Collection, Storage, and Preservation
Algae grow in almost every habitat in every part of the world. They can be found on very different natural substrates, from animals (snails, crabs, sloths, and turtles are algal hosts) to plants (tree trunks, branches and leaves, water plants, and
macroalgae), from springs and rivers to hypersaline lagoons and salt lakes. They also colonize artificial habitats, such as dams and reservoirs, fountains and pools, but cans, bottles, plant pots, or dishes allow algae to extend their natural range. The ubiquity of these organisms together with the plasticity of their metabolic requirements make many algal species easily available for investigation, collection, or simple observation.
Floating microalgae can be collected with a mesh net (e.g., with 25–30 µm pores) or, if in sufficient quantity (i.e., coloring the water), by simply scooping a jar through the water. A small amount of the bottom sediments will also provide many of the algal species that live in or on these sediments. Some algae live attached to other types of substrate, such as dead leaves, twigs,
and any underwater plants, which may be growing in the water. Macroalgae and the attached microalgae can be collected by hand or with a knife, including part or all of the substrate (rock, plant, wood, etc.) if possible. Algae growing on soil are difficult to collect and study, many requiring culturing before sufficient and suitable material is available for identification.
Any sample should be labeled with standard information such as the locality, date of collection, and as many of the following features as possible: whether the water is saline, brackish or fresh; whether the collection site is terrestrial, a river, a stream, or a lake; whether the alga is submerged during water level fluctuations or floods; whether the water is muddy or polluted; whether the alga is free floating or attached, and if the latter, the type of substrate to which it is attached; and the color, texture and size of the alga. Algae can be stored initially in a glass jar, plastic bottle or bag, or in a vial with some water from the collecting site. The container should be left open or only half filled with liquid and wide shallow containers are better than narrow deep jars. If refrigerated or kept on ice soon after collecting most algae can be kept alive for short periods (a day or two). If relatively sparse in the sample, some algae can continue to grow in an open dish stored in a cool place with reduced light. For long-term storage, specimens can be preserved in liquid, dried, or made into a permanent microscope mount. Even with ideal preservation, examination of fresh material is sometimes essential for an accurate determination. Motile algae particularly must be examined while flagella and other delicate structures remain intact, because any kind of preservation procedure causes the detachment of the flagella.
Commercial formalin (40% formaldehyde in water), diluted between 1/10 and 1/20 with the collecting solution, is the most commonly used fixative. As formaldehyde is considered carcinogenic, any contact with skin, eyes, and air passages should be avoided. This compound mixed with other chemicals such as glacial acetic acid and alcohol (FAA 1:1:8 by volume: 40% formaldehyde 1:glacial acetic acid 1:95% alcohol 8) gives better preservation results for some of the more fragile algae, whereas the standard alcohol and water mix (e.g., 70% ethyl alcohol) will ruin all but the larger algae. However, FAA may cause thin-walled cells to burst. Color is an important taxonomic characteristic, especially for cyanobacteria, and formalin is a good preservative for green algae, cyanobacteria and dinoflagellates because cell color remains intact if samples are stored in the dark. Algae can be kept in diluted formalin for a number of years, but the solution is usually replaced by 70% ethyl alcohol with 5% glycerin (the latter to prevent accidental drying out).
Lugol’s solution is the preferred preservative commonly used for short-term (e.g., a few months, but possibly a year or more) storage of microalgae. It is excellent for preserving chrysophytes but it makes the identification of dinoflagellates difficult, if not impossible. Samples can be preserved and kept (in dark bottles away from light) for as long as 1 yr in Lugol’s solution (0.05–1% by volume). The solution is prepared by dissolving 20 g of potassium iodide and 10 g of iodine crystals in 180 ml of distilled water and by adding 20 ml glacial acetic acid. Note that Lugol’s solution has a shelf life that is affected by light of about 6 months.
Dried herbarium specimens can be prepared by “floating out” similar to aquatic flowering plants. Ideally, fresh specimens should be fixed prior to drying. Most algae will adhere to absorbent herbarium paper. Smaller, more fragile specimens or tangled, mat-forming algae may be dried onto mica or cellophane. After “floating out,” most freshwater algae should not be pressed but simply left to air dry in a warm dry room. If pressed, they should be covered with pieces of waxed paper, plastic or muslin cloth so that the specimen does not stick to the drying paper in the press.
To examine a dried herbarium specimen, a few drops of water are added to the specimen to make it swell and lift slightly from the paper. This makes it possible to remove a small portion of the specimen with forceps or a razor blade.
Observations (preferably including drawings or photographs) based on living material are essential for the identification of some genera and a valuable adjunct to more leisurely observations on preserved material for others. The simplest method is to place a drop of the water including the alga onto a microscope slide and carefully lower a coverslip onto it. It is always tempting to put a large amount of the alga onto the slide but smaller fragments are much easier to view under a microscope. Microalgae may be better observed using the “hanging drop method,” that is, a few drops of the sample liquid are placed on a coverslip which is turned over onto a ring of paraffin wax, liquid paraffin or a “slide ring.”
A permanent slide can be prepared with staining such as aniline blue (1% aqueous solution, pH 2.0–2.5), toluidine blue (0.05% aqueous solution, pH 2.0–2.5), and potassium permanganate, KMnO
4 (2% aqueous solution), which are useful stains for macroalgae (different stains suit different species) and India Ink, which is a good stain for highlighting mucilage and some flagella-like structures. Staining time ranges from 30 sec to 5 min (depending on the material), after which the sample is rinsed in water. Mounting is achieved by adding a drop or two of glycerine solution (75% glycerine, 25% water) to a small piece of the sample placed on a microscope slide then carefully lowering the coverslip. Sealing with nail polish is essential. This method is
unsuitable for most unicellular algae which should be examined fresh or in temporary mounts of liquid-preserved material.
Magnifications of between 40 and 1000 times are required for the identification of all but a few algal genera. A compound microscope with 10x – 12x eyepiece and 4x – 10x – 40x objectives is therefore an essential piece of equipment for anyone wishing to discover the world of algal diversity. An oil immersion 100x objective would be a useful addition, particularly when
aiming at identifying to species level. Phase-contrast or interference (e.g., Nomarski) microscopy can improve the contrast for bleached or small specimens. A dissecting microscope providing 20x, 40x up to 60x magnifications is a useful aid but is secondary to a compound microscope. A camera lucida attachment is helpful for producing accurate drawings while an eyepiece micrometer is important for size determinations. Formulas for calculating biomass for various phytoplankton shapes using geometric forms and measurements and shape code for each taxa exists in the literature, and are routinely used in the procedure for phytoplankton analysis that require biovolume calculations (Table 6.1). Moreover, software packages for image recognition and analysis are available, which can process phytoplankton images acquired by means of a TV camera mounted onto a compound microscope. Scanning and transmission electron microscopes are beyond the reach of all but specialist institutions; however, they are an essential tool for identifying some of the very small algae and investigating the details of their ultrastructure.
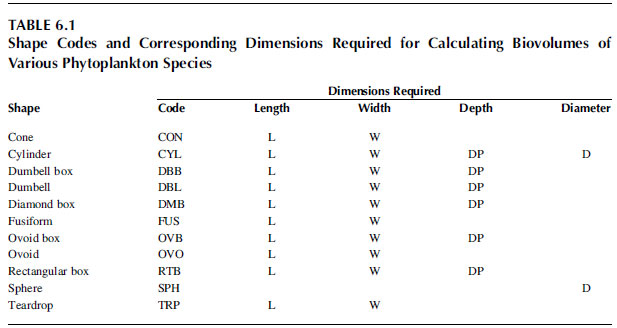
TABLE 6.1 Shape Codes and Corresponding Dimensions Required for Calculating Biovolumes of Various Phytoplankton Species





