Agar Disk Diffusion Method
The testing method most frequently used is the standardized filter paper disk agar diffusion method, also known as the NCCLS (National Committee for Clinical Laboratory Standards) or Kirby-Bauer method. In this test, a number of small, sterile filter paper disks of uniform size (6 mm) that have each been impregnated with a defined concentration of an antimicrobial agent are placed on the surface of an agar plate previously inoculated with a standard amount of the organism to be tested. The plate is inoculated with uniform, close streaks to assure that the microbial growth will be confluent and evenly distributed across the entire plate surface. The agar medium must be appropriately enriched to support growth of the organism tested. Using a disk dispenser or sterile forceps, the disks are placed in even array on the plate, at well-spaced intervals from each other. When the disks are in firm contact with the agar, the antimicrobial agents diffuse into the surrounding medium and come in contact with the multiplying organisms. The plates are incubated at 35°C for 18 to 24 hours.After incubation, the plates are examined for the presence of zones of inhibition of bacterial growth (clear rings) around the antimicrobial disks (see colorplate 14). If there is no inhibition, growth extends up to the rim of the disks on all sides and the organism is reported as resistant (R) to the antimicrobial agent in that disk. If a zone of inhibition surrounds the disk, the organism is not automatically considered susceptible (S) to the drug being tested. The diameter of the zone must first be measured (in millimeters) and compared for size with values listed in a standard chart (Table 15.1). The size of the zone of inhibition depends on a number of factors, including the rate of diffusion of a given drug in the medium, the degree of susceptibility of the organism to the drug, the number of organisms inoculated on the plate, and their rate of growth. It is essential, therefore, that the test be performed in a fully standardized manner so that the values read from the chart provide an accurate interpretation of susceptibility or resistance. In some instances, the organism cannot be classified as either susceptible or resistant, but is interpreted as being of “intermediate” or “indeterminate” (I) susceptibility to a given drug. The clinical interpretation of this category is that the organisms tested may be inhibited by the antimicrobial agent provided that either (1) higher doses of drug are given to the patient, or (2) the infection is at a body site where the drug is concentrated; for example, the penicillins are excreted from the body by the kidneys and reach higher concentrations in the urinary tract than in the bloodstream or tissues. When an interpretation of I is obtained, the physician may wish to select an alternative antimicrobial agent to which the infecting microorganism is fully susceptible or additional tests may be necessary to assess the susceptibility of the organism more precisely.
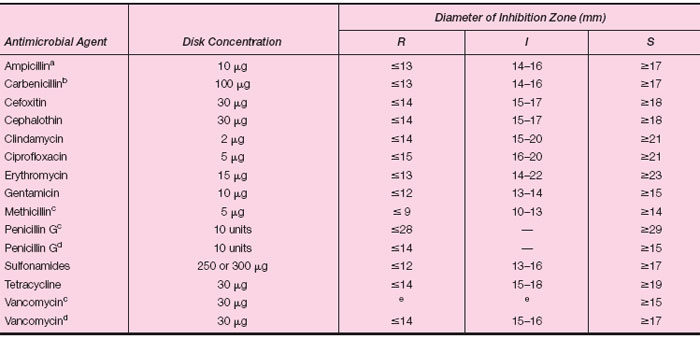 |
| Table 15.1 Zone Diameter Interpretive Table |
| Purpose | To learn the agar disk diffusion technique for antimicrobial susceptibility testing |
| Materials | Nutrient agar plates (Mueller-Hinton if available) Tubes of sterile nutrient broth or saline (5 ml each) Antimicrobial disks (various drugs in standard concentrations) Antimicrobial disk dispenser (optional) McFarland No. 0.5 turbidity standard Sterile swabs Forceps 24-hour plate cultures of Staphylococcus epidermidis and Escherichia coli |
Procedures
- Touch 4 to 5 colonies of S. epidermidis with your sterilized and cooled inoculating loop. Emulsify the colonies in 5 ml of sterile broth or saline until the turbidity is approximately equivalent to that of the McFarland No. 0.5 turbidity standard.
- Dip a swab into the bacterial suspension, express any excess fluid against the side of the tube, and inoculate the surface of an agar plate as follows: first streak the whole surface of the plate closely with the swab; then rotate the plate through a 45° angle and streak the whole surface again; finally rotate the plate another 90° and streak once more. Discard the swab in disinfectant.
- Repeat steps 1 and 2 with the E. coli broth culture on a second nutrient agar plate.
- Heat the forceps in the Bunsen burner flame or bacterial incinerator, and allow to cool.
- Pick up an antimicrobial disk with the forceps and place it on the agar surface of one of the inoculated plates. Press the disk gently into full contact with the agar, using the tips of the forceps.
- Heat the forceps again and cool.
- Repeat steps 5 and 6 until about eight different disks are in place on one plate, spaced evenly away from each other. (If an antimicrobial disk dispenser is available, all disks may be dispensed on the agar surface simultaneously. Be certain to press them into contact with the agar using the forceps tips.)
- Place a duplicate of each disk on the other inoculated plate, using the same procedures.
- Invert the plates and incubate them at 35°C for 18 to 24 hours.
Results
Observe for the presence or absence of growth around each antimicrobial disk on each plate culture. Using a ruler with millimeter markings, measure the diameters of any zones of inhibition and record them in the chart. If the organism grows right up to the edge of a disk, record a zone diameter of 6 mm (the diameter of the disk).
 |




