Cell Culture and Metabolite Production
When we cultivate plant cells In vitro, there are different types of cultures with distinct metabolite productivities. One type produces metabolites in undifferentiated cells cultured In vitro, while another produces metabolites only under differentiated conditions, for example, shoot or root cultures. In some cases, cells lose their biosynthetic potential even after redifferentiation, that is, showing the complete loss of their natural biosynthetic potential.The first type of cell culture is in high demand because cells are easily cultivated under In vitro conditions. Plant cells often do not produce desired secondary metabolites, although they have the potential to regenerate whole plants from single cells. For example, plant growth regulators, such as auxins and cytokinins, can regulate morphological differentiation, but the chemical regulation of functional differentiation in secondary metabolism without morphological differentiation is rather limited. For example, morphinan alkaloids are not produced in cell cultures of P. somniferum without organ differentiation (Facchini and Park, 2003; Grothe et al., 2001; Huang and Kutchan, 2000; Unterlinner et al., 1999), whereas several cell cultures, for example, P. somniferum and C. japonica, produce large quantities of structurally related isoquinoline alkaloids, sanguinarine and berberine, respectively (Facchini and Park, 2003; Huang and Kutchan, 2000; Sato and Yamada, 1984). Interestingly, morphine, sanguinarine, and berberine are derived from tyrosine through the same intermediate, reticuline (Fig. 11.2), indicating that early steps in metabolic pathway do not determine the end-product in cell culture.
Biochemical, molecular biological, and cell biological studies of biosynthetic enzymes have gradually revealed the mechanisms of regulation. For example, all enzymes examined were highly expressed in cultured C. japonica cells, which show a high production of berberine (Ikezawa et al., 2003; Sato et al., unpublished data), and in P. somniferum cells, which do not produce morphinan alkaloids under undifferentiated conditions (Facchini and Park, 2003; Grothe et al., 2001; Huang and Kutchan, 2000; Unterlinner et al., 1999). However, biosynthetic enzymes in sanguinarine biosynthesis in roots have been localized to the immature endodermis and the protodermis of leaf primordia in the rhizome of Thalictrum (Samanani et al., 2005). Similarly, the enzymes in morphinan alkaloid biosynthesis were localized in different cell types (see above); localization of 40OMT and SAT were both in phloem parenchyma cells, but the enzyme catalyzing the penultimate step, COR, in morphine biosynthesis was located in the laticifers (Kutchan, 2005a; Weid et al., 2004). These results suggest that secondary metabolite production in cell cultures is regulated in a complicated manner. The coordinated expression of biosynthetic genes and their enzymes at a high level seems to be an essential requirement for high production of metabolites.
Many medicinal compounds are used as chemical defense agents in whole plants. These metabolites often have activities as phytoalexins which are induced in response to fungal attack, that is, to act as endogenous chemical weapons for defense in plants. For example, berberine and sanguinarine are antibacterial and antifungal agents (Schmeller et al., 1997). These chemicals are also produced in cells/tissues cultured In vitro, even though they are cultivated under aseptic conditions without infection by microbes or attack by animals. The high expression of pathogenesis-related protein genes, as well as of cell proliferation-related genes in cultured cells, has been indicated by protein analysis and expressed sequence tag (EST)- and microarray analyses, indicating that the cell cultures exhibit stress responses (Sasaki et al., 1994; Takeda et al., 1990; Sato et al., unpublished data).
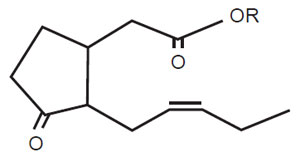 |
| FIGURE 11.5 Active jasmonic acid structure. JA; R=H; MeJA, R=CH3. |
JA (or MeJA) (Fig. 11.5) is an active component in stress responses, especially in elicitation induced by microbial cell walls, heavy metals, and so on (Gundlach et al., 1992; Zhao et al., 2005). Among signal mediators, JA has a pronounced effect in the production of several secondary metabolites, including paclitaxel (Yukimune et al., 1996). However, the application of JA alone is not sufficient to induce the entire biosynthetic pathway for indole alkaloids (Eilert et al., 1987; Van Der Fits and Memelink, 2000): the application of JA induced only a set of biosynthetic genes (Fig. 11.3). Some high-metabolite-producing cells do not respond to JA, suggesting that the signal transduction system and/or the expression of some downstream biosynthetic gene(s) may be highly activated during cell selection (Sato et al., unpublished data). While many of the biosynthetic genes in secondary metabolism respond to JA, PMT in tropane alkaloid synthesis does not (Suzuki et al., 1999a).
Regarding defense responses, we have identified several mediators for such signals other than JA, including salicylic acid (SA) and ethylene. Recent advances in the molecular biology of signal transduction have shown that the overall mechanism of regulation of the expression of defense genes is more complicated and divergent among plant species than expected (Vom Endt et al., 2002; Zhao et al., 2005): for example, the ‘‘ethylene response factor 1’’ in Arabidopsis acts downstream of the intersection between the ethylene and JA pathways, suggesting that these signals are somehow integrated (Lorenzo et al., 2003; Vom Endt et al., 2002). Although a general antagonism between JA and SA and synergistic interaction between JA and ethylene have been noted in plant–pathogen interactions, it is still too early to make any definite conclusions about this topic.




