Mutant Analysis Allowed Identification of Genes for Cellulose Synthases and Other Proteins Required for Cellulose Biosynthesis
Identification and Functional Characterization of Cellulose Synthases in Plants by Analysis of Mutants and Gene Expression StudiesAlthough a majority of the CesA and Csl genes have been identified from genome and EST sequences, at least six of the CesA genes in Arabidopsis were identified by mutant analysis. In a number of cellulose-deficient Arabidopsis mutants, the mutations were mapped to genes that encoded for cellulose synthases (Arioli et al., 1998; Fagard et al., 2000; Scheible et al., 2001; Taylor et al., 1999). Interestingly, although all the mutants exhibited different phenotypes, they all showed a deficiency in the amount of cellulose produced. The first mutant, where the mutation was identified in a gene that encoded for a cellulose synthase, was a temperature-sensitive root-swelling mutant (rsw1) (Arioli et al., 1998). At the nonpermissive temperature, the mutant produced a larger proportion of noncrystalline cellulose in place of crystalline cellulose, and the rosette terminal complexes (TCs) normally associated with cellulose microfibrils were not observed by freeze-fracture electron microscopy. The mutation in the cellulose synthase gene (rsw1 gene; AtCesA1) led to the substitution of valine for alanine at position 549 of the cellulose synthase protein and this change resulted in all the different phenotypes associated with the rsw1 mutant (Williamson et al., 2001). No biochemical changes have been characterized in the mutant protein, but it appears that at the nonpermissive temperature, the cellulose synthase is not assembled into a rosette structure. Although the mutation results in the reduction of crystalline cellulose at the nonpermissive temperature, noncrystalline cellulose still is produced suggesting that the rsw1-encoded cellulose synthase is able to synthesize the β-1,4-glucan chains, but does not allow for their assembly to take place, or alternatively these chains are synthesized by cellulose synthases encoded by other genes, where the assembly of these cellulose synthases is affected by the rsw1 mutation. Changes in cell shapes and sizes suggested that the rsw1 cellulose synthase contributed to cellulose in the primary wall. Interestingly, a number of questions still remain to be answered in terms of how the rsw1 mutation affects cellulose biosynthesis.
A number of irregular xylem mutants (irx mutants) have been isolated by screening cross-sections of stems of Arabidopsis plants (Turner and Somerville, 1997). The mutations resulted in collapse of mature xylem cells in the inflorescence stems, and in many of these mutants there was a significant decrease in the amount of cellulose in the secondary cell wall of cells in the xylem. Genes mutated in some of the irx mutants were identified to encode for cellulose synthases. The null mutation in the irx3 mutant results in a stop codon that truncates the cellulose synthase (irx3; AtCesA7) by 168 amino acids (Taylor et al., 1999) In two irx1 mutants (irx1-1 and irx1-2), the mutations were mapped to a different cellulose synthase gene that altered the amino acids at positions 683 (D683N) in irx1-1 and 679 (S679L) in irx1-2 (Taylor et al., 2000). Both these amino acid positions reside within the conserved region of the irx1 cellulose synthase (AtCesA8). RNA analysis indicated that irx1 and irx3 are highly expressed in stems but not in leaves, suggesting that both genes are involved in cellulose synthesis during secondary cell wall formation. Examination of the phenotypes of the xylem elements by electron microscopy showed that the same cell type is affected in the irx1 and irx3 mutants, indicating that products of both the irx1 and irx3 genes are required within the same cell for normal cellulose synthesis during secondary cell wall formation (Taylor et al., 2000). These results allowed development of the concept regarding the nonredundant nature of cellulose synthases and the requirement of more than a single cellulose synthase in each cell for normal cellulose synthesis. Using biochemical and immunological methods, Taylor et al. (2000) furthermore demonstrated that the irx1 and irx3 cellulose synthases associate with each other, and suggested that this association is required for cellulose synthesis (Taylor et al., 2000). Even as different models to explain the requirement of two different cellulose synthases for cellulose synthesis were being proposed, another gene (irx5) encoding for a different cellulose synthase (irx5; AtCesA4) was identified in a further screen of irx mutants and it was found that the irx1, irx3, and irx5 genes were coexpressed in the same cells (Perrin, 2001; Taylor et al., 2003). Using detergent-solubilized extracts, the proteins encoded by these three genes were shown to interact with each other, and it was suggested that all three gene products probably are required for the formation of the cellulose-synthesizing complexes (rosette TCs) in plants. Interestingly, the presence of all three cellulose synthases (AtCesA8, AtCesA7, and AtCesA4), but not their activity, is required for correct assembly and targeting of the cellulose-synthesizing complex during secondary wall cellulose synthesis (Taylor et al., 2004). Overall, the irx mutants have been crucial in not only identifying the cellulose synthase genes that are required for cellulose synthesis during secondary wall formation, but also in formulating the concept that the assembly of the cellulose-synthesizing complexes (rosette TCs) in plants requires more than a single isoform of cellulose synthase. Fig. 6.3 shows immunogold labeling of the rosette TCs from Vigna angularis using an antibody to a cellulose synthase.
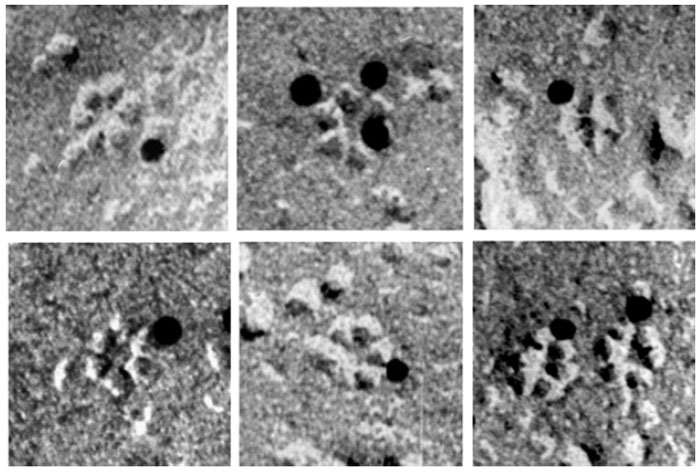 |
| FIGURE 6.3 Rosette terminal complexes from V. angularis that were immunogold labeled with an antibody to cellulose synthase. (Reproduced from Kimura, S., Laosinchai, W., Itoh, T., Cui, X., Linder, R., and Brown, R. M., Jr. (1999). Plant Cell 11, 2075–2085.) |
The protein regulator of cytokinesis 1 (PRC1) gene in Arabidopsis encodes AtCesA6, and like the rsw1 mutant of AtCesA1, mutation in this gene exhibits decreased cell elongation, especially in roots and dark-grown hypocotyls, because of cellulose deficiency in the primary wall (Fagard et al., 2000). In addition to similar mutant phenotypes, both AtCesA1 and AtCesA6 also show similar expression profiles in various organs and growth conditions suggesting coordinated expression of at least two distinct cellulose synthases (AtCesA1 and AtCesA6) in most cells (Fagard et al., 2000). However, differences were observed in the embryonic expression of these two CesA genes (Beeckman et al., 2002). Mutations in the ixr1 and ixr2 genes confer resistance to the cellulose synthesis inhibitor isoxaben and these two genes encode AtCesA3 and AtCesA6, respectively (Desprez et al., 2002; Scheible et al., 2001). The cellulose synthases identified by analysis of the rsw1, ixr1, and PRC1/ixr2 mutants involve members of the CesA family (AtCesA1, AtCesA3, and AtCesA6) required for primary wall cellulose synthesis. Although no physical interactions have been determined for these cellulose synthases, studies of inhibition of cellulose synthesis by isoxaben suggest that AtCesA3 and AtCesA6 together form an active protein complex in which the involvement of AtCesA1 may be required (Desprez et al., 2002).
Brittle culm mutants have been identified in barley, maize, and rice. The cellulose content in the cell walls of cells in the brittle culm mutants of barley was found to be lower than the wild-type plants, but no significant differences were found in the amount of the noncellulosic components of the cell wall (Kokubo et al., 1989, 1991). Brittle culm mutants in rice were useful in identifying three CesA genes (OsCesA4, OsCesA7, and OsCesA9) (Tanaka et al., 2003). The three genes are expressed in seedlings, culms, premature panicles, and roots, but not in mature leaves. The expression profiles are almost identical for these three genes, and decrease in the cellulose content in the culms of null mutants of the three genes indicates that these genes are not functionally redundant (Tanaka et al., 2003).
Identification of Other Genes/Proteins Which May be Required for Cellulose Biosynthesis in Plants
The role of β-1,4-endoglucanase during cellulose synthesis was first proposed by Matthysse et al. (1995a,b) during analysis of cellulose-minus mutants in Agrobacterium tumefaciens (Matthysse et al., 1995a,b). In this bacterium, cellulose synthesis is suggested to proceed via the formation of lipid-linked intermediates, and a β-1,4-endoglucanase is predicted to function as a transferase in the transfer of β-1,4-linked glucan oligomers from a lipid carrier to the growing cellulose chain (Matthysse et al., 1995a). The gene encoding β-1,4-endoglucanase is organized with the cellulose synthase gene in an operon in A. tumefaciens, and a similar organization of these genes is observed in a number of other bacteria (Matthysse et al., 1995b; Römling, 2002). The organization of a β-1,4-endoglucanase gene with the cellulose synthase gene in the same operon in bacteria has been taken as an indication that β-1,4-endoglucanase probably has a role during cellulose synthesis. So far, there is no direct demonstration for this role in bacteria or any other organism. A gene encoding a membrane-anchored β-1,4-endoglucanase called Korrigan also has been identified in a dwarf mutant of Arabidopsis (Nicol et al., 1998). In plants, the Korrigan protein is believed to function during primary or secondary wall cellulose synthesis (Lane et al., 2001;Mølhøj et al., 2002; Nicol et al., 1998; Sato et al., 2001; Szyjanowicz et al., 2004; Zuo et al., 2000). Its exact function during cellulose synthesis remains to be determined, although various roles have been assigned to it such as terminating or editing the glucan chains emerging from the cellulose synthase complex before their crystallization into a cellulose microfibril. Alternately it could cleave sterol from the sterol-glucoside primer that is suggested to initiate glucan chain formation (Peng et al., 2002). However, recent evidence does not support this role (Scheible and Pauly, 2004). A membrane-bound sucrose synthase, which converts sucrose to UDP-glucose, may be physically linked to the cellulose synthase complex for channeling UDP-glucose to the cellulose synthase in plants, and suppression of this gene has been shown to effect cotton fiber initiation and elongation (Amor et al., 1995; Ruan et al., 2003).
Proteins that may indirectly influence cellulose biosynthesis include those that are required for N-glycan synthesis and processing (Lukowitz et al., 2001). One of these proteins is glucosidase I, which trims off the terminal β-1,2-linked glucosyl residue from N-linked glycans and is involved in the quality control of newly synthesized proteins that transit through the endoplasmic reticulum (ER) (Boisson et al., 2001; Gillmor et al., 2002). Another protein could be glucosidase II that removes the two internal β-1,3-linked glucosyl residues subsequent to the action of glucosidase I in the quality control pathway (Burn et al., 2002b). Other proteins that influence cellulose production include KOBITO, a membraneanchored protein of unknown function that is suggested to be a part of the cellulose synthase complex, and COBRA, a putative glycosylphosphatidylinositol (GPI)-anchored protein, which upon being inactivated, dramatically reduces culm strength in rice (Li et al., 2003b; Pagant et al., 2002; Schindelman et al., 2001).




