Pyrrolidine and Tropane Alkaloids
Simple pyrrolidine-containing alkaloid structures are exemplified by hygrine and cuscohygrine, found in those plants of the Solanaceae that accumulate medicinally valuable tropane alkaloids such as hyoscyamine or cocaine ( Figure 3). The pyrrolidine ring system is formed initially as a Δ1-pyrrolinium cation (Figure 2). PLP-dependent decarboxylation of ornithine gives putrescine, which is then methylated to N-methylputrescine. Oxidative deamination of N-methylputrescine by the action of a diamine oxidase gives the aldehyde, and Schiff base (imine) formation produces the N-methyl-Δ1-pyrrolinium cation. Indeed, the aminoaldehyde in aqueous solution is known to exist as an equilibrium mixture with the Schiff base. An alternative sequence to putrescine starting from arginine also operates concurrently as indicated in Figure 2. The arginine pathway also involves decarboxylation, but requires additional hydrolysis reactions to cleave the guanidine portion.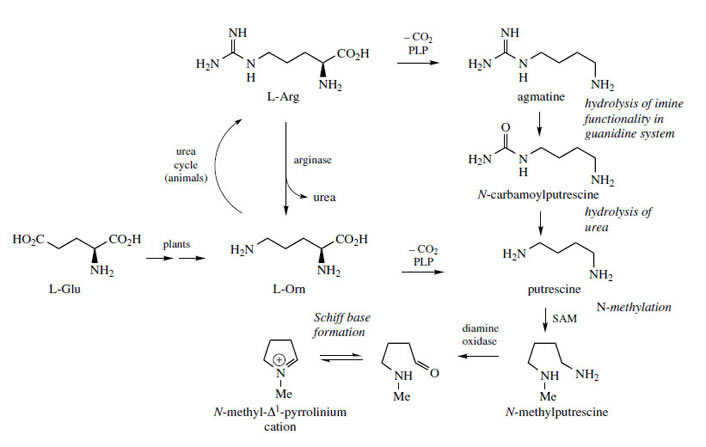 |
| Figure 2 |
 |
| Figure 3 |
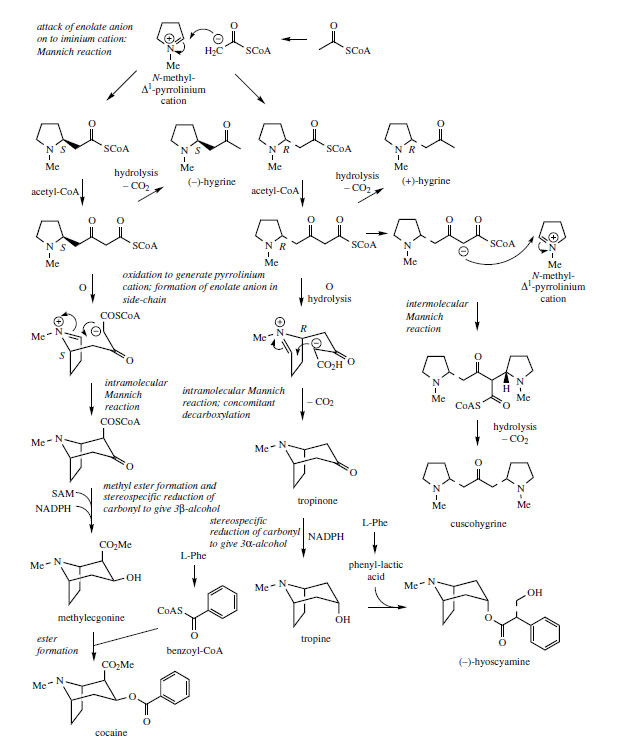
The extra carbon atoms required for hygrine formation are derived from acetate via acetyl-CoA, and the sequence appears to involve stepwise addition of two acetyl-CoA units (Figure 3). In the first step, the enolate anion from acetyl-CoA acts as nucleophile towards the pyrrolinium ion in a Mannich-like reaction, which could yield products with either R or S stereochemistry. The second addition is then a Claisen condensation extending the side-chain, and the product is the 2-substituted pyrrolidine, retaining the thioester group of the second acetyl-CoA. Hygrine and most of the natural tropane alkaloids lack this particular carbon atom, which is lost by suitable hydrolysis/decarboxylation reactions. The bicyclic structure of the tropane skeleton in hyoscyamine and cocaine is achieved by a repeat of the Mannich-like reaction just observed. This requires an oxidation step to generate a new ?1-pyrrolinium cation, and removal of a proton a to the carbonyl. The intramolecular Mannich reaction on the R enantiomer accompanied by decarboxylation generates tropinone, and stereospecific reduction of the carbonyl yields tropine with a 3a- hydroxyl. Hyoscyamine is the ester of tropine with (S )-tropic acid (Figure 4), which is derived from L-phenylalanine. A novel rearrangement process occurs in the phenylalanine ? tropic acid transformation, in which the carboxyl group apparently migrates to the adjacent carbon (Figure 4). Phenylpyruvic acid and phenyl-lactic acid have been shown to be involved and tropine becomes esterified with phenyl-lactic acid (as the coenzyme-A ester) to form littorine before the rearrangement occurs. The mechanism of this rearrangement has yet to be proven, though a free radical process (Figure 4) with an intermediate cyclopropane-containing radical would accommodate the available data. Further modifications to the tropane skeleton then occur on the ester, and not on the free alcohol. These include hydroxylation to 6�-hydroxyhyoscyamine and additional oxidation allowing formation of an epoxide grouping as in hyoscine (scopolamine). Both of these reactions are catalysed by a single 2-oxoglutarate-dependent dioxygenase. Other esterifying acids may be encountered in tropane alkaloid structures, e.g. tiglic acid in meteloidine (Figure 5) from Datura meteloides and phenyl-lactic acid in littorine, above which is a major alkaloid in Anthocercis littorea. Tiglic acid is known to be derived from the amino acid L-isoleucine. The structure of cuscohygrine arises by an intermolecular Mannich reaction involving a second N-methyl-?1-pyrrolinium cation (Figure 3).
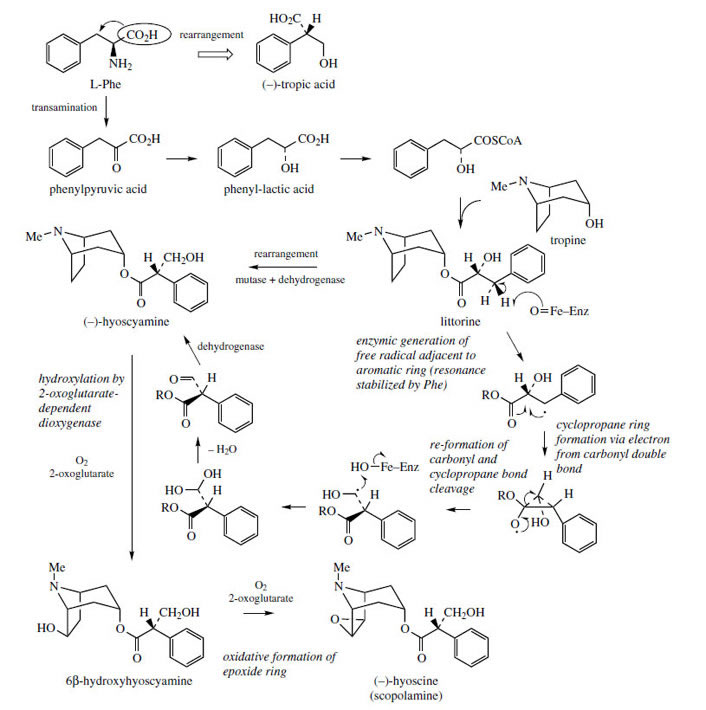 |
| Figure 4 |
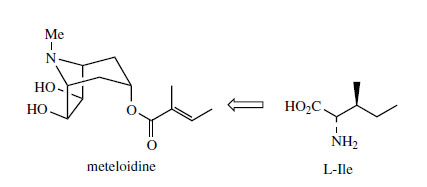 |
| Figure 5 |
Belladonna
The deadly nightshade Atropa belladonna (Solanaceae) has a long history as a highly poisonous plant. The generic name is derived from Atropos, in Greek mythology the Fate who cut the thread of life. The berries are particularly dangerous, but all parts of the plant contain toxic alkaloids, and even handling of the plant can lead to toxic effects since the alkaloids are readily absorbed through the skin. Although humans are sensitive to the toxins, some animals, including sheep, pigs, goats, and rabbits, are less susceptible. Cases are known where the consumption of rabbits or birds that have ingested belladonna has led to human poisoning. The plant is a tall perennial herb producing dull-purple bell-shaped flowers followed by conspicuous shiny black fruits, the size of a small cherry. Atropa belladonna is indigenous to Central and Southern Europe, though it is not especially common. It is cultivated for drug use in Europe and the United States. The tops of the plant are harvested two or three times per year and dried to give belladonna herb. Roots from plants some 3–4 years old are less commonly employed as a source of alkaloids.
Belladonna herb typically contains 0.3–0.6% of alkaloids, mainly (-)-hyoscyamine (Figure 4). Belladonna root has only slightly higher alkaloid content at 0.4–0.8%, again mainly (-)-hyoscyamine. Minor alkaloids including (-)-hyoscine (Figure 4) and cuscohygrine (Figure 3) are also found in the root, though these are not usually significant in the leaf. The mixed alkaloid extract from belladonna herb is still used as a gastrointestinal sedative, usually in combination with antacids. Root preparations can be used for external pain relief, e.g. in belladonna plasters.
Stramonium
Datura stramonium (Solanaceae) is commonly referred to as thornapple on account of its spikey fruit. It is a tall bushy annual plant widely distributed in Europe and North America, and because of its alkaloid content is potentially very toxic. Indeed, a further common name, Jimson or Jamestown weed, originates from the poisoning of early settlers near Jamestown, Virginia. At subtoxic levels, the alkaloids can provide mild sedative action and a feeling of well-being. In the Middle Ages, stramonium was employed to drug victims prior to robbing them. During this event, the victim appeared normal and was cooperative, though afterwards could usually not remember what had happened. For drug use, the plant is cultivated in Europe and South America. The leaves and tops are harvested when the plant is in flower. Stramonium leaf usually contains 0.2–0.45% of alkaloids, principally (-)-hyosycamine and (-)-hyoscine in a ratio of about 2:1. In young plants, (-)-hyoscine can predominate.
The generic name Datura is derived from dhat, an Indian poison used by the Thugs. The narcotic properties of Datura species, especially D. metel, have been known and valued in India for centuries. The plant material was usually absorbed by smoking. Most species of Datura contain similar tropane alkaloids and are potential sources of medicinal alkaloids. In particular, Datura sanguinea, a perennial of treelike stature with blood-red flowers, is cultivated in Ecuador, and yields leaf material with a high (0.8%) alkaloid content inwhich the principal component is (-)-hyoscine. The plants can be harvested several times a year. Datura sanguinea, and several other species of the tree-daturas (now classified as a separate genus Brugmansia) are widely cultivated as ornamentals, especially for conservatories, because of their attractive large tubular flowers. The toxic potential of these plants is not always recognized.
Hyosycamus
Hyoscyamus niger (Solanaceae), or henbane, is a European native with a long history as a medicinal plant. Its inclusion in mediaeval concoctions and its power to induce hallucinations with visions of flight may well have contributed to our imaginary view of witches on broomsticks. The plant has both annual and biennial forms, and is cultivated in Europe and North America for drug use, the tops being collected when the plant is in flower, and then dried rapidly. The alkaloid content of hyoscyamus is relatively low at 0.045–0.14%, but this can be composed of similar proportions of (-)-hyoscine and (-)-hyosycamine. Egyptian henbane, Hyosycamus muticus, has a much higher alkaloid content than H. niger, and although it has mainly been collected from the wild, especially from Egypt, it functions as a major commercial source for alkaloid production. Some commercial cultivation occurs in California. The alkaloid content of the leaf is from 0.35% to 1.4%, of which about 90% is (-)-hyoscyamine.
Duboisia
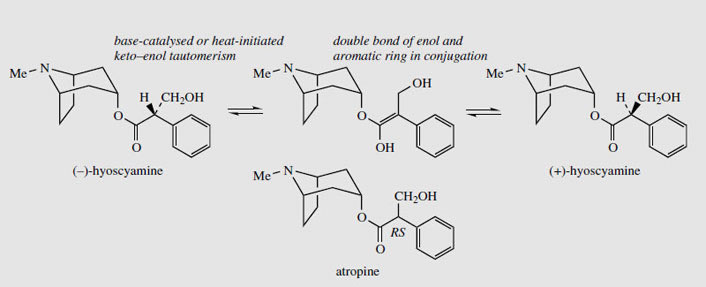
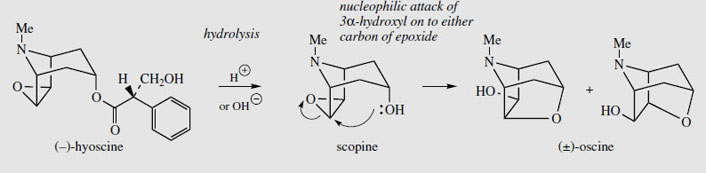
Duboisia is a small genus of trees, containing only three species, found in Australia, again from the family Solanaceae. Two of these, Duboisia myoporoides and D. leichhardtii are grown commercially in Australia for tropane alkaloid production. The small trees are kept as bushes to allow frequent harvesting, with up to 70-80% of the leaves being removed every 7-8 months. The alkaloid content of the leaf is high, up to 3% has been recorded, and it includes (-)-hyoscyamine, (-)-hyoscine, and a number of related structures. The proportion of hyoscyamine to hyoscine varies according to the species used, and the area in which the trees are grown. The hyoscine content is frequently much higher than that of hyoscyamine. Indeed, interest in Duboisia was very much stimulated by the demand for hyoscine as a treatment for motion sickness in military personnel in the Second World War. Even higher levels of alkaloids, and higher proportions of hyoscine, can be obtained from selected D. myoporoides x D. leichhardtii hybrids, which are currently cultivated. The hybrid is superior to either parent, and can yield 1-2.5% hyoscine and 0-1% hyoscyamine. Duboisialeaf is an important commercial source of medicinal tropane alkaloids. The third species of Duboisia, D. hopwoodi, contains little tropane alkaloid content, but produces mainly nicotine and related alkaloids, e.g. nornicotine. Leaves of this plant were chewed by aborigines for their stimulating effects. Allied Drugs Tropane alkaloids, principally hyoscyamine and hyoscine, are also found in two other medicinal plants, scopolia and mandrake, but these plants find little current use. Scopolia (Scopolia carniolica; Solanaceae) resembles belladonna in appearance, though it is considerably smaller. Both root and leaf materials have been employed medicinally. The European mandrake (Mandragora officinarum; Solanaceae) has a complex history as a hypnotic, a general panacea, and an aphrodisiac. Its collection has been surrounded by much folklore and superstition, in that pulling it from the ground was said to drive its collector mad due to the unearthly shrieks emitted. The roots are frequently forked and are loosely likened to a man or woman. Despite the Doctrine of Signatures, which teaches that the appearance of an object indicates its special properties, from a pharmacological point of view, this plant would be much more efficient as a pain-reliever than as an aphrodisiac. Hyoscyamine, Hyoscine and Atropine All the above solanaceous plants contain as main alkaloidal constituents the tropane esters (-)-hyoscyamine and (-)-hyoscine, together with other minor tropane alkaloids. The piperidine ring in the bicyclic tropane system has a chairlike conformation, and there is a ready inversion of configuration at the nitrogen atom so that the N-methyl group can equilibrate between equatorial and axial positions (Figure 6). An equatorial methyl is strongly favoured provided there are no substituents on the two-carbon bridge, in which case the axial form may predominate. (-)-Hyoscyamine is the ester of tropine (Figure 4) with (-)- (S)-tropic acid, whilst (-)-hyoscine contains scopine (Figure 8) esterified with (-)-(S)-tropic acid. The optical activity of both hyoscyamine and hyoscine stems from the chiral centre in the acid portion, (S)-tropic acid. Tropine itself, although containing chiral centres, is a symmetrical molecule and is optically inactive; it can be regarded as a meso structure. The chiral centre in the tropic acid portion is adjacent to a carbonyl and the aromatic ring, and racemization can be achieved under mild conditions by heating or treating with base. This will involve an intermediate enol (or enolate) which is additionally favoured by conjugation with the aromatic ring (Figure 7). Indeed, normal base assisted fractionation of plant extracts to isolate the alkaloids can sometimes result in production of significant amounts of racemic alkaloids. The plant material itself generally contains only the enantiomerically pure alkaloids. Hyoscyamine appears to be much more easily racemized than hyoscine. Hydrolysis of the esters using acid or base usually gives racemic tropic acid. Note that littorine (Figure 4), in which the chiral centre is not adjacent to the phenyl ring, is not readily racemized, and base hydrolysis gives optically pure phenyl-lactic acid. The racemic form of hyoscyamine is called atropine (Figure 7), whilst that of hyoscine is called atroscine. In each case, the biological activity of the (+)-enantiomer is some 20-30 times less than that of the natural (-)-form. Chemical hydrolysis of hyoscine in an attempt to obtain the alcohol scopine is not feasible. Instead, the alcohol oscine is generated because of the proximity of the 3a-hydroxyl group to the reactive epoxide function (Figure 8).
|
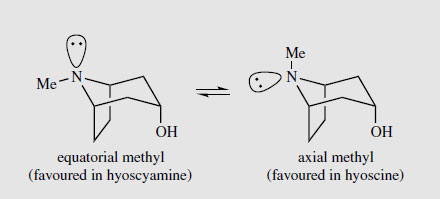 |
||||||
| Figure 6 | ||||||
|
Probably for traditional reasons, salts of both (-)-hyoscyamine and (±)-hyoscyamine (atropine) are used medicinally, whereas usage of hyoscine is restricted to the natural laevorotatory form. These alkaloids compete with acetylcholine for the muscarinic site of the parasympathetic nervous system, thus preventing the passage of nerve impulses, and are classified as anticholinergics. Acetylcholine binds to two types of receptor site, described as muscarinic or nicotinic, from the specific triggering of a response by the Amanita muscariaalkaloid muscarine or the tobacco alkaloid nicotine respectively. The structural similarity between acetylcholine and muscarine (Figure 9) can readily be appreciated, and hyoscyamine is able to occupy the same receptor site by virtue of the spatial relationship between the nitrogen atom and the ester linkage (Figure 9). The side-chain also plays a role in the binding, explaining the difference in activities between the two enantiomeric forms. The agonist properties of hyoscyamine and hyoscine give rise to a number of useful effects, including antispasmodic action on the gastrointestinal tract, antisecretory effect controlling salivary secretions during surgical operations, and as mydriatics to dilate the pupil of the eye. Hyoscine has a depressant action on the central nervous system and finds particular use as a sedative to control motion sickness. One of the side-effects from oral administration of tropane alkaloids is dry mouth (the antisecretory effect) but this can be much reduced by transdermal administration. In motion sickness treatment, hyoscine can be supplied via an impregnated patch worn behind the ear. Hyoscine under its synonym scopolamine is also well known, especially in fiction, as a ‘truth drug’. This combination of sedation, lack of will, and amnesia was first employed in child-birth, giving what was termed ‘twilight sleep’, and may be compared with the mediaeval use of stramonium. The mydriatic use also has a very long history. Indeed, the specific name belladonna for deadly nightshade means ‘beautiful lady’ and refers to the practice of ladies at court who applied the juice of the fruit to the eyes, giving widely dilated pupils and a striking appearance, though at the expense of blurred vision through an inability to focus. Atropine also has useful antidote action in cases of poisoning caused by cholinesterase inhibitors, e.g. physostigmine and neostigmine and organophosphate insecticides.
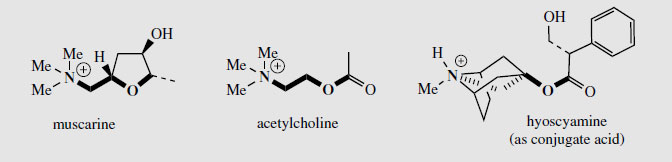 |
| Figure 9 |
It is valuable to reiterate here that the tropane alkaloid-producing plants are all regarded as very toxic, and that since the alkaloids are rapidly absorbed into the blood stream, even via the skin, first aid must be very prompt. Initial toxicity symptoms include skin flushing with raised body temperature, mouth dryness, dilated pupils, and blurred vision.
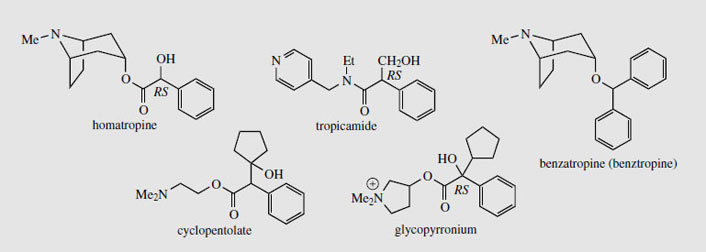 |
| Figure 10 |
Homatropine (Figure 10) is a semi-synthetic ester of tropine with racemic mandelic (2- hydroxyphenylacetic) acid and is used as amydriatic, as are tropicamide and cyclopentolate(Figure 10). Tropicamide is an amide of tropic acid, though a pyridine nitrogen is used to mimic that of the tropane. Cyclopentolate is an ester of a tropic acid-like system, but uses a non-quaternized amino alcohol resembling choline. Glycopyrronium (Figure 10) has a quaternized nitrogen in a pyrrolidine ring, with an acid moiety similar to that of cyclopentolate. This drug is an antimuscarinic used as a premedicant to dry bronchial and salivary secretions. Hyoscine butylbromide (Figure 11) is a gastro-intestinal antispasmodic synthesized from (-)-hyoscine by quaternization of the amine function with butyl bromide. The quaternization of tropane alkaloids by N-alkylation proceeds such that the incoming alkyl group always approaches from the equatorial position. The potent bronchodilator ipratropium bromide(Figure 11) is thus synthesized from noratropine by successive isopropyl and methyl alkylations whilst oxitropium bromide is produced from norhyoscine by N-ethylation and then N-methylation. Both drugs are used in inhalers for the treatment of chronic bronchitis.
Benzatropine (benztropine) (Figure 10) is an ether of tropine used as an antimuscarinic drug in the treatment of Parkinson’s disease. It is able to inhibit dopamine reuptake, helping to correct the deficiency which is characteristic of Parkinsonism.
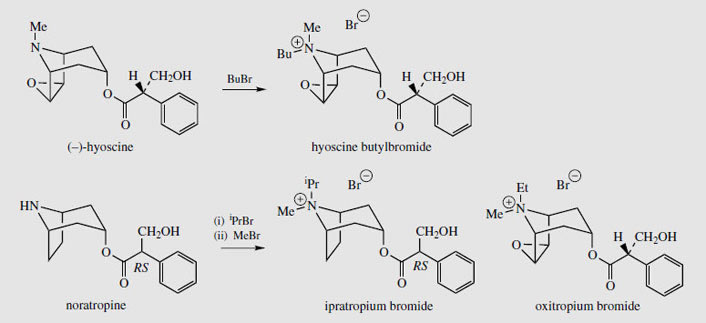 |
| Figure 11 |
The calystegines (Figure 12) are a group of recently discovered, water-soluble, polyhydroxy nortropane derivatives that are found in the leaves and roots of many of the solanaceous plants, including Atropa, Datura, Duboisia, Hyoscyamus, Mandragora, Scopolia, and Solanum. They were first isolated from Calystegia sepium (Convolvulaceae). These compounds, e.g. calystegin A3 and calystegin B2 (Figure 12), are currently of great interest as glycosidase inhibitors and have similar potential as the polyhydroxyindolizidines such as castanospermine and the aminosugars such as deoxynojirimycin in the development of drugs with activity against the AIDS virus HIV. It is likely that these alkaloids are produced by a similar pathway to that which yields tropine, but the stereochemistry of reduction of tropinone (or nortropinone) yields the 3�-alcohol, and further hydroxylation steps are necessary. Examples of tri-, tetra-, and penta-hydroxy calystegines are currently known.
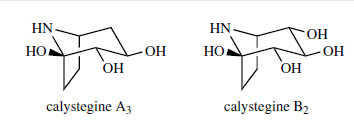 |
| Figure 12 |
Cocaine (Figure 3) is a rare alkaloid restricted to some species of Erythroxylum (Erythroxylaceae). Erythroxylum coca (coca)* is the most prominent as a source of cocaine, used medicinally as a local anaesthetic, and as an illicit drug for its euphoric properties. Coca also contains significant amounts of cinnamoylcocaine (cinnamylcocaine) (Figure 13), where cinnamic acid rather than benzoic acid is the esterifying acid, together with some typical tropine derivatives without the extra carboxyl, e.g. tropacocaine (Figure 13). Tropacocaine still retains the 3�-configuration, showing that the stereospecific carbonyl reduction is the same as with the cocaine route, and not as with the hyoscyamine pathway.
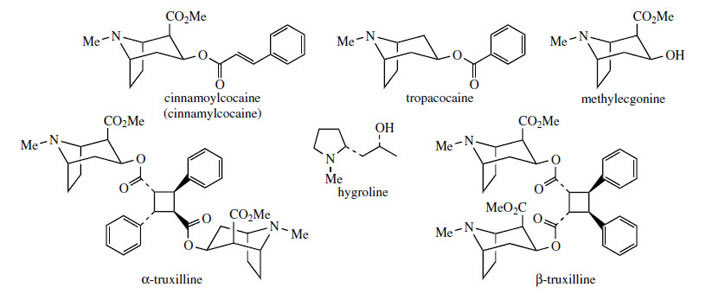 |
| Figure 13 |
Coca
Coca leaves are obtained from species of Erythroxylum (Erythroxylaceae), small shrubs native to the Andes region of South America, namely Colombia, Ecuador, Peru, and Bolivia. Plants are cultivated there, and in Indonesia. Two main species provide drug materials, Erythroxylumcoca (Bolivian or Huanaco coca) and E. truxillense (Peruvian or Truxillo coca). Cultivated plants are kept small by pruning and a quantity of leaves is harvested from each plant three or more times per year. Coca-leaf chewing has been practised by South American Indians for many years and is an integral part of the native culture pattern. Leaf is mixed with lime, this liberating the principal alkaloid cocaine as the free base, and the combination is then chewed. Cocaine acts as a potent antifatigue agent, and this allows labourers to ignore hunger, fatigue, and cold, enhancing physical activity and endurance. Originally the practice was limited to the Inca high priests and favoured individuals, but became widespread after the Spanish conquest of South America. It is estimated that 25% of the harvest is consumed in this way by the local workers, who may each use about 50 g of leaf per day (= 350 mg cocaine). Only a tiny amount (1-2%) of the coca produced is exported for drug manufacture. The rest contributes to illicit trade and the world's drug problems. Efforts to stem the supply of illicit coca and cocaine have been relatively unsuccessful. Coca leaf contains 0.7-2.5% of alkaloids, the chief component (typically 40-50%) of which is (-)-cocaine (Figure 3), a diester of (-)-ecgonine. Note that although tropine is an optically inactive meso structure, ecgonine contains four chiral centres, is no longer symmetrical, and is therefore optically active. Cinnamoylcocaine (cinnamylcocaine), a-truxilline, �-truxilline, and methylecgonine (Figure 13) are minor constituents also based on ecgonine. The truxillines contain dibasic acid moieties, a-truxillic and �-truxinic acids, which are cycloaddition products from two cinnamic acid units (Figure 14). Other alkaloids present include structures based on f-tropine (the 3�-isomer of tropine) such as tropacocaine (Figure 13) and on hygrine, e.g. hygrine, hygroline (Figure 13) and cuscohygrine (Figure 3). Cuscohygrine typically accounts for 20-30% of the alkaloid content. Illegal production of cocaine is fairly unsophisticated, but can result in material of high quality. The alkaloids are extracted from crushed leaf using alkali (lime) and petrol. The petrol extract is then re-extracted with aqueous acid, and this alkaloid fraction is basified and allowed to stand, yielding the free alkaloid as a paste. Alternatively, the hydrochloride or sulphate salts may be prepared. The coca alkaloids are often diluted with carrier to give a preparation with 10-12% of cocaine. The illicit use of cocaine and cocaine hydrochloride is a major problem worldwide. The powder is usually sniffed into the nostrils, where it is rapidly absorbed by the mucosa, giving stimulation and short-lived euphoria through inhibiting reuptake of neurotransmitters dopamine, noradrenaline, and serotonin, so prolonging and augmenting their effects. Regular usage induces depression, dependence, and damage to the nasal membranes. The drug may also be injected intravenously, or the vapour inhaled. For inhalation, the free base or 'crack' is employed to increase volatility. The vaporized cocaine is absorbed extremely rapidly and carried to the brain within seconds, speeding up and enhancing the euphoric lift. Taken in this form, cocaine has proved highly addictive and dangerous. Cocaine abuse is currently a greater problem than heroin addiction, and, despite intensive efforts, there is no useful antagonist drug available to treat cocaine craving and addiction.
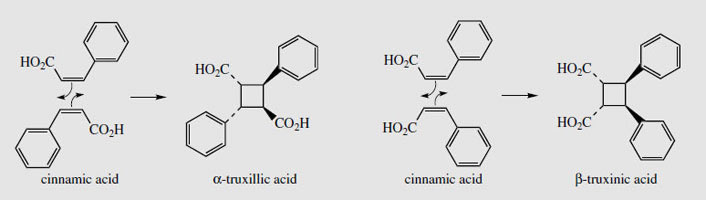 |
| Figure 14 |
In the 1800s, coca drinks were fashionable, and one in particular, Coca-Cola, became very popular. This was originally based on extracts of coca (providing cocaine) and cola (supplying caffeine), but although the coca content was omitted from 1906 onwards, the name and popularity continue. Medicinally, cocaine is of value as a local anaesthetic for topical application. It is rapidly absorbed by mucous membranes and paralyses peripheral ends of sensory nerves. This is achieved by blocking ion channels in neural membranes. It was widely used in dentistry, but has been replaced by safer drugs, though it still has applications in ophthalmic and ear, nose, and throat surgery. As a constituent of Brompton's cocktail (cocaine and heroin in sweetened alcohol) it is available to control pain in terminal cancer patients. It increases the overall analgesic effect, and its additional CNS stimulant properties counteract the sedation normally associated with heroin. The essential functionalities of cocaine required for activity were eventually assessed to be the aromatic carboxylic acid ester and the basic amino group, separated by a lipophilic hydrocarbon chain. Synthetic drugs developed from the cocaine structure have been introduced to provide safer, less toxic local anaesthetics (Figure 15). Procaine, though little used now, was the first major analogue employed. Benzocaine is used topically, but has a short duration of action. Tetracaine (amethocaine), oxybuprocaine, and proxymetacaineare valuable local anaesthetics employed principally in ophthalmic work. The ester function can be replaced by an amide, and this gives better stability towards hydrolysis in aqueous solution or by esterases. Lidocaine (lignocaine) is an example of an amino amide analogue and is perhaps the most widely used local anaesthetic, having rapid action, effective absorption, good stability, and may be used by injection or topically. Other amino amide local anaesthetic structures include prilocaine, with similar properties to lidocaine and very low toxicity, and bupivacaine, which has a long duration of action. Ropivacaine, mepivacaine, and articaine(carticaine) are some recently introduced amide-type local anaesthetics, the latter two being used predominantly in dentistry. Cinchocaine is often incorporated into preparations to soothe haemorrhoids. Lidocaine, although introduced as a local anaesthetic, was subsequently found to be a potent antiarrhythmic agent, and it now finds further use as an antiarrhythmic drug, for treatment of ventricular arrhythmias especially after myocardial infarction. Other cocainerelated structures also find application in the same way, including tocainide, procainamide, and flecainide (Figure 15). Tocainide is a primary amine analogue of lidocaine, whilst procainamide is an amide analogue of procaine. In mexiletene, a congener of lidocaine, the amide group has been replaced by a simple ether linkage.
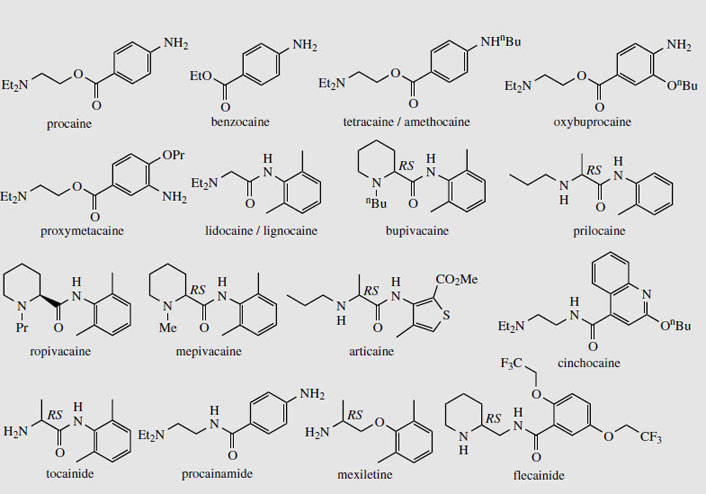 |
| Figure 15 |
Anatoxin-a (Figure 16) is a toxic tropanerelated alkaloid produced by a number of cyanobacteria, e.g. Anabaena flos-aquae and Aphanizomenon flos-aquae, species which proliferate in lakes and reservoirs during periods of hot, calm weather. A number of animal deaths have been traced back to consumption of water containing the cyanobacteria, and ingestion of the highly potent neurotoxin anatoxin-a, which has been termed Very Fast Death Factor. Anatoxina is a powerful agonist at nicotinic acetylcholine receptors, and has become a useful pharmacological probe. The ring system may be regarded as a homotropane, and it has been suggested that the pyrrolidine ring originates from ornithine via putrescine and ?1 pyrroline, in a way similar to the tropane alkaloids (Figure 16). The remaining carbons may originate from acetate precursors. A remarkable compound with a nortropane ring system has been isolated in tiny amounts from the highly coloured skin of the Ecuadorian poison frog Epipedobates tricolor. This compound, called epibatidine (Figure 17), is exciting considerable interest as a lead compound for analgesic drugs. It is 200-500 times more potent than morphine, and does not act by the normal opioid mechanism, but is a specific agonist at nicotinic acetylcholine receptors. Whether or not this structure is ornithine derived remains to be established.
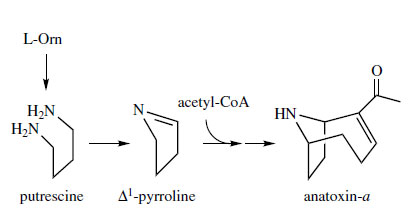 |
| Figure 16 |
 |
| Figure 17 |