Terpenoid Alkaloids
A variety of alkaloids based on mono-, sesqui-, di-,
and tri-terpenoid skeletons have been characterized,
but information about their formation in nature is
still somewhat sparse.Monoterpene alkaloids are in themain structurally related to iridoidmaterials , the oxygen heterocycle being replaced
by a nitrogen-containing ring. β-Skytanthine from
Skytanthus acutus (Apocynaceae) and actinidinefrom Actinidia polygama (Actinidiaceae) serve as
examples (Figure 122). The iridoid loganin, so
important in the biosynthesis of terpenoid indole
alkaloids and the ipecac alkaloids, is not a precursor of these structures,
and a modified series of reactions starting from
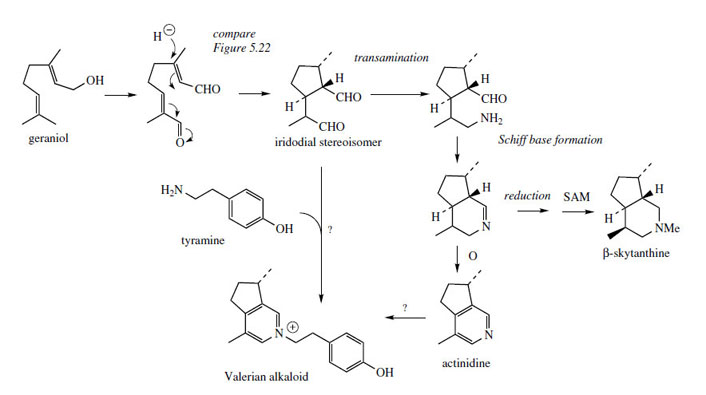
geraniol is proposed (Figure 122). The formation
of the dialdehyde follows closely elaboration of its
stereoisomer in loganin biosynthesis.
This could then act as a substrate for amination
via an amino acid, followed by ring formation as seen with coniine. |
Reduction and methylation would yield �-skytanthine, whereas further oxidation could provide the pyridine ring of actinidine. Valerian root (Valeriana officinalis; Valerianaceae) is known to contain alkaloids such as that shown in Figure 122, as well as iridoid structures. Whilst this alkaloid may be the result of N-alkylation on actinidine, an alternative pathway in which tyramine is condensed with the dialdehyde could be proposed. In the latter case, this alkaloid could be regarded as an alkaloid derived from tyrosine, rather than in this group of terpenoid alkaloids.
 |
| Figure 122 |
 |
| Figure 123 |
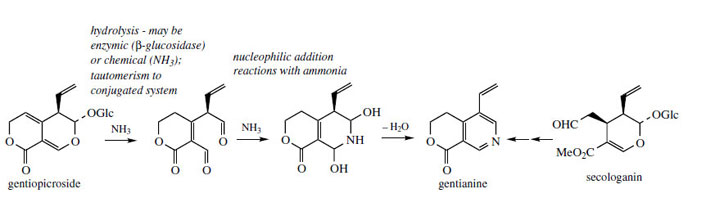
Gentianine (Figure 123) is probably the most common of the monoterpene alkaloids, but it is sometimes formed as an artefact when a suitable plant extract is treated with ammonia, the base commonly used during isolation of alkaloids. Thus, gentiopicroside from Gentiana lutea (Gentianaceae) is known to react with ammonia to give gentianine (Figure 123). Many other iridoid structures are known to react with ammonia to produce alkaloid artefacts. In some plants, gentianine can be found when no ammonia treatment has been involved, and one may speculate that loganin and secologanin may be precursors.
 |
| Figure 124 |
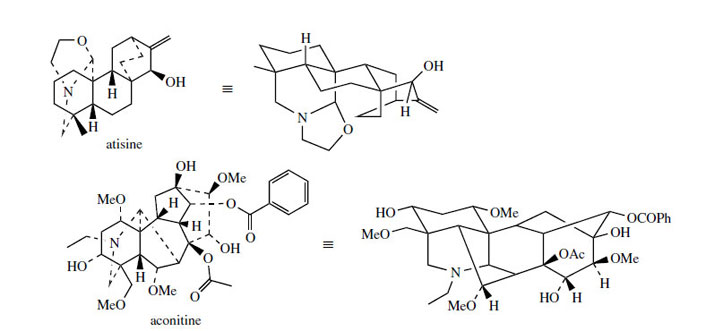
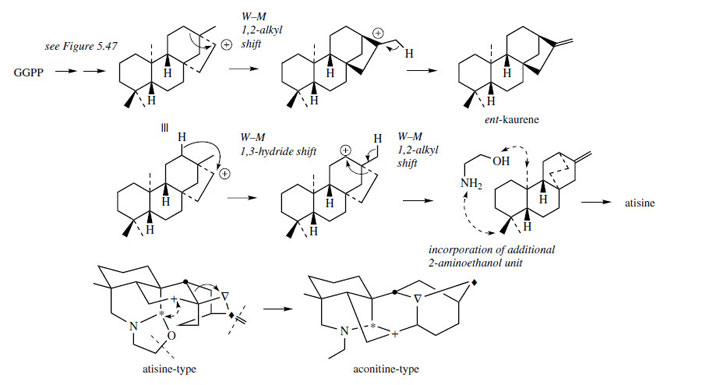
Perhaps the most important examples of terpenoid alkaloids from a pharmacological point of view are those found in aconite* or wolfsbane (Aconitum species; Ranunculaceae) and species of Delphinium (Ranunculaceae). Whilst Aconitum napellus has had some medicinal uses, plants of both genera owe their highly toxic nature to diterpenoid alkaloids. Aconite in particular is regarded as extremely toxic, due to the presence of aconitine (Figure 124) and related C19 norditerpenoid alkaloids. Species of Delphinium accumulate diterpenoid alkaloids such as atisine (Figure 124), which tend to be less toxic than aconitine. An appreciation of their structural relationship to diterpenes, e.g. ent-kaurene, is given in Figure 125, though little experimental evidence is available. It appears feasible that a pre-ent-kaurene carbocation undergoesWagner–Meerwein rearrangements, and that the atisine skeleton is then produced by incorporating an N−CH2CH2−O fragment (e.g. from 2-aminoethanol) to form the heterocyclic rings. The aconitine akeleton is probably formed from the atisine skeleton by further modifications as indicated. Note that a rearrangement process converts two fused six-membered rings into a (7 + 5)-membered bicyclic system, and that one carbon, that from the exocyclic double bond, is lost.
Aconite
Aconites, commonly called wolfsbane or monkshood, are species of Aconitum (Ranunculaceae), valued ornamental herbaceous plants, grown for their showy blue or purple flowers, which are shaped like a monk's cowl. Their alkaloid content, mainly in the roots, makes them some of the most toxic plants commonly encountered. The dried roots of Aconitum napellus were once used, mainly externally for relief of pain, e.g. in rheumatism. The toxic alkaloids (0.3-1.5%) are complex diterpene-derived esters. Aconitine (Figure 124) is the principal component (about 30%) and is a diester of aconine with acetic and benzoic acids. Hydrolysis products benzoylaconine and aconine are also present in dried plant material. These alkaloids appear to behave as neurotoxins by acting on sodium channels. All species of Aconitum and Delphinium are potentially toxic to man and animals and must be treated with caution.
 |
| Figure 125 |




