A distinction should be made between monoploidy and haploidy.
Monoploids have a single basic set of chromosomes e.g. 2
n =
x = 7 in barley or 2
n =
x = 10 in corn. Haploids, on the other hand represent individuals having half the somatic chromosome number found in normal individual. Therefore, individuals having 2
n =
3x = 21 in wheat would also be haploids. These latter kind of haploids obtained from polyploids are often called
polyhaploids in order to distinguish them from monoploids.
While reviewing the work on haploids in flowering plants in 1963,
G. Kimber and
R. Riley of Plant Breeding Institute, Cambridge, England (now, Institute of Plant Science Research or IPSR, Norwich, UK), gave a classification for haploids. They classified haploids in
euhaploids and
aneuhaploids which as the terms indicate are derived from euploids and aneuploids respectively. A modified classification recently given by K.J.
Kasha is presented in Figure 20.6.
Euhaploids will include
monoploids as well as
polyhaploids.
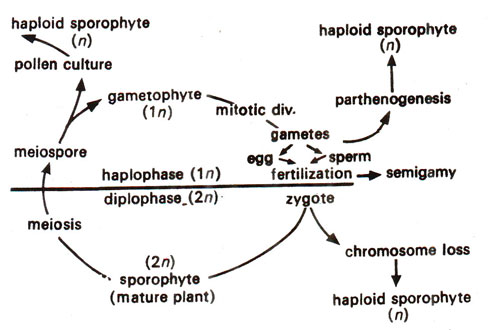
Fig. 20.7. Life cycle of a flowering plant illustrating different stages at which haploids can be produced (after Kasha, 1983).
Origin and production of haploids. Haploids in some cases as in male insects (Hymenoptera) are found as a routine and are produced due to parthenogenesis. In these insects, queen and drones are diploid females. Haploids may also originate spontaneously due to parthenogenetic development of egg in flowering plants. Such rare haploids have actually been obtained in tomatoes and cotton under cultivation.
Rarely haploids may originate from pollen tube rather than form egg, synergids or antipodals of embryo sac. These haploids will be called
androgenic haploids. Haploids can be artificially produced by any one of the following methods: (i) X-rays treatment, (ii) delayed pollination, (iii) temperature shocks, (iv) colchicine treatment, (v) distant (interspecific or intergeneric) hybridization, (vi) anther or pollen culture.
Among different available methods for haploid production as listed above, the most important are distant hybridization and anther culture. Thus haploids can be produced at different points in the life cycle of a flowering plant. The details of methods used are given in a life cycle shown in Figure 20.7. It may be noticed that haploids may originate due to : (i) parthenogenetic or androgenic development of gametes, (ii) due to chromosome loss in hybrid embryos and (iii) by pollen culture.

Fig. 20.7. Life cycle of a flowering plant illustrating different stages at which haploids can be produced (after Kasha, 1983).
Interspecific crosses in genera of
Solanaceae, (e.g.
Solarium and
Nicotiana)have been successfully used for production of haploids, both parthenogenetic and androgenic. Haploids by this method have also been produced in potato in large number. K.J.
Kasha from University of Guelph, Canada evolved an excellent technique for producing haploids in large number in barley (Kasha and Kao, 1970). He discovered that if the cross
Hardeum vulgare x
H. bulbosum, is made, chromosomes of
H. bulbosum are eliminated in early zygotic divisions, so that a few days after pollination, embryos can be cultured to get haploids. This technique is being extensively utilized now all over the world.
The production of haploids by anther and pollen culture was demonstrated for the first time in the laboratory of S.C.
Maheshwari of Delhi University (Guha and Maheshwari, 1970). Subsequently haploids by this technique could be produced in other plant genera e.g.
Oryza (rice) and
Nicotiana (tobacco). This technique is now considered to be a very potential source of haploid production.
Morphology of
haploids. Haploids are characterized morphologically by a reduction in size of all vegetative and floral parts. Only rarely a haploid may equal or exceed the diploid in size. When haploids were studied in
Nicotiana by
D. Kostoff, it was found that leaves, flowers, and overall plant size were smaller. The seed size, stomata size and pollen diameter were also generally smaller in haploids than in the diploids. In many cases in haploids, the size of nucleus measured as nuclear volume was half that in diploids.
Cytology of haploids. Since in a haploid set, the chromosomes are non- homologous and have no homologues to pair with, they are found as univalents at metaphase I of meiosis. Consequently, these univalents distribute at random during anaphase I. For instance a haploid in maize (2n = 20) will have 10 chromosomes and the number of chromosomes in a gamete of haploid plant can range from 0-10. Consequently, considerable sterility will be found. Moreover, since univalents are scattered all over the cell, they may constitute a restitution nucleus including all chromosomes and may thus give rise to gametes having a complete haploid set of chromosomes. Haploids (polyhaploids; 2
n = 3x = 21) were used by E.R. Sears for production of monosomics by pollinating the haploid by pollen from a diploid individual (hexaploid; 2
n = 6x = 42). If the egg has a chromosome number, one less than the complete set, this will result into a monosomic.
Uses of
haploids. When the chromosome number of a haploid is doubled by colchicines treatment, derived diploid will be completely homozygous for all genes so that the most important use of haploids is in the production of homozygous diploids. Although this theoretical expectation was earlier used only for the production of experimental material, but during the last few years a number of varieties for cultivation have been produced using this method. A number of such cultivars, were listed by K.J. Kasha in a section written in the genetics section'
Cytogenetics of Crop Plants' published by McMillan India Ltd. in 1983. Some of these varieties produced as homozygous diploids derived from haploids are listed in Table 20.2. The most extensive use of haploids for developing superior cultivars has been made in rice crop in China. The details of some rice varieties developed from pollen in China are presented in Table 20.3.








