Nature and Transformations of Soil Phosphorus
Soils contain organic and inorganic phosphorus compounds. Because organic compounds are largely derived from plant residues, microbial cells, and metabolic products, components of soil organic matter are often similar to these source materials. Approximately 1% of the organic phosphorus is in the phospholipid fraction; 5 to 10% is in nucleic acids or degradation products, and up to 60% is in an inositol polyphosphate fraction (11). A significant portion of the soil organic fraction is unidentified.Phospholipids and nucleic acids that enter the soil are degraded rapidly by soil microorganisms (12,13). The more stable, and therefore more abundant, constituents of the organic phosphorus fraction are the inositol phosphates. Inositol polyphosphates are usually associated with high-molecular- weight molecules extracted from the soil, suggesting that they are an important component of humus (14,15).
Soils normally contain a wide range of microorganisms capable of releasing inorganic orthophosphate from organic phosphates of plant and microbial origin (16,17). Conditions that favor the activities of these organisms, such as warm temperatures and near-neutral pH values also favor mineralization of organic phosphorus in soils (16,18). The enzymes involved in the cleavage of phosphate from organic substrates are collectively called phosphatases. Microorganisms produce a variety of phosphatases that mineralize organic phosphate (19).
Phosphorus released to the soil solution from the mineralization of organic matter might be taken up by the microbial population, taken up by growing plants, transferred to the soil inorganic pool, or less likely lost by leaching and runoff (Figure 3.1). Phosphorus, like nitrogen, undergoes mineralization and immobilization. The net phosphorus release depends on the phosphorus concentration of the residues undergoing decay and the phosphorus requirements of the active microbial population (16).
In addition to phosphorus mineralization and immobilization, it appears that organic matter has indirect, but sometimes inconsistent, effects on soil phosphorus reactions. Lopez-Hernandez and Burnham (20) reported a positive correlation between humification and phosphate-sorption capacity. Wild (21) concluded that the phosphorus-sorption capacity of organic matter is negligible. It is observed more commonly that organic matter hinders phosphorus sorption, thereby enhancing availability. Humic acids and other organic acids often reduce phosphorus fixation through the formation of complexes (chelates) with Fe, Al, Ca, and other cations that react with phosphorus (22-24). Studies have shown that organic phosphorus is much more mobile in soils than inorganic sources (25). The interaction between the organic and inorganic phosphorus fractions is understood poorly. It is generally presumed that phosphorus availability to plants is controlled by the inorganic phosphorus fraction, although the contribution of organic phosphorus to plant nutrition should not be dismissed.
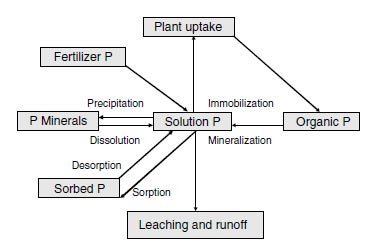 |
| FIGURE 3.1 Phosphorus cycle in agricultural soils. |
Inorganic phosphorus entering the soil solution, by mineralization or fertilizer additions, is rapidly converted into less available forms. Sorption and precipitation reactions are involved. The sorption of inorganic phosphorus from solution is closely related to the presence of amorphous iron and aluminum oxides and hydrous oxides (26-30) and the amounts of calcium carbonate (CaCO3) (24,31,32). Hydrous oxides and oxides of aluminum and iron often occur as coatings on clay mineral surfaces (27,28,33), and these coatings may account for a large portion of the phosphorus sorption associated with the clay fraction of soils. Even in calcareous soils, hydrous oxides have been demonstrated as being important in phosphorus sorption, as was demonstrated by Shukla (34) for calcareous lake sediments, Holford and Mattingly (24) for calcareous mineral soils, and Porter and Sanchez (35) for calcareous Histosols.
In calcareous soils, phosphorus (or phosphate) sorption to CaCO3 may be of equal or greater importance than sorption to aluminum and iron oxides (35). In a laboratory investigation with pure calcite, Cole (31) concluded that the reaction of phosphorus with CaCO3 consisted of initial sorption reactions followed by precipitation with increasing concentrations of phosphorus. Phosphorus sorption may occur in part as a multilayer phenomenon on specific sites of the calcite surface (24,32). As sorption proceeds, lateral interactions occur between sorbed phosphorus, eventually resulting in clusters. These clusters in turn serve as centers for the heterogeneous nucleation of calcium phosphate crystallites on the calcite surface.
Phosphorus sorption is probably limited to relatively low initial phosphorus solution concentrations and precipitation is likely a more important mechanism of phosphorus removal from the soil solutions at higher concentrations (31). Lindsay (36) identified, by x-ray crystallography, what he considered to be an incomplete list of 32 forms of phosphate compounds as reaction products from phosphorus fertilizers. The nature of the reaction products formed when phosphorus fertilizer is added to soil depends primarily on the coexisting cation, the pH of the saturated solution, the quantity of phosphorus fertilizer added, and the chemical characteristics of the soil (37). In acidic soils, aluminum and iron will generally precipitate phosphorus. In calcareous soils, an acidic fertilizer solution would dissolve calcium, and it is anticipated that most of the added phosphorus fertilizer would precipitate initially as dicalcium phosphate dihydrate (DCPD) and dicalcium phosphate (DCP) (38,39). These products are only moderately stable and undergo a slow conversion into compounds such as octacalcium phosphate, tricalcium phosphate, or one of the apatites.
As discussed above, soil transformations of phosphorus are complex and often ambiguous. Phosphorus availability has often been characterized in general terms (a) as solution phosphorus, often known as the intensity factor, (b) as readily available or labile phosphorus, often known as the quantity factor, and (c) as nonlabile phosphorus. The labile fraction might include easily mineralizable organic phosphorus, low-energy sorbed phosphorus, and soluble mineral phosphorus. The nonlabile fraction might include resistant organic phosphorus, high-energy sorbed phosphorus, and relatively insoluble phosphate minerals. As plants take up phosphorus from the solution, it is replenished from the labile fraction, which in turn is more slowly replenished by the nonlabile fraction. The soil buffer capacity, known as the capacity factor, governs the distribution of phosphorus among these pools. As will be shown in a subsequent section, although some soil tests aim to characterize only the intensity factor, most aim to characterize quantity and capacity factors as indices of phosphorus availability.




