Cyclic AMP and Ca++ mediated signal transduction.
For several decades, it is known that responses to several regulatory molecules (e.g. hormones, neurotransmitters, growth factors and cytokines like interferons) are mediated by alterations in the concentration of simple chemicals like cyclic AMP (cAMP) and calcium ions (Ca
++) within the cell. This is achieved by activation of cell surface receptors due to either the attachment of regulatory molecules (e.g. hormones) or due to other physical factors. The activation of receptor leads to activation of a G-protein, which in its turn activates the amplifier enzyme, adenylate cyclase.
This enzyme converts ATP into cAMP, leading to an increased concentration (inside the cell) of cyclic
AMP, Ca
++ or whatever the internal mediator is. Since cyclic AMP conveys the message in the interior of the cell, it was described as a
'second messenger' In most cases, cAMP activates a protein kinase A (PKA), which phosphorylates several specific enzymes (e.g. lipase for mobilization of fat; phosphorylase for enzymatic breakdown of glycogen in the liver). In addition to its role in activation of several cellular enzymes, cAMP often causes an elevation in cytosolic level of Ca
++, which is another second messenger.
Epinephrine (adrenaline hormone) also acts through cAMP to modulate the intracellular level of Ca
++ in muscles and nerve tissues.
Lipids (DAG, IP3) and Ca ++ mediated signal transduction. Some external signals have been found to stimulate the turnover of a membrane lipid and change the intracellular concentration of Ca
++. In this connection, the metabolism of
phosphatidylinositides (Pls) has been shown toplay an important role in the regulation of cell growth. It was also shown that increased PI metabolism is a reflection of signal transduction across the cell membrane. In 1980s, the activation of receptors could be linked to the rapid hydrolysis of PIs and mobilization of Ca
++ in many tissues in response to a variety of external stimuli.
This hydrolysis of a PI (phosphatidylinositol 4,5 bisphosphate =
PIP2) utilizes an amplifier enzyme, the Pi-specific
phospholipase C
(PLC) and produces various messenger molecules, of which the following three 'lipid second messengers' have been documented : (i)
1, 2-diacylglycerol (
DAG)
; (ii)
inositol 1, 4, 5,-triphosphate (
Ins 1, 4, 5, - P3 =
IP3)and (iii)
archidonic acid. Hydrolysis of PIP
2, upon binding of extracellular ligands to cell surface receptors, is also coupled to G-protein activation. Activated G-protein modulates PLC, which in turn hydrolysis PIP
2 into DAG and IP
3. Although, the signal transduction utilizing these lipid messengers has been shown to be complex,
it has been shown that DAG serves as a co-factor and together with Ca
++ activates
protein kinase C (PKC). During this process, DAG itself is metabolized to archidonic acid, a precursor of other mediators. (PKC plays a central role in the transduction of extracellular signals into a cellular response). On the other hand, IP
3 promotes the release of Ca
++ into the cytoplasm through binding to a specific receptor on the surface of endoplasmic reticulum and/or other intracellular vesicles that store Ca
++ This interaction induced a conformational change in proteins specific to Ca
++ channel and allows Ca
++ ions to exit from intracellular storage sites into the cytoplasm.
This calcium influx following stimulation of the PI signal pathway is a widespread and physiologically most important phenomenon. Ca
+ + in the cytoplasm has several roles to play in the cellular regulation, including activation of PKC by DAG. In view of the role of IP3 as a 'second messenger' in operating Ca
+ + channels as above, these Ca
++ channels have been described, in the past, as SMOCs (second messenger operated calcium channels). However, since the role of 'second messengers' is not fully established, the usage of the term SMOC has recently been questioned.
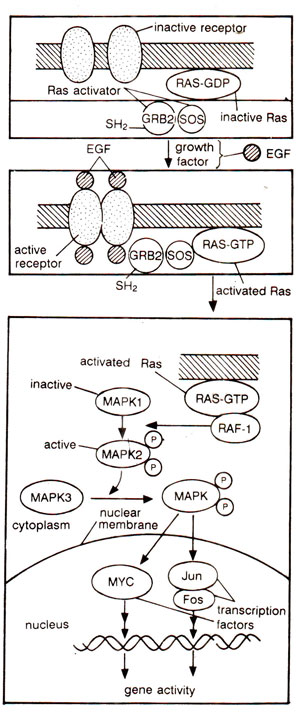
Fig. 37.23. Different steps involved in Ras mediated signal transduction pathway for inducing cell division (for details, see text).
Ras mediated signal transduction for cell division. It has been known for some years, that signals received at the cell surface are transmitted via activated tyrosine kinase (associated with the receptor) on the cell surface, through serine/threonine kinase in the cytoplasm, to the transcription factor in the nucleus (among other targets). One such signal transduction pathway, discovered fully in 1992-1993, involves a flow of signal via Ras (product of proto-oncogene
c-ras;consult
Genetics of Cancer : Proto-oncogenes, Oncogenes and Tumour Suppressor Genes for proto-oncogenes) to Raf-1 kinase (product of another oncogene,
raf-1)
, thence of MAPkinases and pp
90rsk and finally onto nuclear proto-oncogene products like c-Fos, c-Jun, etc. (MAP = mitogen activated protein; pp
90rsk = ribosomal S6 kinase).
Following steps, shown in Figure 37.23 are involved in Ras signal transduction pathway, which is the only pathway, most fully worked out : (i) EGF binds to receptor to form activated EGF-receptor complex, which is an active tyrosine kinase and phosphorylates itself; (ii) phosphorylation of receptor,
enables its internal segment (cytosolic) to bind to GRB2 protein in the GRB2-mSOS complex (GRB2 and mSOS are two intermediary proteins, which remain linked together in unstimulated cells); (iii) mSOS of the GRB2-mS0S complex now attaches to Ras, which is anchored to the inside of the cell membrane; mSOS activates Ras by changing it from inactive Ras-GDP to active Ras-GTP; (iv) Raf-1 now binds to activated Ras-GTP and gets activated; (v) Activated Raf-1 now activates MAP kinases (MAPK) forming a 'kinase cascade', (each MAPK phosphorylates, and thus activates the next member of the series); (vi) The last MAP kinase phosphorylates several cell proteins, including some transcription factors (e.g. Myc, Fos, Jun, etc.); (vii) Phosphorylation of transcription factors leads to expression of specific genes for cell division.

Fig. 37.23. Different steps involved in Ras mediated signal transduction pathway for inducing cell division (for details, see text).





