Determination of Iron and Manganese in Water
B. DETERMINATION OF MANGANESE
A. DETERMINATION OF IRON
AimTo determine the quantity of iron present in the given sample of water.
Principle
Iron is usually present in natural water and is not objectionable, if concentration is less than 0.3 ppm. It may be in true solution in colloidal state that may be peptized by organic matter, in the inorganic and organic iron complexes, or in relatively coarse suspended particles. It may be either ferrous or ferric, suspended or filterable. Iron exists in soils and minerals mainly as insoluble ferric oxide and iron sulphide (pyrite). It occurs in some areas, also as ferrous carbonate (siderite), which is very slightly soluble.
Phenanthroline Method
Apparatus- Colorimetric equipment; one of the following is required:
(a) Spectrophotometer, for use at 510 nm, providing a light path of 1 cm or longer.
(b) Nessler tubes, matched, 100 mL, tall form. - Glassware like conical flasks, pipettes and glass beads.
Reagents (click to check the preparation of reagents)
1. Hydrochloric acid
2. Hydroxylamine solution
3. Ammonium acetate buffer solution
4. Sodium acetate solution
5. Phenanthroline solution
6. Stock iron solution
7. Standard iron solution (1 mL = 10 µg Fe)
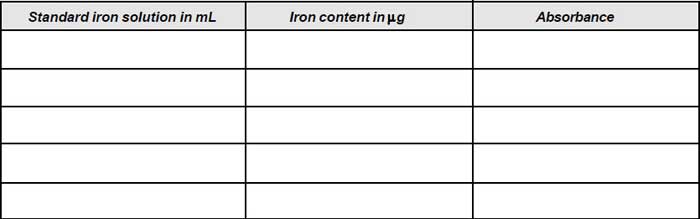
- Pipette 10, 20, 30 and 50 mL. Standard iron solution into 100 mL conical flasks.
- Add 1 mL hydroxylamine solution and 1 mL sodium acetate solution to each flask.
- Dilute each to about 75 mL with distilled water.
- Add 10 mL phenanthroline solution to each flask.
- Make up the contents of each flask exactly to 100mL by adding distilled water and left stand for 10 minutes.
- Take 50 mL distilled water in another conical flask.
- Repeat steps 2 to 5 described above.
- Measure the absorbance of each solution in a spectrophotometer at 508 nm against the reference blank prepared by treating distilled water as described in steps 6 and 7. Prepare a calibration graph taking meter reading on y-axis and concentration of iron on x-axis.
- For visual comparison, pour the solution in 100 mL tall form Nessler tubes and keep them in a stand.
- Mix the sample thoroughly and measure 50 mL into a conical flask.
- Add 2 mL conc. hydrochloric acid (HCl) and 1mL hydroxylamine solution. Add a few glass beads and heat to boiling. To ensure dissolution of all the iron, continue boiling until the volume is reduced to 15 to 20 mL.
- Cool the flask to room temperature and transfer the solution to a 100 mL Nessler tube.
- Add 10 mL ammonium acetate buffer solution and 2 mL phenanthroline solution and dilute to the 100 mL mark with distilled water.
- Mix thoroughly and allow at least 10 to 15 minutes for maximum colour development.
- Measure the absorbance of the solution in a 1cm cell in a spectrophotometer at 508 nm.
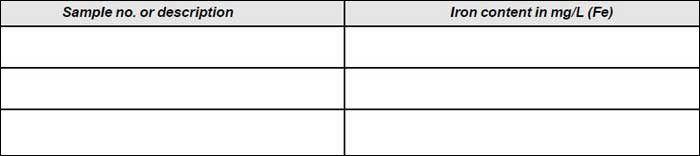
- Read off the conc. of iron (mg Fe) from the calibration graph for the corresponding meter reading.
- For visual comparison, match the colour of the sample with that of the standard prepared in steps 1 to 7 above.
- The matching colour standard will give the concentration of iron in the sample (µg Fe).
Observation

Sample Calculation
| iron (Fe) in mg/L | = μg Fe/mL of sample |
| = ____________mg/L |
B. DETERMINATION OF MANGANESE
To determine the quantity of manganese present in the given sample.
Principle
Manganese exists in the soil principally as manganese dioxide, which is very insoluble in water containing carbon dioxide. Under anaerobic conditions, the manganese in the dioxide form is reduced from an oxidation state of IV to II and solution occurs. The manganese in ground water is of divalent form. It may be present in trivalent or quadrivalent form also.
Periodate method is used when concentrations are below 0.1 mg/L. To obtain complete oxidation of small amounts of manganese, silver nitrate is added and the heating time is increased.
- Colorimetric equipment: one of the following is required:
(a) Spectrophotometer, for use at 252 nm, providing a light path of 1 cm or longer.
(b) Nessler tubes, matcheds, 100 mL tall form. - Glassware like conical flasks, measuring cylinder and pipette.
Reagents (click to check the preparation of reagents)
1. Special reagent
2. Ammonium persulphate
3. Standard manganese solution
4. Hydrogen peroxide 30%.
- Take 50 mL of the sample in a conical flask. Add 50 mL distilled water to it.
- Pipette 1, 2, 3, 4, and 8 mL of standard manganese solution to different flasks, and dilute each to 100 mL using distilled water.
- Add 5 mL special reagent to all the flasks.
- Concentrate the solutions in all the flasks to about 90 mL boiling.
- Add 1g ammonium persulphate to all the flasks, bring to boiling and boil for 1 minute.
- Remove all the flasks from the heat source and let stand for 1 minute.
- Then cool the flasks under the tap water.
- Dilute the contents in all the flasks to 100 mL with distilled water and mix. Pour the contents into 100mL Nessler tubes.
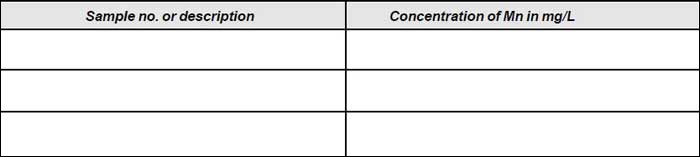
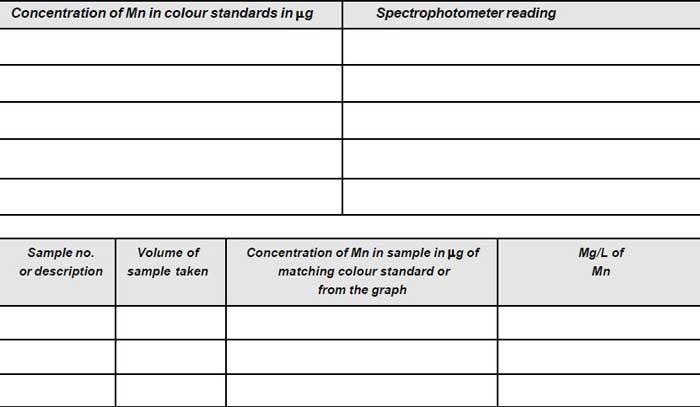
- Match the colour of the sample with that of the colour standards. Note down the concentration of Mn in µg.
- If the spectrophotometer is used, one distilled water blank has to be prepared along with the colour standards.
- Measure the absorbance of each solution in a 1cm cell at 525 nm against the reference blank prepared by treating distilled water.
- Prepare the calibration graph taking meter reading along y-axis and concentration of manganese (in µg) in colour standards on x-axis.
- Keep the sample in the spectrophotometer and note down the meter reading.
- Read off from the graph, the corresponding concentration of manganese in µg.
Observation
| Mn in mg/L = | µg of Mn |
| mL of sample |