Mutations induced due to radiations or due to chemicals could be broadly of two types - (i) lethal mutations, (ii) visible mutations. Both these types could be located either on sex chromosomes or on autosomes (autosomes are chromosomes other than sex chromosomes). Different methods for detection of sex linked lethals, sex linked visibles and autosomal mutations are available.
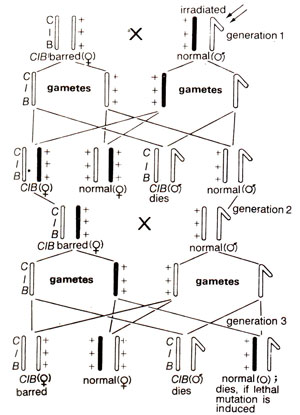
Fig. 21.3. Muller's CIB method for detection of sex linked lethal mutations in Drosophila.
Detection of sex linked lethals
The ClB method. This method involves use of a
ClB stock which carries (i) an inversion in heterozygous state to work as crossover
suppressor (C), (ii) a
recessive lethal (
l) on X-chromosome in heterozygous state, and (iii) a dominant marker,
Barred (
B)for the
barred eye (narrow eye). One of the two X-chromosomes in a female fly carried all these three features and the other X-chromosome was normal. Male flies irradiated for induction of mutations were crossed to
ClB females. Male progeny receiving
ClB X-chromosome will die (Fig. 21.3).

Fig. 21.3. Muller's CIB method for detection of sex linked lethal mutations in Drosophila.
The
ClB female flies obtained in progeny can be detected by barred phenotype. These are crossed to normal males. In the next generation 50% of males receiving
ClB X-chromosome will die. The other 50% males will receive X-chromosome, which may or may not carry the induced mutation. In case lethal mutation was induced no males will be observed. On the other hand, if no lethal mutation was induced, 50% males will survive. Thus, the
ClB method was the most efficient method for detecting sex linked lethal mutations. This has since been improved upon and therefore, has been replaced by Muller-5 method.
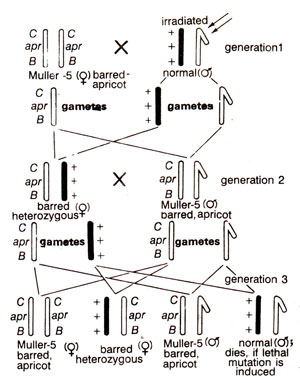
Fig. 21.4. Muller-5 method for detection of sex linked mutations in Drosophila.
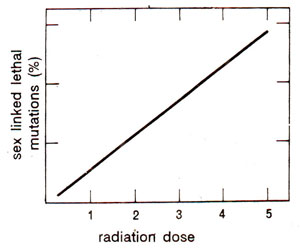
Fig. 21.5. A linear relationship between the frequency of sex linked lethal mutations and the dose of radiation in kilo Roentgens (kR)
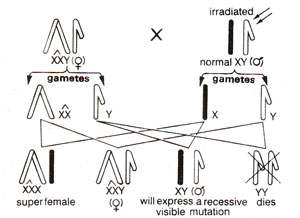
Fig. 21.6. Attached X-method for detection of sex linked visible mutations.
MuIIer-5 method. This method makes use of a
Muller-5 Drosophila stock, which carries two marker genes, dominant 'Bar'
(barred eye) and recessive
'apricot', but does not carry a lethal as in
CIB. Moreover, Muller-5 contains a more complex inversion and therefore, has a better crossover suppressor than in
ClB. As shown in Figure 21.4, in F
2 generation, 50% males are Muller-5 in phenotype and remaining 50% are wild type. If a lethal mutation is induced in X-chromosome of irradiated male no wild type males would appear in F
2 generation. Therefore, absence of wild type males in F
2 is an indication of an induced lethal mutation. Using different doses of irradiation and working out the corresponding frequencies of lethal mutations, a linear relationship could be observed between the radiation dose and mutation frequency (Fig. 21.5). When different kinds of radiations were used, it was found that alpha (α) particles gave the lowest mutation frequencies (.0084 per individual per 1000 roentgen) and beta (β), gamma (γ) and hard X-rays gave the highest frequency (.029).

Fig. 21.4. Muller-5 method for detection of sex linked mutations in Drosophila.

Fig. 21.5. A linear relationship between the frequency of sex linked lethal mutations and the dose of radiation in kilo Roentgens (kR)
Detection of sex linked visible mutations Attached X-method. The methods described in the preceding section were meant for detection of sex linked lethals. For detection of sex linked visible mutations, Muller-5 and attached X-chromosomes were used. The attached X females (XXY) have a special advantage. When these females are crossed to an irradiated male, X-chromosome of irradiated male goes either to superfemale daughters or to the sons. Since in sons there is a single X-chromosome, any visible induced mutation will immediately express itself and can be easily scored (Fig. 21.6).

Fig. 21.6. Attached X-method for detection of sex linked visible mutations.
Detection of autosomal mutations (lethal or visible)
Balanced lethal system and detection of mutations. Detection of mutations on autosomes (chromosomes other than sex chromosomes) makes use of a balanced lethal stock. Balanced lethal stock is one which carries recessive lethal alleles on two specific homologous chromosomes, their normal allele being present on its homologue; consequently homozygous individuals will not survive. The concept of balanced lethal would be better understood using a specific example. For detection of visible mutations in
Drosophila, a stock carried the dominant genes
Cy (
curly wing)and
L (
lobed eye)on one chromosome and
Pm (
plum eye i.e. brownish eye) on the other homologue so that the organism can be designated as
Cy L/Pm. Phenotype of the stock which was heterozygous for
Cy, L and
Pm, was curly, lobe and plum. If such individuals are crossed among themselves, progeny is always heterozygous, because each of the two homologous chromosomes carried recessive lethal genes (Fig. 21.7). Through the presence of inversion, crossing over was also suppressed, so that
Cy and/or
L may not be transferred to the chromosome carrying
Pm and vice versa.
For detection of an autosomal mutation,
Cy L/Pm stock is crossed to irradiated fly. From F
1,
Cy L flies were again backcrossed to
Cy LIPm in order to obtain curly, lobe (heterozygotes) male and female flies carrying the same autosome to be tested. Such
Cy L heterozygotes when crossed among themselves would give 1
CyL/CyL : 2 CyL/++ : 1 ++/++. Since
CyL homozygotes are lethal, phenotypic ratio will be 2 : 1. However, if lethal mutation was induced, only curly will appear. If a recessive visible mutation was induced, this will appear in homozygous state in one-third of the progeny (Fig. 21.8).
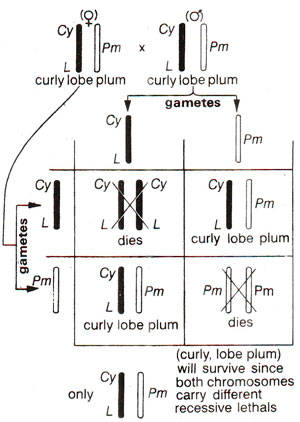
Fig. 21.7. Balanced lethal system in Cy LIPm Drosophila.
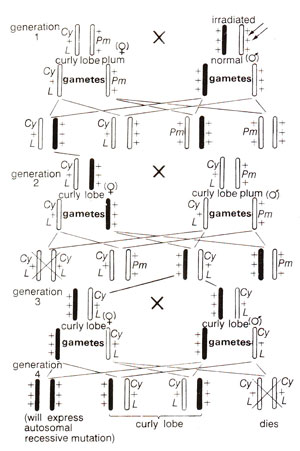
Fig. 21.8. Detection of autosomal mutations in Drosophila.












