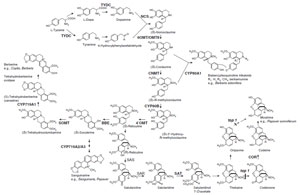
Isoquinoline Alkaloid Biosynthesis
Isoquinoline alkaloids are a large and diverse group of alkaloids with~2500 defined structures. They include the analgesic morphine from Papaver somniferum L., the antigout colchicine from Colchicum autumnale L., the emetic and antiamoebic emetine from Cephaelis ipecacuanha (Brot.) A. Rich., the skeletal muscle relaxant tubocurarine from Strychonos toxifera Bentham, and the antimicrobial compounds berberine and sanguinarine from divergent plant species including Berberis spp. and Sanguinaria spp., many of which are used as pharmaceuticals.Isoquinoline alkaloid biosynthesis begins with the conversion of tyrosine to both dopamine and 4-hydroxyphenylacetaldehyde by decarboxylation, orthohydroxylation, and deamination (Fig. 11.2; Facchini, 2001). Among these early steps, only tyrosine/dopa decarboxylase (TYDC; an aromatic L-amino acid decarboxylase), which converts tyrosine and dopa to their corresponding amines, has been purified and characterized. This small family of genes (~15 genes) was isolated from opium poppy (P. somniferum) and each subfamily has been shown to have distinct developmental and inducible expression patterns (Facchini and De Luca, 1994, 1995; Park et al., 1999). Members of the TYDC gene family are classified into two groups (TYDC1 and TYDC2) that are differentially expressed in opium poppy. In the mature plant, TYDC2-like transcripts are predominant in the stem and are also present in roots, whereas TYDC1-like transcripts are abundant only in roots. The localization of TYDC transcripts in the phloem is consistent with the expected developmental origin of laticifers, which are specialized internal secretory cells that accompany vascular tissues in all organs of select species and contain alkaloid-rich latex in aerial organs (Facchini and De Luca, 1995).
Dopamine and 4-hydroxyphenylacetaldehyde are condensed by norcoclaurine synthase (NCS) to yield (S)-norcoclaurine, which is the central precursor to all isoquinoline alkaloids. Recently, NCS has been purified and characterized (Samanani and Facchini, 2002) from cultured Thalictrum flavum spp., and a TfNCS cDNA belonging to PR10 family was isolated from T. flavum (Samanani et al., 2004), whereas a novel dioxygenase-like protein (CjNCS) from cultured Coptis japonica cells was also shown to catalyze this NCS reaction (Minami et al., 2007). The presence of TYDC (Facchini, 2001) and TfNCS (Samanani et al., 2004) and CjNCS homologues (Minami et al., 2007) in Arabidopsis or rice suggests that these genes either have other basic biological roles or that the isoquinoline biosynthesis pathway is relatively universal in the plant kingdom, although sequence homology of TfNCS or CjNCS with Arabidopsis or rice homologues were relatively low (less than 20% in amino acid basis) (Liscombe et al., 2005; Minami et al., 2007) and no isoquinoline alkaloid has been found in Arabidopsis or rice. Strictosidine synthase (STR) (the key reaction in terpenoid indole alkaloid)- like genes has also been found in animals and Arabidopsis (De Luca and Laflamme, 2001).
(S)-Norcoclaurine is sequentially converted to coclaurine by S-adenosyl methionine (SAM)-dependent norcoclaurine 6-O-methyltransferase (6OMT) (Morishige et al., 2000), to N-methylcoclaurine by coclaurine N-methyltransferase (Choi et al., 2002), to 3'-hydroxy-N-methyl coclaurine by P450 hydroxylase (Pauli and Kutchan, 1998), and then to (S)-reticuline by 3'-hydroxy N-methylcoclaurine 4'-O-methyltransferase (4'OMT; see Fig. 11.2; Morishige et al., 2000). All of the cDNAs for these reactions have been isolated and functional recombinant proteins subsequently produced. Detailed biochemical studies using recombinant enzymes have shown their strict reaction specificities, and these enzymes regulate biosynthesis sequentially and in a coordinated manner. For example, CNMT prefers coclaurine than 6-O-methylnorlaudanosoline and 4'OMT prefers an N-methylated substrate, which suggest that the pathway in Fig. 11.2 is preferable to a sequence of N-methylation, hydroxylation, and 4'-O-methylation. On the other hand, Thalictrum cells may show some variation since Thalictrum O-methyltransferases can form heterodimers and exhibit broad substrate specificity (Frick and Kutchan, 1999). Current data also indicate that all of these enzymes, except the membrane-bound P450 CYP80B1, are located in the cytosol.
While dimeric bisbenzylisoquinoline alkaloids, such as berbamunine and tubocurarine, are produced from the intermediates of the (S)-reticuline pathway by the action of a phenol coupling P450-dependent oxidase (berbamunine synthase, CYP80A1) (Kraus andKutchan, 1995), reticuline is the central intermediate in branch pathways that lead to benzophenanthridine alkaloids (e.g., sanguinarine and marcarpine), protoberberine alkaloids (e.g., berberine and palmatine), and morphinan alkaloids (e.g., morphine and codeine) (Fig. 11.2). Many of the enzymes involved in these branch pathways have been purified and the corresponding cDNAs have been cloned.
The first committed step in the biosynthesis of benzophenanthridine, a protoberberine alkaloid, involves conversion of the N-methyl group of (S)-reticuline into the methylene bridge moiety of (S)-scoulerine by the berberine bridge enzyme (BBE) (Dittrich and Kutchan, 1991). This unique enzyme is soluble but localized in vesicles (Bock et al., 2002). Immunocytological staining of P. somniferum tissue with antibodies against BBE led to a characteristic labeling of electron-dense aggregates in idioblasts that are not connected to the laticifer system, which demonstrates that benzophenanthridine and morphine biosyntheses show strict cytological separation within this plant (Bock et al., 2002).
In benzophenanthridine alkaloid biosynthesis, (S)-scoulerine can be converted to (S)-stylopine by two P450-dependent oxidases, (S)-cheilanthifoline and (S)-stylopine synthase, which result in the formation of two methylenedioxy groups (not shown) (see Facchini, 2001; Ikezawa et al., 2007). On the other hand, in protoberberine biosynthesis, (S)-scoulerine is converted to (S) tetrahydrocolumbamine by the SAM-dependent scoulerine 9-O-methyltransferase (SOMT) (Takeshita et al., 1995) and then to tetrahydroberberine (canadine) by a P450-dependent canadine synthase (CDS or CYP719A1) (Ikezawa et al., 2003). The isolation and characterization of these enzymes and the corresponding cDNAs have confirmed that berberine biosynthesis proceeds via canadine and not via columbamine. Again, the enzyme substrate specificity shows a clear preference for this pathway.While the hydrophobic N-terminal region of SOMT suggests that this enzyme may be targeted to the membrane fraction, its localization in both the cytosol (Muemmler et al., 1985) and within the lumen of alkaloid-specific vesicles (Galneder et al., 1988) has been reported. Note that the CYP719A1 family as well as CYP80 were not found in Arabidopsis and rice and that these members of the cytochrome P450 superfamily are unique for benzylisoquinoline alkaloid biosynthesis (Nelson et al., 2004).
In morphinan alkaloid biosynthesis, (S)-reticuline is converted to its (R)-enantiomer via the stereospecific reduction of 1,2-dehydroreticuline with NADPHdependent cytosolic 1,2-dehydroreticuline reductase. Subsequent intramolecular carbon–carbon phenol coupling of (R)-reticuline by a P450-dependent salutaridine synthase results in the formation of salutaridine. Salutaridine: NADPH 7-oxidoreductase then reduces salutaridine to (7S)-salutaridinol. Transformation of salutaridinol into thebaine involves the closure of an oxide bridge between C-4 and C-5 by acetylcoenzyme A:salutaridinol-7-O-acetyltransferase (SAT) (Grothe et al., 2001). Furthermore, thebaine can be converted to codeinone and then reduced to codeine by cytosolic NADPH-dependent codeinone reductase (COR) (Unterlinner et al., 1999). Finally, codeine is demethylated to give morphine. Interestingly, COR genes have been found in some Papaver spp. that do not produce morphine (Unterlinner et al., 1999), whereas SAT transcript was detected in Papaver spp. that accumulate alkaloids with a morphinan nucleus, consistent with the expected distribution (Grothe et al., 2001; Unterlinner et al., 1999). The recent isolation of the top1 mutant from poppy, and the demonstration of the activity of the protein, has illustrated that thebaine can be demethylated in two steps either through codeinone or oripavine to morphine (Millgate et al., 2004).
Northern blot analysis using the eight available genes in morphinan alkaloid biosynthesis showed that while all of the transcripts are detected in every organ, the highest levels are seen in stems and flower buds and the lowest levels are seen in leaves (Millgate et al., 2004; Unterlinner et al., 1999). The accumulation of each transcript, with the exception of COR, was markedly induced in response to treatment with an elicitor or wounding of cultured cells. All known enzymes in the morphine pathway have been detected in cultured cells derived from the fruit capsule (Facchini and Park, 2003; Grothe et al., 2001; Huang and Kutchan, 2000; Unterlinner et al., 1999). Conversely, different cell type-specific localizations of biosynthetic enzymes have been reported in the capsule and stem of intact opium poppy plants. In situ localization of alkaloid biosynthetic gene transcripts indicated seven biosynthetic enzymes: 6OMT, CNMT, CYP80B, 4'OMT and BBE involved in reticuline biosynthesis, and SAT and COR in morphine pathway. These proteins have apparently been localized to sieve elements in opium poppy and the corresponding gene transcripts to adjacent phloem companion cells (Bird et al., 2003; Facchini and St-Pierre, 2005). In contrast, a different immunocytochemical analysis clearly showed that 4'OMT and SAT were located in phloem parenchyma cells in vascular bundles. COR, catalyzing the penultimate step in morphine biosynthesis, was localized to laticifers, the site of morphinan alkaloid accumulation (Weid et al., 2004). Although this discrepancy in the cell type-specific localization of enzymes remains to be clarified, it is noteworthy that both studies showed different localizations of the biosynthetic gene transcripts and their corresponding enzymes.
Cell type-specific expression has recently been reported in protoberberine alkaloid biosynthesis in T. flavum subsp. (Samanani et al., 2005). While gene transcripts for biosynthetic enzyme were most abundant in rhizomes, they were also detected at lower levels in roots and other organs. Further In situ RNA hybridization analysis revealed that all transcripts were mainly localized to immature endodermis cells, the pericycle of roots, and restricted to the protoderm of leaf primordia in rhizomes. These data and analysis of alkaloid accumulation clearly indicated that distinct and different cell types are involved in the biosynthesis and accumulation of benzylisoquinoline alkaloids in T. flavum and P. somniferum.