Phenylalanine-derived Alkaloids
Whilst the aromatic amino acid L-tyrosine is
a common and extremely important precursor of alkaloids, L-phenylalanine is
less frequently utilized, and usually it contributes
only carbon atoms, e.g. C6C3, C6C2, or C6C1
units, without providing a nitrogen atom from its
amino group. Ephedrine (Figure 117), the main
alkaloid in species of Ephedra* (Ephedraceae) and
a valuable nasal decongestant and bronchial dilator,
is a prime example. Whilst ephedrine contains
the same carbon and nitrogen skeleton as
seen in phenylalanine, and L-phenylalanine is a precursor, only seven carbons, a C6C1 fragment,
are actually incorporated. It is found that phenylalanine
is metabolized, probably through cinnamic
acid to benzoic acid, and
this, perhaps as its coenzyme A ester, is acylated
with pyruvate, decarboxylation occurring during
the addition (Figure 117). The use of pyruvate as a nucleophilic reagent in this way is unusual in secondary metabolism, but occurs in primary metabolism during isoleucine and valine biosynthesis. |
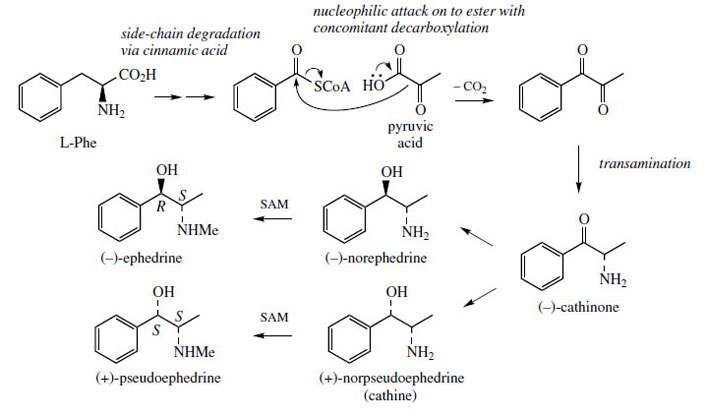 |
| Figure 117 |
A thiamine PP-mediated mechanism is suggested (Figure 118; compare decarboxylation of pyruvate, page 21, and formation of deoxyxylulose phosphate, page 170). This process yields the diketone, and a transamination reaction would then give cathinone (Figure 115).
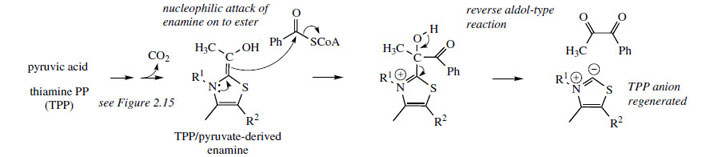 |
| Figure 118 |
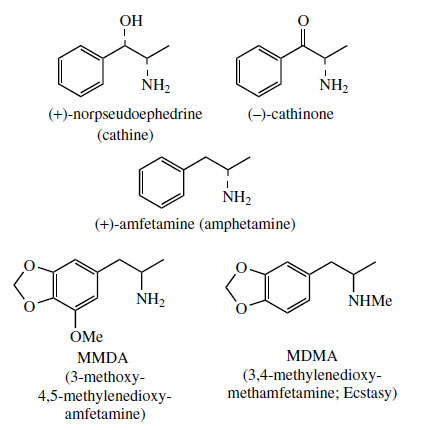
Reduction of the carbonyl group from either face provides the diastereomeric norephedrineor norpseudoephedrine (cathine). Finally, Nmethylation would provide ephedrine or pseudoephedrine(Figure 117). Typically, all four of the latter compounds can be found in Ephedraspecies, the proportions varying according to species. Norpseudoephedrine is also a major constituent of the leaves of khat* (Catha edulis; Celastraceae), chewed in African and Arab countries as a stimulant. Most of the CNS stimulant action comes from the more active cathinone, the corresponding carbonyl derivative. These natural compounds are structurally similar to the synthetic amfetamine/ dexamfetamine; (amphetamine/dexamphetamine) (Figure 119) and have similar properties.
 |
| Figure 119 |
Ephedra
Ephedra or Ma Huang is one of the oldest known drugs, having being used by the Chinese for at least 5000 years. It consists of the entire plant or tops of various Ephedra species (Ephedraceae), including E. sinica and E. equisetina from China, and E. geriardiana, E. intermedia and E. major from India and Pakistan. The plants are small bushes with slender aerial stems and minute leaves, giving the appearance of being effectively leafless. The plants typically contain 0.5-2.0% of alkaloids, according to species, and from 30-90% of the total alkaloids is (-)-ephedrine (Figure 117). Related structures, including the diastereoisomeric (+)-pseudoephedrine and the demethyl analogues (-)-norephedrine and (+)-norpseudoephedrine (Figure 117) are also present. In E. intermedia, the proportion of pseudoephedrine exceeds that of ephedrine.
Ephedrine is an indirectly acting sympathomimetic amine with effects similar to noradrenaline. Lacking the phenolic groups of the catecholamines, it has onlyweak action on adrenoreceptors, but it is able to displace noradrenaline from storage vesicles in the nerve terminals, which can then act on receptors. It is orally active and has a longer duration of action than noradrenaline. It also has bronchodilator activity, giving relief in asthma, plus a vasoconstrictor action on mucous membranes, making it an effective nasal decongestant. Pseudoephedrine is also widely used in compound cough and cold preparations and as a decongestant. The ephedrine and pseudoephedrine used medicinally are usually synthetic. One commercial synthesis of ephedrine involves a fermentation reaction on benzaldehyde using brewer's yeast (Saccharomyces sp.), giving initially an alcohol, then reductive condensation with methylamine yields (-)-ephedrine with very high enantioselectivity (Figure 120). The fermentation reaction is similar to that shown in Figure 118, in that an activated acetaldehyde bound to TPP is produced by the yeast by decarboxylation of pyruvate, and this unit is added stereospecifically to benzaldehyde in an aldol-like reaction.
The herbal drug ephedra/Ma Huang is currently being traded as 'herbal ecstasy'. Consumption gives CNS stimulation, but in high amounts can lead to hallucinations, paranoia, and psychosis.
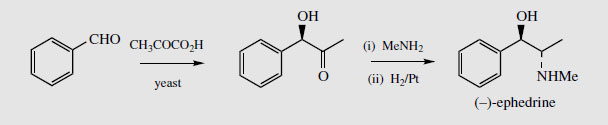 |
| Figure 120 |
Khat
Khat, or Abyssinian tea, consists of the fresh leaves of Catha edulis (Celastraceae), a small tree cultivated in Ethiopia, East and South Africa, and the Yemen. The leaves are widely employed in African and Arabian countries, where they are chewed for a stimulant effect. This traditional use alleviates hunger and fatigue, but also gives a sensation of general well-being (compare coca, page 302). Users become cheerful and talkative, and khat has become a social drug. Prolonged usage can lead to hypertension, insomnia, or even mania. Khat consumption may lead to psychological dependence, but not normally physical dependence. There is presently little usage outside of Africa and Arabia, although this is increasing due to immigration from these areas. However, for maximum effects, the leaves must be fresh, and this somewhat restricts international trade. Dried leaves contain up to 1% cathine ((+)-norpseudoephedrine (Figure 119)), but young fresh leaves contain (-)-cathinone (Figure 119) as the principal CNS stimulant. Cathinone has similar pharmacological properties as the synthetic CNS stimulant (+)-amfetamine/dexamfetamine (amphetamine/dexamphetamine) (Figure 119), with a similar potency. Both compounds act by inducing release of catecholamines.
Medicinal use of amfetamine has declined markedly as drug dependence and the severe depression generated on withdrawal have been appreciated. Nevertheless, amfetamine abuse is significant. Amfetamines are taken orally, sniffed, or injected to give a long period of CNS stimulation (hours to days). Users often then take a depressant drug (alcohol, barbiturates, opioids) to terminate the effects. Users rapidly become dependent and develop tolerance. The consumption of khat is not yet restricted in the UK, even though both cathine and cathinone are now controlled drugs. It remains to be seen whether khat will be reclassified and its use restricted in any way. Other amfetamine-like derivatives of note are methoxymethylenedioxyamfetamine (MMDA) and methylenedioxymethamfetamine (MDMA) (Figure 119). MMDA is thought to be formed in the body after ingestion of nutmeg (Myristica fragrans; Myristicaceae), by an amination process on myristicin, and it may be the agent responsible for the euphoric and hallucinogenic effects of nutmeg. MDMA is the illicit drug Ecstasy, a synthetic amfetamine-like stimulant popular among young people. The use of Ecstasy has resulted in a number of deaths, brought about by subsequent heatstroke and dehydration.
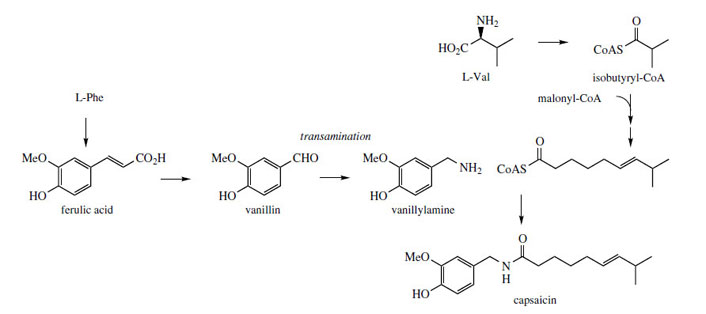 |
| Figure 121 |
The amide capsaicin (Figure 121) constitutes the powerfully pungent principle in chilli peppers (Capsicum annuum; Solanaceae). Apart from its culinary importance, it is also used medicinally in creams to counter neuralgia caused by herpes infections and in other topical pain-relieving preparations. The initial burning effect of capsaicin is found to affect the pain receptors, making them less sensitive. The aromatic portion of capsaicin is derived from phenylalanine through ferulic acid and vanillin (Figure 121, compare page 141), this aldehyde being the substrate for transamination to give vanillylamine. The acid portion of the amide structure is of polyketide origin, with a branched-chain fatty acyl-CoA being produced by chain extension of isobutyryl-CoA. This starter unit is valine derived.




