Soil Water
Phosphorus availability is affected by soil water conditions. Soil water affects soil reactions governing the release and diffusion of phosphorus in the soil solution and ultimately the positional availability of phosphorus relative to root growth. Generally, maximum availability of phosphorus for most crops has been associated with a soil water tension of about 1/3 bar (76).The dissolution of fertilizer phosphorus and all amorphous and mineral phosphorus compounds in the soil depends on soil water. Furthermore, under anaerobic conditions, the reduction of ferric phosphates to ferrous phosphates might result in additional increased phosphorus solubility (77,78). Nevertheless, it is the general view that with the exception of aquatic crops, excessive water resulting in poor aeration would actually restrict phosphorus uptake by crops in spite of this enhanced solubility. However, Bacon and Davey (79), using trickle irrigation in an orchard, noted increased phosphorus availability during and immediately after each irrigation and noted that available phosphorus decreased rapidly as soil moisture declined below field capacity. These authors attributed this increased phosphorus availability to the reduction of amorphous iron phosphates in anaerobic micro-sites.
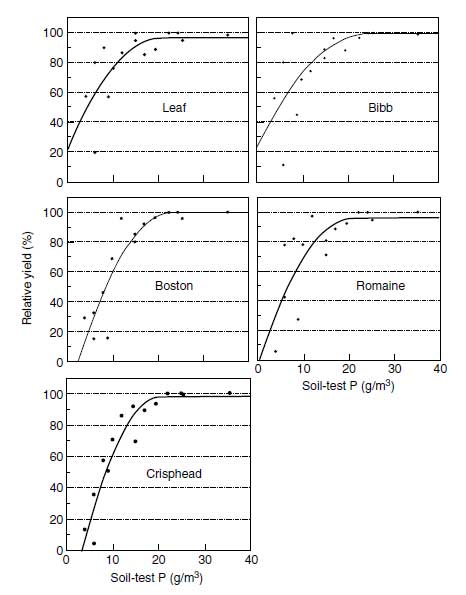 |
| FIGURE 3.7 Response of five lettuce types to soil-test phosphorus. (Adapted from C.A. Sanchez and N.M. El-Hout, HortScience 30:528–531, 1995.) |
The volume of soil that is occupied by water affects the cross-sectional area through which phosphorus can diffuse (80). Thus, the lower the soil moisture, the more tortuous the path of diffusion and the greater the likelihood of contact with soil constituents that render phosphorus insoluble.
Under most conditions, phosphorus is applied near the soil surface. Thus, during dry periods in nonirrigated production systems, crops largely draw soil moisture from lower soil depths, and phosphorus deficiencies can arise (81). This condition is generally not a problem in irrigated production systems where root growth extends to near the soil surface.




