gradient into the cell. An important example of proton-linked transport is that of sucrose loading into the vascular element, the phloem, that transports sucrose from the leaves to the remainder of a plant. The concentration of sucrose in phloem cells near leaves that are actively carrying out photosynthesis can be 0.5 M or higher, whereas that in the intracellular space, just 0.001 M. The energy cost of generating this gradient is 3.7 kcal/mol at 25°C. The immediate source of energy is proton flow, and the ultimate source, ATP hydrolysis by the H+-ATPase.
The concentration of free Ca2+ (meaning that unbound to proteins and membrane lipids) in the cytoplasm of cells is normally maintained at a very low level. Under certain circumstances, however, transient increases in the cytoplasmic Ca2+ concentration are triggered. Ca2+ is a major player in the transmission of some hormonally induced signals in plants and animals. Muscle contraction is also induced by release of Ca2+ from internal membranes within muscle cells.
The plasma membrane contains an enzyme that catalyzes the export of Ca2+ from the cytoplasm at the expense of ATP hydrolysis. The Ca2+-ATPase has features that place it in the category of plasma membrane enzymes that also includes the Na+/K+-ATPase and the H+-ATPase. The Ca2+-ATPase functions to keep the cytosolic Ca2+concentration low ( < 1 µM). It is not a major contributor to the generation of the membrane potential or to the energetics of the transport of bioorganic molecules.
Inhibitors of the enzyme responsible for the acidification of the stomach are well known and equally well-advertised alleviators of “heartburn.” This enzyme is present in the parietal cells of the stomach and resembles the Na+/K+-ATPase. Instead of catalyzing the ATP-dependent exchange of Na+ and K+, the stomach acid pump excretes H+ into the lumen of the stomach in exchange for K+.
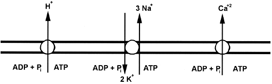 |
| Figure 12 Some ion pumps in the plasma membrane. The Na+/K+-ATPase of animal cells uses the energy of ATP hydrolysis to move three Na+ ions out of the cells and two K+ ions in, which results in the generation of ion gradients and a membrane potential. Plant, yeast, and fungal cells do not have a Na+/K+-ATPase, but instead have a H+-ATPase, as the electrogenic pump. The plasma membrane also contains a Ca2+-ATPase that pumps Ca2+ out of cells to help keep the intracellular Ca2+ concentration low. |




