Terpenoid Tetrahydroisoquinoline Alkaloids
The alkaloids found in ipecacuanha*, the dried rhizome and roots of Cepahaelis ipecacuanha(Rubiaceae), have a long history of use in the treatment of amoebic dysentery, and provide unusual principal alkaloids, e.g. emetine and cephaeline(Figure 67), possess a skeleton with two tetrahydroisoquinoline ring systems, plus a further fragment that has its origin in a terpenoid-derived molecule. This terpenoid substrate is the secoiridoid secologanin, a compound that also features in the biosynthesis of many complex indole alkaloids. Secologanin is an aldehyde and can condense with dopamine to give the tetrahydroisoquinoline alkaloid N-deacetylisoipecoside (Figure 67). Ipecosideitself is found in ipecacuanha, though it has the opposite stereochemistry at C-1 to this biosynthetic intermediate. The secologanin fragment contains an acetal function, which can be restored to its component aldehyde and alcohol fragments by hydrolysis of the glucosidic bond. The newly liberated aldehyde can then bond with the secondary amine to give the quaternary Schiff base. This intermediate is converted into an aldehyde by a sequence of reactions: reduction of iminium, reduction of alkene, plus hydrolysis of ester and subsequent decarboxylation, though not necessarily in that order.The decarboxylation step is facilitated by the β-aldehyde function, shown as an enol in Figure 67. Most of the reactions taking place in the secologanin-derived part of the structure are also met in discussions of terpenoid indole alkaloids. The resultant aldehyde is now able to participate in formation of a second tetrahydroisoquinoline ring system, by reaction with a second dopamine molecule. Methylation gives cephaeline and emetine.
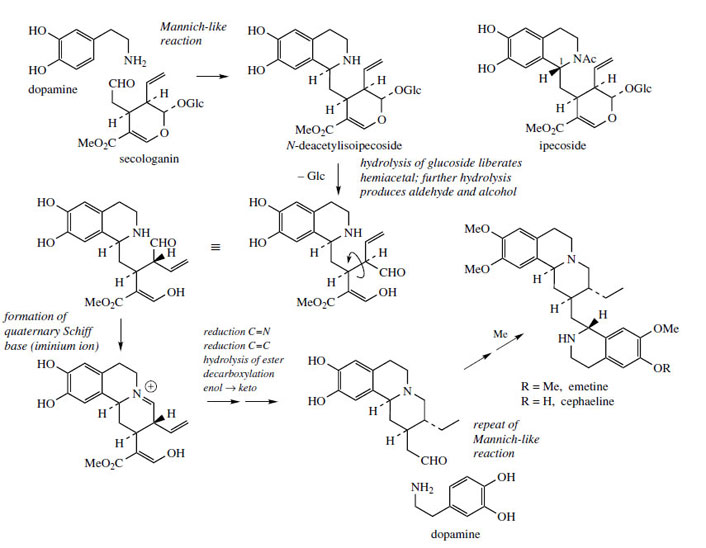 |
| Figure 67 |
Ipecacuanha
Ipecacuanha or ipecac is derived from the dried rhizome and roots of Cephaelis ipecacuanhaor C. acuminata (Rubiaceae). These are low straggling shrubs having horizontal rhizomes with prominently ridged roots. Cephaelis ipecacuanha yields what is termed Rio or Brazilian ipecac, and is cultivated mainly in Brazil, whilst C. acuminata gives Cartagena, Nicaragua, or Panama ipecac, and comes principally from Colombia and Nicaragua. Most of the commercial ipecac now derives from C. acuminata. Ipecac is an age-old remedy of the South American Indians, who used it for the treatment of dysentery. More recently it was mixed with powdered opium to give Dover's powder, where the ipecac content functioned as a diaphoretic.
Ipecac contains 2-2.5% of alkaloids, the principal ones being emetine and cephaeline (Figure 67). Typically, in C. ipecacuanha the emetine to cephaeline ratio might be about 2:1, whereas in C. acuminata the ratio ranges from about 1:2 to 1:1. Minor alkaloids characterized include psychotrine and O-methylpsychotrine (Figure 68), which are dehydro variants of cephaeline and emetine respectively.
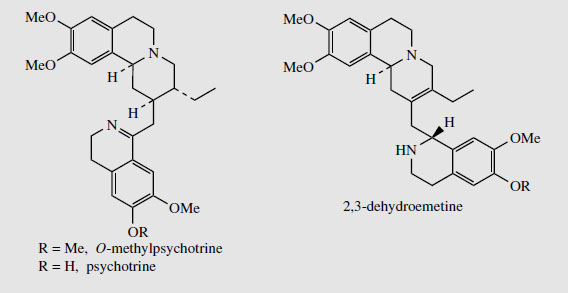 |
| Figure 68 |
Both emetine and the synthetic 2,3-dehydroemetine (Figure 68) have been useful as anti-amoebics, particularly in the treatment of amoebic dysentery. However, they also cause nausea, and this has now made other drugs preferable. The emetic action of the alkaloids is particularly valuable though, and the crude drug extract in the form of ipecacuanha emetic mixture is an important preparation used for drug overdose or poisoning. The emetic mixture is often a standard component in poison antidote kits. Ipecacuanha also has expectorant activity and extracts are still components of a number of compound expectorant preparations. Emetine has more expectorant and less emetic action than cephaeline, and thus the Brazilian drug is preferred for such mixtures. If required, emetine may be obtained in larger amounts by also methylating the cephaeline component of the plant material.
Emetine and cephaeline are both potent inhibitors of protein synthesis, inhibiting at the translocation stage. They display antitumour and antiviral as well as antiamoebic activity, but are too toxic for therapeutic use. In recent studies, O-methylpsychotrine has displayed fairly low effects on protein synthesis, but a quite potent ability to curb viral replication through inhibition of HIV-reverse transcriptase. This may give it potential in the treatment of AIDS.




