Estimation of Reducing Sugars by the Dinitro Salicylic Acid (DNS) Method
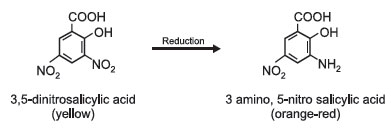
PrincipleReducing sugars have the property to reduce many of the reagents. One such reagent is 3,5-dinitrosalicylic acid (DNS). 3,5-DNS in alkaline solution is reduced to 3 amino 5 nitro salicylic acid.
Reagents Required
- Sodium potassium tartrate: Dissolve 45 gms of sodium potassium tartrate in 75 mL of H2O.
- 3,5-DNS solution: Dissolve 1.5 gm of DNS reagent in 30 mL of 2 M/liter NaOH.
- 2 molar NaOH: 80 gms of NaOH dissolved in 1 liter of water.
- DNS reagent: Prepare fresh by mixing the reagents (1) and (2) make up the volume to 150 mL with water.
- Standard sugar sodium:
(i) Stock standard sugar sodium: 250 mg of glucose in water and make up the volume to 100 mL.
(ii) Working standard sodium: Take 10 mL from this stock solution and make up the volume to 100 mL.
Procedure
Take 7 clean, dry test tubes. Pipette out standard sugar solution in the range of 0 to 3 mL in different test tubes and make up the volume of all test tubes to 3 mL with distilled water concentrations ranging from 0 to 750 mg. Add 1 mL DNS reagent to all the test tubes and mix plug the test tube with cotton or marble and keep the test tube in a boiling water bath for 5 minute. Take the tubes and cool to room temperature. Read extinction at 540 mm against the blank. Please note that all the tubes must be cooled to room temperature before reading, since the absorbance is sensitive to temperature. '
Prepare standard curves of the sugars provided and use them to estimate the concentration of the unknowns provided.
Result:
The 100 mL of unknown solution contains ........... mg of glucose.




