Mollusc pests
Content
Slugs This group of serious pests belongs to the phylum Mollusca, a group including the octopus and whelk and the slug’s close relatives, the snails, which cause some damage to plants in greenhouses and private gardens.
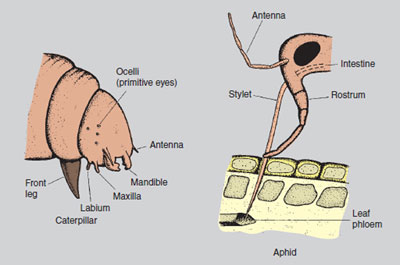
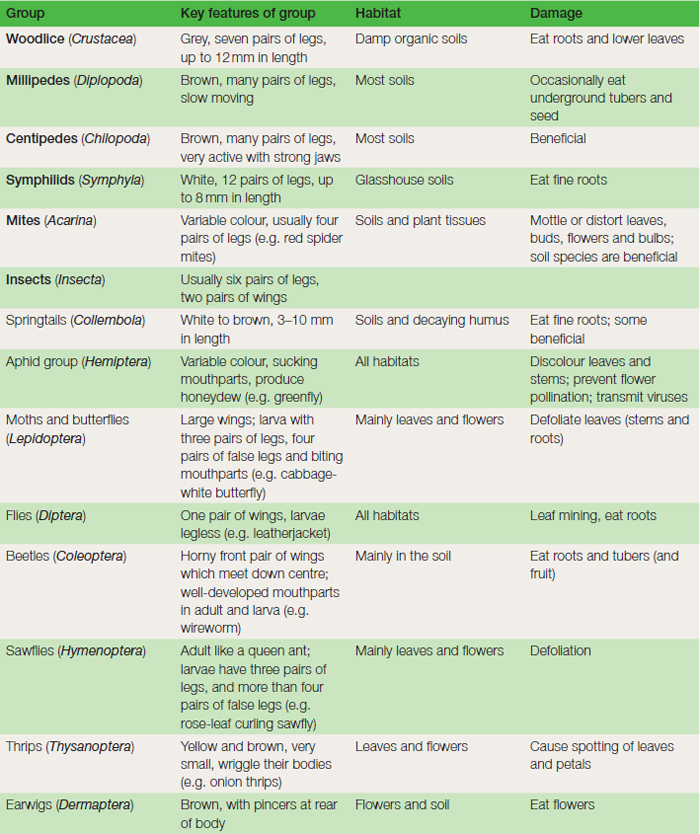
Life cycle: Slugs are hermaphrodite (bearing in their bodies both male and female organs), mate in spring and summer, and lay clusters of up to 50 round, white eggs in rotting vegetation, the warmth from which protects this sensitive stage during cold periods. Slugs range in size from the black keeled slug (Milax) 3 cm long, to the garden slug (Arion) which reaches 10 cm in length. The mottled carnivorous slug (Testacella) is occasionally found feeding on earthworms. Control: Private gardeners use many non-chemical forms of control, ranging from baits of grapefruit skins and stale beer to soot sprinkled around larger plants. A nematode (Phasmarhabditis hermaphrodita) is increasingly being used to limit slug numbers. The most effective methods available to both amateur and professional horticulturists at the moment are three chemicals, aluminium sulphate (an irritant), metaldehyde (which dehydrates the slug) and methiocarb (which acts as a stomach poison). The chemicals are most commonly used as small-coloured pellets (which include food attractants such as bran and sugar), but metaldehyde may also be applied as a drench. Some growers estimate the slug population using small heaps of pellets covered with a tile or flat stone (to prevent bird poisoning) before deciding on general control. Use of metaldehyde and methiocarb in gardens has recently been claimed as a major contribution to the decline of the thrush numbers. A simple device such as that seen in Figure 16.2, using a modified plastic milk carton (containing slug pellets) prevents the entry of mammals and birds. Insect pests Belonging to the large group of Arthropods, which include also the woodlice, mites, millipedes and symphilids (see Table 14.1), insects are horticulturally the most important arthropod group, both as pests, and also as beneficial soil animals. Structure and biology The body of the adult insect is made up of segments, and is divided into three main parts: the head, thorax and abdomen (see Figure 14.4). The head bears three pairs of moving mouthparts. The first, the mandibles in insects (such as in caterpillars and beetles) have a biting action (see Figure 14.5). The second and third pairs, the maxillae and labia in these insects help in pushing food into the mouth. In the aphids, the mandibles and maxillae are fused to form a delicate tubular stylet, which sucks up liquids from the plant phloem tissues. Insects remain aware of their environment by means of compound eyes which are sensitive to movement (of predators) and to colour (of flowers). Their antennae may have a touching and smelling function. The thorax bears three pairs of legs and, in most insects, two pairs of wings. The abdomen bears breathing holes (spiracles) along its length, which lead to a respiratory system of tracheae. The blood is colourless, circulates digested food and has no breathing function. The digestive system, in addition to its food absorbing role, removes waste cell products from the body by means of fine, hair-like growths (malpighian tubules) located near the end of the gut.
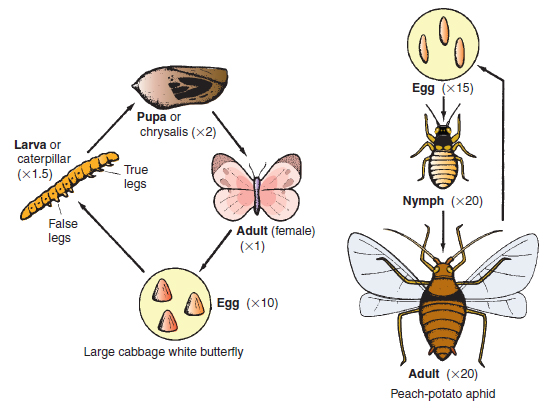
Since the animal has an external skeleton made of tough chitin, it must shed and replace its ‘skin’ (cuticle, see Figure 14.4) periodically by a process called ecdysis, in order to increase in size. The two main groups of insect develop from egg to adult in different ways. In the first group (Endopterygeta), typified by the aphids, thrips and earwigs, the egg hatches to form a first stage, or instar, called a nymph, which resembles the adult in all but size, wing development and possession of sexual organs. Successive nymph instars more closely resemble the adult. Two to seven instars (growth stages) occur before the adult emerges (see Figure 14.6). This development method is called incomplete metamorphosis.
The method of overwintering differs between insect groups. The aphids survive mainly as the eggs, while most moths, butterflies and flies survive as the pupa. The speed of increase of insects varies greatly between groups. Aphids may take as little as 20 days to complete a life cycle in summer, often resulting in vast numbers in the period June–September. On the other hand, the wireworm, the larva of the click beetle, usually takes four years to complete its life cycle. Insect groups are classified into their appropriate order (Table 14.1) according to their general appearance and life cycle stages. There follows now a selection of insect pests in which each species’ particular features of life cycle are given. Whilst comments on control are mentioned here, the reader should also refer to Plant protection for details of specific types of control (cultivations, chemicals, etc.) and for explanations of terms used.
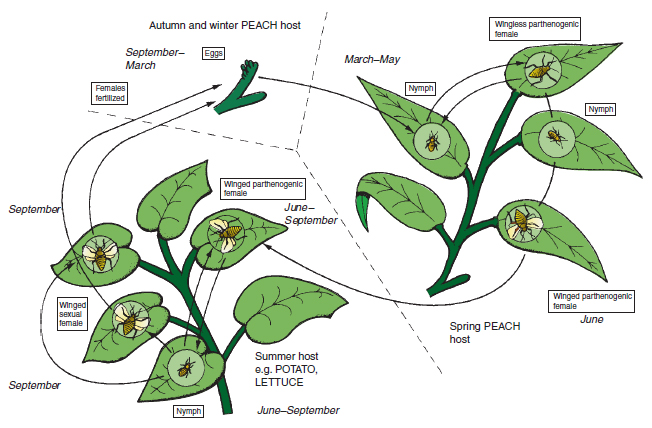
Aphids and their relatives ( Order Hemiptera ) This important group of insects has the egg–nymph–adult life cycle and sucking mouthparts. Peach-potato aphid (Myzus persicae) This and similar species are often referred to by the name ‘greenfly’ (Figure 14.8). Damage: It is common in market gardens and greenhouses. The nymph and adult of this aphid may cause three types of damage. Using its sucking stylet, it may inject a digestive juice into the plant phloem, which in young organs may cause severe distortion. Having sucked up sugary phloem contents, the aphid excretes a sticky substance called honey-dew, which may block up leaf stomata and reduce photosynthesis, particularly when dark-coloured fungi (sooty moulds) grow over the honeydew. Thirdly, the aphid stylets may transmit viruses such as virus Y on potatoes and tomato aspermy virus on chrysanthemums. Life cycle: The aphid varies in colour from light green to orange, measures 3 mm in length (see Figure 14.9) and has a complex life cycle, shown in Figure 14.8, alternating between the winter host (peach) and the many summer hosts such as potato and bedding plants. In spring and summer, the females give birth to nymphs directly without any egg stage (a process called vivipary), and without fertilization by a male (a process called parthenogenesis). Spread is by the summer flighted females. Only in autumn, in response to decreasing daylight length and outdoor temperatures, are both sexes produced, which having wings, fly to the winter host, the peach. Here, the female is fertilized and lays thick-walled black eggs. In glasshouses, the aphid may survive the winter as the nymph and adult female on plants such as begonias and chrysanthemums, or on weeds such as fat hen.
Control: The peach-potato aphid can be controlled in several ways. In outdoor crops, several organisms, e.g. ladybirds, lacewings, hoverflies and parasitic fungi (see biological control, Plant protection), naturally found in the environment, may reduce the pest’s importance in favourable seasons. In the greenhouse, an introduced parasitic wasp (Aphidius matricariae) is available to amateurs and professionals. The Amateur gardener uses an aphicide containing pyrethrins. Two other chemicals are used by both amateur and professional horticulturists: a contact chemical bifenthrin and a soap concentrate containing fatty acids. For the professional, there is a biological control using the fungus Verticillium lecanii, and many chemical controls, including pirimicarb used outdoors and in glasshouses as a spray, and nicotine used as a smoke in glasshouses. There are many other horticulturally important aphid species. The black bean aphid (Aphis fabae), which overwinters on Euonymus bushes, may seriously damage broad beans, runner beans and red beet. The rose aphid (Macrosiphum rosae) attacks young shoots of rose. Spruce-larch adelgid (Adelges viridis) This relative of the aphid may cause serious damage on spruce grown for Christmas trees. Damage: Although nursery trees less than four years of age are rarely badly damaged, early infestation in the young plant may result in serious damage as it gets older. In June–September the adults move to larch , acquire woolly white hairs and may cause defoliation of the leaves. Life cycle: The green female adult develops from overwintering nymphs on spruce, and in May (year 1) lays about 50 eggs on the dwarf shoots. The emerging nymphs, injecting poisons into the shoots, cause abnormal growth into pineapple galls, which spoil the tree’s appearance. After a further year (year 2) on this host, the female adelges returns to the Figure 14.9 R ose aphid spruce, where it lives for another year (year 3) before the gall-inducing stages are produced. Spread is by flighted females. Control: In Christmas tree production, the adelges may be controlled by sprays of deltamethrin in May, when the gall-inducing nymphs are developing. Glasshouse whitefly (Trialeurodes vaporariorum) This small insect, looking like a tiny moth, was originally introduced from the tropics, but now causes serious problems on a range of glasshouse food and flower crops. It should not be confused with the very similar, but slightly larger cabbage whitefly on brassicas. Damage: All stages after the egg have sucking stylets, which extract a sugary liquid from the phloem, often causing large amounts of honeydew and sooty moulds on the leaf surface. Plants that are seriously attacked include fuchsias, cucumbers, chrysanthemums and pelargoniums. Chickweed, a common greenhouse weed, may harbour the pest over winter in all stages of the pest’s life cycle.
Spread is mainly by introduced plants, or more rarely by chance arrivals of adult through doors or vents. Control of glasshouse whitefly is achieved in several ways. Amateur and Professional horticulturalists should remove weeds (such as chickweed or sowthistle) harbouring the pest from crop to crop. Careful inspection of the lower leaves of introduced plants achieves a similar aim. There is a reliable form of biological control. This involves a minute wasp (Encarsia formosa) which lays an egg inside the last scale stage of the white-fly. The developing whitefly is eaten away by the wasp grub and the scale turns black and soon releases the next generation of wasps (see Plant protection for more details). Control is usually most effective when whitefly numbers are low. Amateur gardeners can use spray products containing specially formulated fatty acids to control young and adult pest stages. Professional horticulturists can choose between an insecticide that physically blocks the insect’s breathing holes such as alginate/polysaccharide, or a contact insecticide such as deltamethrin, or the above-mentioned fatty acids It is suggested that serious infestations of this pest receive a regular weekly chemical spray to catch the more sensitive scale and adult stages.
Damage: This pest, a distant relative of the aphid, spoils the appearance of glasshouse crops, particularly orchids, Coleus species, cacti and Solanum species. All of the stages except the egg suck phloem juices by means of a tubular mouthpart (stylet), and when this pest is present in dense masses it produces honey dew and may cause leaf drop (see Figure 14.11). Life cycle: Being a tropical species, it develops most quickly in high temperatures and humidities, and at 30°C completes a life cycle within about 22 days. The adult measures about 3 mm in length and produces fine waxy threads. Spread. Adults have wings, but the most important spread is by introduction of plant material newly infested with small nymphs, or from plant to plant when leaves are touching. Control: Mealy bugs are difficult pests to control, as the thick cuticle resists chemical sprays, and the droplets fall off the waxy threads. An introduced tropical ladybird, Cryptolaemus montrouzieri, is commercially available for controlling the pest and is most effective at temperatures above 20°C. Spray products containing pure fatty acids are effective against this pest and are available to both amateur and professional horticulturists. A recently reported related problem on beech has been the woolly beech aphid (Phyllapis fagi). Brown scale (Parthenolecanium corni) The female scale, measuring up to 6 mm, is tortoise shaped (see Figure 14.11) and has a very thick cuticle. It may be a serious pest outdoors on vines, currants, top fruit and cotoneasters, sucking phloem sugars out of the plants. It is also found in greenhouses on vines, peaches and Amaryllis, causing stunted growth and leaf defoliation. The nymphs are mobile, but do not spread far. Transport of infested plant material is the main cause of outbreaks in greenhouses. As with mealy-bug, control is made more difficult because of the thick cuticle, but products containing fatty acids give good control. A recently reported scale problem is soft brown scale (Coccus hesperidum) in greenhouses.
These slender, light green insects, about 3 mm long, well known in their nymph stage as ‘cuckoo spit’, are found on a wide variety of crops, e.g. potato, rose, Primula and Calceolaria. The adults can fly from plant to plant. They live on the undersurface of leaves, causing a mottling of the upper surface. In strawberries, they are vectors of the green-petal disease, while in rhododendron they carry the serious bud blast disease that kills off the flower buds (see Figure 14.12). August and September sprays of products containing fatty acids prevent egg laying inside buds of rhododendron, and thus reduce the entry points for the fungus disease. Common green capsid (Lygocoris pabulinus) This very active, light green pest measuring 5 mm in length and resembling a large aphid, occurs on fruit trees and shrubs and flower crops, most commonly outdoors. Owing to the poisonous nature of its salivary juices, young foliage shows distorted growth with small holes, even when relatively low insect numbers are present and fruit is scarred (see Figure 14.12). The adult flies from plant to plant. The chemicals used against aphids control this pest. Thrips (Order Thysanoptera) Whilst classified under the broad grouping of the Exopterygota (like the aphids), thrips also produce a survival stage which is often soil borne, and is loosely called a pupa (a life cycle stage normally reserved for members of the other main insect group, the Endopterygota). Due to their increased activity during warm humid weather, thrips are sometimes called ‘thunder flies’ (and are known for their ability to get into human’s hair in sultry summer weather and cause itching). Onion thrips (Thrips tabaci) Damage: Thrips’ mouthparts are modified for piercing and sucking, and the toxic salivary juices cause silvering in onion leaves, straw-brown spots several mm in diameter on cucumber leaves, and white streaks on carnation petals. Life cycle: The 1 mm long, narrow-bodied insect has feather-like wings. The last instar of the life cycle, called the pupa, descends to the soil, and it is this stage which overwinters. In greenhouses there may be seven generations per year, while outdoors one life cycle is common. Spread. Adults may be blown considerable distances from nursery to nursery in the wind. (The occurrence in Britain of Western flower thrip (Frankliniella occidentalis) on both greenhouse and outdoor flower and vegetable crops has created serious problems for the industry, particularly because it transmits the serious tomato spotted wilt virus, and is able to pupate on the plant, deep within dense flowers such as carnation.) Control: Thrips infestations may be reduced in greenhouses by the use of fine screens over vents, and by a double door system. Amateur gardeners are able to use a recently introduced product for thrip control containing natural plant extracts which block the insect’s breathing holes. Professional growers use the predatory mite Neoseiulus cucumeris, and the predatory bug Orius laevigatus. Chemicals used include the ingredient abamectin and the above named natural plant extract. Western flower thrip has shown greater resistance to chemical control than the other thrips and a careful rotation of chemical groups has proved necessary. Earwigs (Forficula auricularia) These pests belong to the order Dermaptera, and bear characteristic ‘pincers’ (cerci) at the rear of the 15 mm long body. They gnaw away at leaves and petals of crops such as beans, beet, chrysanthemums and dahlias, usually from July to September, when the nymphs emerge from the parental underground nest. They usually spread by crawling on the surface of the soil, but they can also fly. Upturned flower pots containing straw are sometimes used in greenhouses for trapping these shy nocturnal insects. The professional grower may use pirimiphos-methyl as a spray or smoke. Moths and butterflies This group of insects belongs to the large grouping, the Endopterygota which has different life cycle details from the aphid group. The order (Lepidoptera) characteristically contains adults with four large wings and curled feeding tubes. The larva (caterpillar), with six small legs and eight false legs, is modified for a leaf-eating habit (see Figures 14.6 and 14.7). Some species are specialized for feeding inside fruit (codling moth on apple, see Figure 14.15), underground (cutworms), inside leaves (oak leaf miner), or inside stems (leopard moth). The gardener may find large webbed caterpillar colonies of the lackey moth (Malacosoma neustria) on fruit trees and hawthorns, or the juniper webber (Dichomeris marginella) causing webs and defoliating junipers. Large cabbage-white butterfly (Pieris brassicae) Damage: Leaves of cabbage, cauliflower, Brussels sprouts and other hosts such as wallflowers and the shepherd’s-purse weed are progressively eaten away. The defoliating damage of the larva may result in skeletonized leaves.
Spread. The species is spread by the adults. (Care should be taken not to confuse the cabbage-white larva with the large smooth green or brown larva of the cabbage moth, or the smaller light green larva of the diamond-backed moth, both of which may enter the hearts of cabbages and cauliflowers, presenting greater problems for control.) Control: There are several forms of control against the cabbagewhite butterfly. A naturally occurring small wasp (Apantales glomeratus) lays its eggs inside the pest larva. A virus disease may infect the pest, causing the larva to go grey and die. Birds such as starlings eat the plump larvae. When damage becomes severe, amateur gardeners and professional growers can use spray products containing bifenthrin. Winter moths (Operophthera brumata) Damage: These are pests which may be serious on top fruit and ornamentals, especially woody members of the Rosaceae family. The caterpillars eat away leaves in spring and early summer and often form other leaves into loose webs, reducing the plant’s photosynthesis. They occasionally scar young apple fruit. Life cycle: This pest’s timing of life cycle stages is unusual. The pest emerges as the adult form from a soil-borne pupa in November and December. The male is a greyish-brown moth, 2.5 cm across its wings, while the female is wingless. The female crawls up the tree to lay 100– 200 light-green eggs around the buds. The eggs hatch in spring at bud burst to produce green larvae with faint white stripes. These larvae move in a characteristic looping fashion and when fully grown, descend on silk threads at the end of May before pupating in the soil until winter. Spread is slow because the females do not have wings. Control: A common control is a grease band wound around the main trunk of the tree in October which is effective in preventing the flightless female moth’s progress up the tree. In large orchards, professional growers use springtime sprays of an insecticide such as deltamethrin to kill the young caterpillars. Cutworm (e.g. Noctua pronuba) Damage: The larvae of the yellow underwing moth, unlike most other moth larvae, live in the soil, nipping off the stems of young plants and eating holes in succulent crops, e.g. bedding plants, lawns, potatoes, celery, turnips and conifer seedlings. The damage resembles that caused by slugs. Life cycle (see Figure 14.13). The adult moth, 2 cm across, with brown fore-wings and yellow or orange hind wings, emerges from the shiny soil-borne chestnut brown pupa from June to July, and lays about 1000 eggs on the stems of a wide variety of weeds. The first instar caterpillars, having fed on weeds, descend to the soil and in the later instars cause the damage described above, eventually reaching about 3.5 cm in length. They are grey to grey-brown in colour, with black spots along the sides. Several other cutworm species such as heart and dart moth (Agrotis exclamationis) and turnip moth (Agrotis segetum) may cause damage similar to that of the yellow underwing. In all three species, their typical caterpillar-shaped larvae should not be confused with the legless leatherjacket which is also a common underground larva. The cutworm species normally have two life cycles per year, but in hot summers this may increase to three. Spread. The larvae are able to crawl from plant to plant, but most spread is by the actively flying adults. Control: Amateur gardeners remove a good proportion of the cutworm larvae as they dig plots over. Good weed control reduces cutworm damage. A soil-directed spray of bifenthrin will achieve some control of cutworm. For the professional horticulturist , soil drenches of residual insecticides such as chlorpyrifos have proved successful against the larva stage of this pest. Leopard moth (Zeuzera pyrina) Damage: The caterpillar of this species tunnels into the branches and trunk of a wide range of tree species, such as apple, ash, birch, and lilac. The tunnelling may weaken the branches of trees which in high winds commonly break.
Control: Where tunnels are observed, a piece of wire may be pushed along the tunnel to kill the larva. Other moths worthy of mention here are the fruit-invading species such as codling moth (Cydia pomonella) on apple, plum moth (Cydia funebrana) and pea moth (Cydia nigricana) each of which needs accurately timed insecticidal control to avoid fruit damage by the pest (see also pheremone traps), because insecticidal control inside the fruit is not possible.
Flies This group of insects, of the order Diptera, are characterized by having only a single pair of functioning wings. The hind wings are modified into little stubs which act as balancing organs. The larvae are legless, elongated, and their mouthparts, where present, are simple hooks. The larvae are the only stage causing crop damage. Carrot fly (Psila rosae) Damage: This is a widespread and serious pest on umbelliferous crops (carrots, celery and parsnips). The grubs emerging from the eggs eat fine roots and then enter the mature root using fine hooks in their mouths. Damage is similar in all crops. In carrots, seedlings may be killed, while in older plants the foliage may become red, and wilt in dry weather. Stunting is often seen, and affected roots, when lifted, are riddled with small tunnels (see Figure 14.16) that make the carrots unsaleable. Damage should not be confused in carrots with cavity spot, a condition associated with a Pythium species of fungi which produces elongated sunken spots partly circling the root.
Spread. The adult is the stage which spreads the pest. Control: The Amateur gardener can use a variety of controls. Planting carrots after the May emergence has occurred reduces infestation. Covering carrot plots with horticultural fleece (see Plant protection, Figure 16.2), prevents the adut from laying eggs next to carrots. Inter-planting carrots with onions is said to prevent carrot fly from homing in on the carrot crop. For the professional grower, high levels of carrot fly can be prevented by keeping hedges and nettle beds trimmed, thus reducing sheltering sites for the flies. A seed treatment containing tefluthrin reduces early larval infestation. A ground-directed spray at a high volume rate of lambda-cyhalothrin reaches the larva in the soil. Chrysanthemum leaf miner (Phytomyza syngenesiae) Damage: The leaf miners are a group of small flies, the larvae of which can do serious damage to horticultural crops by tunnelling through the leaf. This species is found on members of the plant family Asteraceae. Plants attacked include chrysanthemum, cineraria and lettuce.
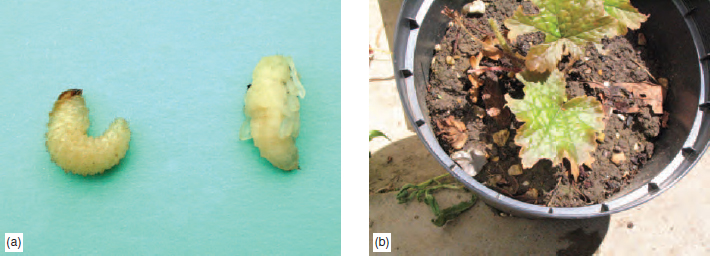
Spread. This pest is spread by the adult stage. Control: Weed hosts such as groundsel and sowthistle should be controlled. Yellow sticky traps remove many of the adult flies. Certain chrysanthemum cultivars show some resistance. Amateur gardeners have no effective insecticide product to control the larva inside the leaf. Professional growers use tiny wasps such as Diglyphus isaea and Dacnusa sibirica to parasitize the tunnelling leaf-miner larvae. Products containing abamectin may be used outdoors and in greenhouses. (The occurrence of South American leaf miner (Liriomyza huidobrensis) and American serpentine leaf miner (Liriomyza trifolii) which are able to damage a wide variety of greenhouse plants has, in recent years, created many problems for horticulture). Leatherjacket (Tipula paludosa) Damage: This is an underground pest which is a natural inhabitant of grassland and causes most problems on golf greens. After ploughing up of grassland, leatherjackets may also cause damage to the crops such as potatoes, cabbages, lettuce and strawberries. This pest is particularly damaging in prolonged wet periods when the roots of young or succulent crops may be killed off. Occasionally lower leaves may be eaten. Life cycle. The adult of this species is the crane fly, or ‘daddy-longlegs’, commonly seen in late August. The females lay up to 300 small eggs on the surface of the soil at this period, and the emerging larvae feed on plant roots during the autumn, winter and spring months, reaching a length of 4 cm by June. They are cylindrical, grey-brown in colour, legless and possess hooks in their mouths for feeding. During the summer months, they survive as a thick-walled pupa. Spread is achieved by the adults. Control: Amateur gardeners remove this pest as they dig in autumn and spring. Crops sown in autumn are rarely affected, as the larvae are very small at this time. There are no pesticide products recommended to control this pest. Professional growers and groundsmen use products containing the residual chlorpyrifos, which is drenched into soil to reduce the larval numbers. Sciarid fly (Bradysia spp.) Damage: The larvae of this pest (sometimes called fungus gnat) feed on fine roots of greenhouse pot plants such as cyclamen, orchid and freesia, causing the plants to wilt. Fungal strands of mushrooms in commercial houses may be attacked in the compost. Life cycle: The slender black females, which are about 3 mm long, fly to suitable sites (freshly steamed compost, moss on sand benches and wellfertilized compost containing growing plants), where about 100 minute eggs are laid. The emerging legless larvae are translucent-white with a black head, and during the next month grow to a length of 3 mm before briefly pupating and starting the next life cycle. Spread is achieved by the adults. Control: Amateur gardeners and Professional horticulturists use yellow sticky traps to catch the flying adults in greenhouses. The pest can be reduced by avoiding overwatering of plants. Biological control by the tiny nematode Steinernema feltiae is now available. Professional growers in mushroom houses attempt to exclude the flies from mushroom houses by means of fine mesh screens placed next to ventilator fans. A predatory mite (Hypoaspis mile) is used to control the larvae. The larvae also may be controlled by the insecticide, diflubenzuron, incorporated into composts. Beetles This group of insects in the order Coleoptera is characterized in the adult by hard, horny forewings (elytrae) which, when folded, cover the delicate hind wings used for flight. The meeting point of these hard wing cases produces the characteristic straight line down the beetles back over its abdomen. Most beetles are beneficial, helping in the breakdown of humus, e.g. dung beetles, or feeding on pest species (see ground beetle). A few beetles, e.g. wireworm, raspberry beetle and vine weevil, causes crop damage. Vine weevil (Otiorhyncus sulcatus) This species belongs to the beetle group but, as with all weevils, possesses a longer snout on their heads than other beetles. Damage. The larva stage is the most damaging, eating away roots of crops such as cyclamen and begonias in greenhouses, primulas, strawberries, young conifers and vines outdoors, causing above-ground symptoms similar to root diseases such as vascular wilt. Close inspection of the plant’s root zone will, however, quickly show the unmistakable white grubs (see Figure 14.18). The adults may eat out neat holes or leaf edges of the foliage of hosts such as rhododendron, raspberry and grapes, and many herbaceous perennials (see Figure 14.18). Several related species, e.g. the clay-coloured weevil (Otiorhyncus singularis) cause similar damage to that of the vine weevil.
Life cycle: The adult is 9 mm long, black in colour, with a rough textured cuticle (see Figure 14.19). The forewings are fused together, the pest being incapable of flight. No males are known. The female lays eggs (mainly in August and September) in soil or compost, next to the roots of a preferred plant species. Over a period of a few years, she may lay a thousand eggs as she visits many plants. The emerging larvae are white, legless and with a characteristic chestnut-brown head. They reach 1 cm in length in December when they pupate in the soil before developing into the adult.
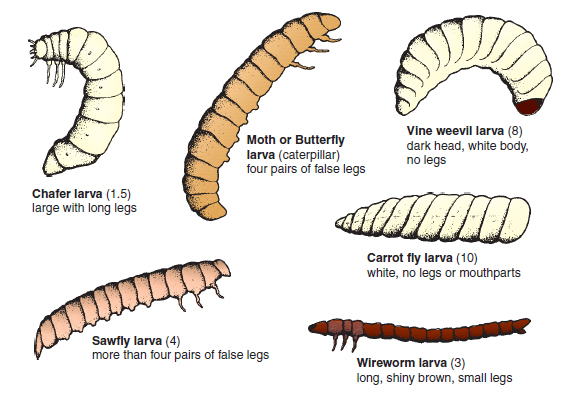
Spread is achieved by the female adult crawling around at night, or by the movement of pots containing grubs. Control: Amateur gardeners sometimes use traps of corrugated paper placed near infested crops. Inspection of plants at night by torchlight may reveal the feeding adult. Amateurs and Professionals can use the nematode ( Steinemena carpocapsae ) by incorporating it into compost or soil. Professional growers have residual chemicals, imidacloprid or chlorpyrifos incorporated or drenched into compost or soil. Wireworm (Agriotes lineatus) Damage: This beetle species is commonly found in grassland, but will attack most crops. Turf grass may be eaten away by the larvae (wireworms) resulting in dry areas of grass. The pest also bores through potatoes to produce characteristic narrow tunnels, while in onions, brassicas and strawberries the roots are eaten. In tomatoes, the larvae bore into the hollow stem. Life cycle: The 1 cm long adult (click beetle) is brown-black and has the unusual ability of flicking itself in the air when placed on its back. The female lays eggs in weedy ground in May and June and the larvae, after hatching, develop over a four year period. Fully grown wireworms are about 2.5 cm long, shiny golden-brown in colour, and possess short legs (see Figure 14.7). After a three week pupation period in the soil, usually in summer, the adult emerges and in this stage survives the winter. Spread is by means of the flighted adults. Control. Some Amateur gardeners dig in green manure crops to lure wireworms away from underground roots and tubers. Professional growers may reduce serious damage to young crops by using a seed dressing containing tefluthrin. Garden Chafer (Phyllopertha horticola) Damage: This pest is increasingly proving a problem on turf where the large white grubs eat the roots. Small yellow patches appear in the lawn or sports area, notably in summer when the grubs are becoming fully grown. Further damage can occur when starlings, crows, moles, foxes and even badgers dig up parts of the lawn to reach the succulent prey.
Spread is by means of the flighted adults. Control: For the Amateur gardener, maintenance of a healthy, wellfertilized lawn will lessen chafer attack. Groundsmen may use a biological control, involving a nematode, Heterorhabditis bacteriophora available for application during the summer. The nematode enters the chafer grub through its spiracles, and proceeds to release bacteria which digest the grub’s body. A soil-applied insecticide containing imadocloprid is used against this pest. Raspberry beetle (Byturus tomentosus) The developing fruit of raspberry, loganberry and blackberry may be eaten away by the 8 mm long, golden-brown larvae of this pest. Only one life cycle per year occurs, the larva descending to the soil in July and August, pupating in a cell from which the golden-brown adult emerges to spend the winter in the soil. The adult female lays eggs in the host flower the following June. Spread is by means of the flighted adult. Control: Since the destructive larval stage may enter the host fruit and thus escape insecticidal control, the timing of the spray is vital. In raspberries, a contact chemical such as bifenthrin, applied when the fruit is pink, achieves good control. Other beetle pests Springtime attack of flea beetle (Phyllotreta species) on leaves of young cruciferous plants (e.g. cabbages and stocks) is a serious problem to amateur and professional horticulturist alike (see Figure 14.21). In recent years, four other increasingly common beetle problems have been reported. These are viburnum beetle on Viburnum opulus, V. tinus and V. lantana, rosemary leaf beetle (Chrysolina americana) on lavender, rosemary and thyme, red lily beetle (Lilioceris lilii) on lilies, and asparagus beetle (Crioceris asparagi ).
This group, together with bees (see Pollination and fertilization), wasps and ants, are classified in the order Hymenoptera, characterized by adults with two pairs of translucent wings and with the fore- and hind-wings being locked together by fine hooks. The slender waist-like first segments of the abdomen give these insects a characteristic appearance. A sawfly larva is shown in Figure 14.22 . Gooseberry sawfly (Nematus ribesii) Damage: This is an important pest on gooseberries, redcurrants and whitecurrants, but not on blackcurrants. Extensive damage to foliage may be caused by the caterpillars. In some cases, the leaves of the whole bush may be skeletonized. Life cycle: The adults emerge from their overwintering soil-borne cocoons in late April. Adults measure about 1 cm in length and resemble flying ants. The male has a black abdomen, the female a yellow abdomen. The female, in early May, lays elongate 1 mm long, light-green eggs in rows along the veins of the leaves situated low down in the host bush. Emerging young caterpillars eat small holes in these leaves. Maturing caterpillars (reaching up to 2 cm in length and identifiable by their green appearance with numerous black spots and with a black head) consume whole leaves and move outwards and upwards from their original positions. After 3–4 weeks of feeding, the mature caterpillars drop to the soil and pupate. There are 2–3 more lifecycle generations of the sawfly from the second emergence in early June to last soil pupation in late September.
Control: Private gardeners often pick off the first few young caterpillars found on the base leaves of the bush at egg-germination times (in late April, early June, early July and late August). In autumn and winter periods, the removal of any mulch from around the bushes and disturbance of soil in the area encourage birds to seek out overwintering pupae. Insecticide products containing pyrethrins control this pest. Professional growers use two ingredients derived from plants. The insecticides, derris or nicotine, are effective against the young caterpillars if directed on the under-leaf surface, and with special attention being paid to the lower, central leaves. No biological control is currently available for gooseberry sawfly, and the insecticide formulated from the bacterium, Bacillus thuringiensis, whilst effective on many caterpillars of moths, is ineffective against this sawfly species. Rose leaf-rolling sawfly (Blennocampa pusilla) The black shiny adults, resembling winged queen ants, emerge from the soil-borne pupa in May and early June. Eggs are inserted into the leaf lamina of roses, which, in responding to the pest, rolls up tightly. The emerging larva, which is pale green with a white or brown head, feeds on the rolled foliage. It reaches a length of 1 cm by August, when it descends to the ground and forms an underground cocoon to survive the winter until it pupates in March. All types of roses are affected, although climbing roses are preferred. Damage caused by leaf-rolling tortrix caterpillars, e.g. Cacoecia oparana, may be confused with the sawfly, although the leaves are less curled. Spread of the sawfly is achieved by means of the flighted adults. Amateur gardeners may need to control this pest with an insecticide containing pyrethrins. Nursery stock growers may use products containing pirimiphos-methyl.
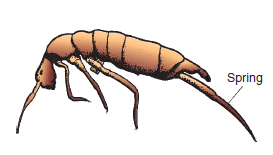
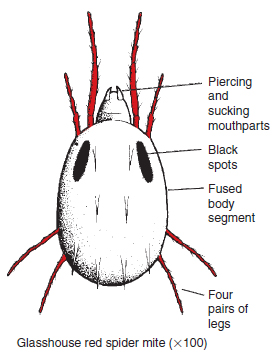
This group of primitive wingless insects, about 2 mm in length ( Figure 14.23 ) has a spring-like appendage at the base of the abdomen. They are very common in soils, and normally aid in the breakdown of soil organic matter. Two genera, Bourletiella and Collembola, however, may do serious damage to conifer seedlings and cucumber roots respectively. Spread is slow since all stages are wingless. Mite pests The mites (Acarina) are classified with spiders and scorpions in the Arachnida. Although similar to insects in many respects they are distinguished from them by the possession of four pairs of legs, a fused body structure and by the absence of wings (see Figure 14.24). Many of the tiny soil-inhabiting mites serve a useful purpose in breaking down plant debris. Several above-ground species are serious pests on plants. The life cycle is composed of egg, larva, nymph and adult stages.
Damage. The piercing mouthparts of the mites inject poisonous secretions which cause localized death of leaf mesophyll cells. This results in a fine mottling symptom on the leaf (see Figure 14.1), not to be confused with the larger spots caused by thrips. In large numbers the mites can kill off leaves and eventually whole plants. Fine silk strands are produced in severe infestations, appearing as ‘ropes’ (see Figure 14.25) on which the mites move down the plant. On flowering crops such as chrysanthemums, these ropes make the plant unsaleable. Life cycle: This pest is of tropical origin and thrives best in high greenhouse temperatures. Both species are 1 mm in length. The first species (T. urticae) is yellowish in colour, with two black spots (see Figure 14.25). The female lays about 100 tiny spherical eggs on the underside of the leaf, and after a period of three days the tiny six-legged larva moults to produce the nymph stage that resembles the adult. The life cycle length varies markedly from 62 days at 10°C, to 6 days at 35°C when the pest’s multiplication potential is extremely high. In autumn, when the daylight period decreases to 14 h and temperatures fall, egg production ceases and the fertilized females, which are now red in colour, move into the greenhouse structures to hibernate (diapause), representing foci for the next spring’s infestation.
Spread occurs when adults and nymphs crawl from plant to plant when leaves are touching. Wind currents can move mites attached to their silk strands. The small size and under-leaf habitat of the pest combine to keep its existence on introduced plants away from the gaze of growers, especially older growers. The second species (T. cinnabarinus), which is dark reddish-brown, has a similar life cycle to T. urticae, but does not hibernate. The hibernation habit of T. urticae leads to it being a common pest on annual crops such as tomatoes, cucumbers and chrysanthemums, while T. cinnabarinus is found more commonly on the perennial crops such as carnations, arums and hothouse pot plants. The two species often occur together on summer hosts. Control may be achieved in several ways. Amateur gardeners and Professional growers should carefully check incoming plants for the presence of the mite, using a hand lens if necessary. A predatory mite, Phytoseiulus persimilis is commonly introduced into cucumber, chrysanthemum and tomato crops in spring. For the Amateur there are products containing fatty acids professional, winter fumigation of greenhouse structures with chemicals such as formalin or burning sulphur kills off many of the hibernating females. A pesticide containing abamectin is commonly used. Gall mite of blackcurrant (Cecidophyopsis ribis) Damage: Mites living inside the blackcurrant bud damage the meristem and induce the bud to produce many scale leaves, which gives the bud its unusual swollen appearance (see Figure 14.26). These buds either fail to open or produce distorted leaves. In addition to the mechanical damage, the mite carries the virus responsible for reversion disease (see Horticultural diseases and disorders), which stunts the plant and reduces fruit production.
Life cycle: Unlike red spider mite, this species, sometimes called bigbud mite, is elongated in shape and is minute (0.25 mm) in size. It spends most of the year living inside the buds of blackcurrants and, to a lesser extent, other Ribes species. Breeding takes place inside the buds from June to September, and January to April. Spread. In May the mites emerge and are spread on silk threads and on the bodies of aphids to infest newly emerging buds and plants. Control: The mite is controlled in three ways. Clean planting material is essential for the establishment of a healthy crop. Pruning out of stems with big bud and destruction of reversion-infected plants slows down the progress of the pest. Amateur gardeners and Professional growers can spray a fine formulation of sulphur during the May–June period when the mites are migrating. There are no chemical ways to control the mite in the bud. Recently there has been a reported problem on hazel in UK caused by the hazel big bud mite (Phytopus avellanae). Tarsonemid mite (Tarsonemus pallidus) Damage. Distortion of developing leaves and flowers resulting from small feeding holes and injected toxins are the main symptom of this pest. This may happen to such an extent that leaves and petals are stunted and misshapen, and flowers may not open properly. Plant species affected are Amaranthus , Fuchsia , pelargonium and cyclamen (the pest is sometimes referred to as ‘cyclamen mite’) A closely related but distinct strain is found on strawberries. Life cycle: This spherical mite, only 0.25 mm in length, lives in the unex-panded buds of a wide variety of pot plants. In greenhouses, the adults may lay eggs all the year round, and the 2 weeks life cycle period can cause a rapid increase in its numbers. Spread occurs mainly on transported bulbs, corms and plants. Control: Care should be taken to prevent introduction of infested plants and propagation material into greenhouses. For the amateur, there is no recommended chemical product. For the professional grower, the contact acaricide, abamectin, is effective against the mite. Addition of a wetter/spreader may help the spray penetrate the tight-knit scale leaves of buds. Other mites Four other horticulturally important mites require a mention. The fruit tree red spider mite (Panonychus ulmi) causes serious leaf mottling of ornamental Malus and apple. Conifer spinning mite (Oligonychus ununguis) causes spruce leaves to yellow, and the mite spins a web of silk threads. Bulb-scale mite (Steneotarsonemus laticeps) causes internal discoloration of forced narcissus bulbs. Bryobia mite (e.g. Bryobia rubrioculus) attacks fruit trees, and may cause damage to greenhouse crops, e.g. cucumbers, if blown in from neighbouring trees. Other arthropods In addition to insects and mites, the phylum Arthropoda contains three other horticulturally relevant classes, the Crustacea (woodlice), Symphyla (symphilids) and Diplopoda (millipedes). The Chilopoda (centipedes) superficially resemble millipedes, but are unrelated and are useful general predators. Woodlouse (Armadillidium nasutum) The damage is confined mainly to stems and lower leaves of succulent glasshouse crops such as cucumbers, but occasionally young transplants may be nipped. A relative of marine crabs and lobsters, the woodlouse has adapted for terrestrial life, but still requires damp conditions to survive. In damp soils it may number over a million per hectare, and greatly helps the breakdown of plant debris, as do earthworms. In greenhouses, where plants are grown in hot, humid conditions, this species may multiply rapidly, producing two batches of 50 eggs per year. The adults roll into a ball when disturbed. Partial soil sterilization by steam effectively controls woodlice. Symphilid (Scutigerella immaculata) In greenhouse crops, root hairs are removed and may cause lettuce to mature without a heart. Infectious fungi, e.g. Botrytis, may enter the roots after symphilid damage. These delicate white creatures, with 12 pairs of legs, resemble small millipedes. The adult female, 6 mm long, lays eggs in the soil all the year round, and the development through larvae to the adult takes about 3 months. Symphilids may migrate 2 m down into soil during hot, dry weather. The recognition of this pest is made easier by dipping a suspect root and its surrounding soil into a bucket of water and searching for symphilids which float on the water surface. Millipedes These elongated, slow-moving creatures are characterized by a thick cuticle and the possession of many legs, two pairs to each body segment. Many species are useful in breaking down soil organic matter, but two pest species, the flat millipede (Brachydesmus superus) and a tropical species (Oxidus gracilus), can cause damage to roots of strawberries and cucumbers respectively. Centipedes These animals resemble millipedes, but are much more active. They help control soil pests by searching for insects, mites and nematodes in the soil. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||