Major nutrients
Content
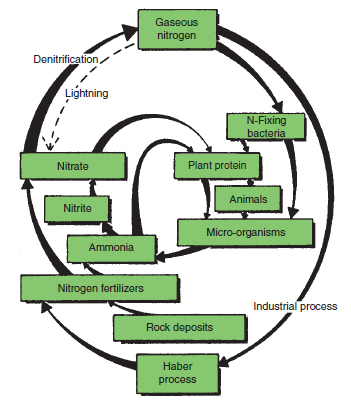
Nitrogen Nitrogen is taken up by plants as the nitrate and, to a lesser extent, the ammonium ion. Nitrates and ammonium ions are utilized in the plant to form protein. Plants use large quantities of nitrogen; it is associated with vegetative growth. Consequently large dressings of nitrogen are given to leafy crops, whereas fruit, flower or root crops require limited nitrogen balanced by other nutrients to prevent undesirable characteristics occurring. The nitrogen cycle Although plants live in an atmosphere largely made up of nitrogen they cannot utilize gaseous nitrogen. They are able to take up soluble nitrogen from the soil water as nitrates and ammonium ions. Both are derived from proteins by a chain of bacterial reactions as shown in Figure 21.2.
Ammonifying bacteria convert the proteins they attack to ammonia. Ammonia from the breakdown of protein in organic matter or from inorganic nitrogen fertilizers is converted to nitrates by nitrifying bacteria. This is accomplished in two stages. Ammonia is first converted to nitrites by Nitrosomonas spp. Nitrites are toxic to plants in small quantities, but they are normally converted to nitrates by Nitrobacter spp. before they reach harmful levels. Ammonifying and nitrifying bacteria thrive in aerobic conditions. Where there is no oxygen, anaerobic organisms dominate. Many anaerobic bacteria utilize nitrates and in doing so convert them to gaseous nitrogen. This denitrification represents an important loss of nitrate from the soil, which is at its most serious in well-fertilized, warm and waterlogged land. Nitrogen fixation Although plants cannot utilize gaseous nitrogen, it can be converted to plant nutrients by some micro-organisms. Azotobactor are free-living bacteria that obtain their nitrogen requirements from the air. As they die and decompose, the nitrogen trapped as protein is converted to ammonia and then to nitrates by other soil bacteria. Rhizobia spp. which live in root nodules on some legumes (see Figure 21.3) also trap nitrogen to the benefit of the host plant. Finally, nitrogen gas can be converted to ammonia industrially in the Haber process, which is the basis of the artificial nitrogen fertilizer industry. Excess nitrogen produces soft, lush growth making the plant vulnerable to pest attack and more likely to be damaged by cold. Very large quantities of nitrogen are undesirable since they can harm the plant by producing high salt concentrations at the roots (see conductivity) and are lost by leaching. Large quantities are usually applied as a split dressing, e.g. some in base dressing and the rest in one or more top dressings. Nitrates are mobile in the soil, which makes them vulnerable to leaching. In the British Isles it is assumed that all nitrates are removed by the winter rains so that virtually none are present until the soils warm up and nitrification begins or artificial nitrogen is applied (see nitrogen cycle). Nitrates leached through the root zone may find their way into the groundwater that is the basis of the water supply in some areas. Nitrification also leads to the loss of bases; for every 1 kg N in the ammonia form that is oxidized to nitrate and leached, up to 7 kg of calcium carbonate or its equivalent is lost. Nitrogen is also lost from the root zone by denitrification, especially in warm, waterlogged soil conditions. When in contact with calcareous material, ammonium fertilizers are readily converted to ammonia gas which is lost to the soil unless it dissolves in surrounding water. For this reason urea or ammonia-based fertilizers should not be applied to such soils as a top dressing or used in contact with lime. Nitrogen fertilizers used in horticulture and their nutrient content are given in Table 21.2.
Ammonium nitrate is now commonly used in horticulture. In pure form it rapidly absorbs moisture to become wet; on drying it ‘cakes’ and can be a fire risk. Pure ammonium nitrate can be safely handled in polythene sacks and as prills. Ammonium sulphate has a highly acid reaction in the growing medium. Urea has a very high nitrogen content and in contact with water it quickly releases ammonia. Its use as a solid fertilizer is limited, but it is utilized in liquid fertilizer or foliar sprays. The addition of a sulphur coating to urea not only creates a controlled release action, but also a fertilizer with an acid reaction. Other manufactured organic fertilizers include urea formaldehydes (nitroform, ureaform, etc.) which release nitrogen as they are decomposed by microorganisms, isobutylidene urea (IBDU) which is slightly soluble in water and releases urea and crotonylidene (CDU, e.g. Crotodur). The latter breaks down very slowly and evenly, which makes it ideal for applying to turf. Natural organic sources of nitrogen, including dried blood, hoof and horn and shoddy, amongst others, are generally considered to provide slow release nitrogen, but in warm greenhouse conditions decomposition is quite rapid. Phosphorus Phosphorus is taken up by plants in the form of the phosphate anion H2PO43-. Phosphorus is mobile in the plant and is constantly being recycled from the older parts to the newer growing areas. In practice this means that, although seeds have rich stores of phosphorus, phosphate is needed in the seedbed to help establishment. Older plants have a very low phosphate requirement compared with quick growing plants harvested young. Most soils contain very large quantities of phosphorus, but only a small proportion is available to plants. The concentration of available phosphate ions in the soil water and on soil colloids is at its highest between pH 6 and 7. Phosphorus is released from soil organic matter by micro-organisms (see mineralization), but most of it and any other soluble phosphorus, including that from fertilizers, is quickly converted to insoluble forms by a process known as phosphate fixation. Insoluble aluminium, iron and manganese phosphates are formed at low pH and insoluble calcium phosphate at high pH. The carbonic acid in the vicinity of respiring roots and organisms in the rhizosphere, such as mycorrhizae, facilitate phosphorus uptake. The low solubility of phosphorus in the soil makes it virtually immobile, with the result that roots have to explore for it. Soils should be cultivated to allow roots to explore effectively; compacted or waterlogged areas deny plants phosphorus supplies. Phosphate added to the soil should be placed near developing roots (see band placement) in order to reduce phosphorus fixation and ensure that it is quickly found. If applied to the surface, phosphate fertilizers should be cultivated into the root zone. Unlike soils, most artificial growing media have no reserves of phosphorus and when added in soluble form it remains mobile and subject to leaching. Incorporating phosphorus in liquid feeds in hard water is complicated by the precipitation of insoluble calcium phosphates that lead to blocked nozzles. Slow release phosphates are often selected in these situations to reduce losses and to eliminate the need for phosphorus in the liquid feeds. Phosphorus nutrition used to be based on organic sources such as bones, but now phosphate fertilizers are mainly derived from rock phosphate ore (see Table 21.2). Slow-acting forms, such as rock phosphate, bone meal and basic slag, can be analysed in terms of their ‘citric soluble’ phosphate content, this being a good guide to their usefulness in the first season. Such materials should be finely ground to enhance their effectiveness. These phosphates are applied mainly to grassland, tree plantings and in the preparation of herbaceous borders, to act as longterm reserves, particularly on phosphate deficient soils. Magnesium ammonium phosphate (MagAmp, Enmag), calcium metaphosphate and potassium metaphosphate contain other nutrients, but are slow release phosphates for use in soilless growing media. Treating rock phosphates with acids produces water-soluble phosphates. Superphosphate, derived from rock phosphate by treating with sulphuric acid, is composed of a water-soluble phosphate and calcium sulphate (gypsum), whereas triple superphosphate, derived from a phosphoric acid treatment, is a more concentrated source of phosphorus with fewer impurities. Both superphosphate and triple superphosphate are widely used in horticulture and are available in granular or powder form. Whilst they have a neutral effect on soil pH they tend to reduce the pH of composts. High-grade monoammonium phosphate is used as a phosphorus source in liquid feeds because it is low in iron and aluminium impurities that lead to blockage in pipes and nozzles.
Potassium Potassium is taken up by the roots as the potassium cation and is distributed throughout the plant in inorganic form where it plays an important role in plant metabolism. For balanced growth the nitrogen to potassium ratio should be 1:1 for most crops, but 2:3 for roots and legumes. Leafy crops take up large amounts of potassium, especially when given large amounts of nitrogen. Where potassium supplies are abundant some plants, especially grasses, take up ‘luxury’ levels, i.e. more than needed for their growth requirements. Consequently, if large proportions of the plant are taken off the land, e.g. as grass clippings, there is a rapid depletion of potassium reserves. Potassium forms part of clay minerals and is released by chemical weathering. The potassium in soil organic matter is very rapidly recycled and exchangeable potassium cations, held on the soil colloids and in the soil solutions, are readily available to plant roots. Potassium is easily leached from sands low in organic matter and from most soilless growing media (see Figure 21.4). Potassium and magnesium ions mutually interfere with uptake of each other. This ion antagonism is avoided when the correct ratio between 3:1 and 4:1 available potassium to magnesium is present in the growing medium. Availability of potassium is also reduced by the presence of calcium (see induced deficiency). The main potassium fertilizers used in horticulture are detailed in Table 21.2. Although cheaper and widely used in agriculture, potassium chloride causes scorch in trees and can lead to salt concentration problems because the chloride ion accumulates as the potassium is taken up. Commercial potassium sulphate can be used in base dressings for composts, but only the more expensive refined grades should be used in liquid feeding. More usually potassium nitrate is used to add both potassium and nitrate to liquid feeds, but it is hygroscopic. Most potassium compounds are very soluble so that the range of slow release formulations is limited to resin-coated compounds.
Magnesium Magnesium is an essential plant nutrient in leaves and roots and is taken up as a cation. There are large reserves in most soils, especially clays, and those soils receiving large dressings of farmyard manure. Deficiencies (see Figure 21.5) are only likely on intensively cropped sandy soils if little organic manure is used. Magnesium ion uptake is also interfered with by large quantities of potassium or calcium ions because of ion antagonism. Chalky and over-limed soils are less likely to yield adequate magnesium for plants. Magnesium fertilizers include magnesian limestones containing a mixture of magnesium and calcium carbonate that raise soil pH (see liming). Magnesium sulphate, as kieserite, provides magnesium ions without affecting pH levels and in a purer form, Epsom salts, is used for liquid feeding and foliar sprays Figure 21.5 Magnesium deficiency (see Table 21.2). Calcium Calcium is an essential plant nutrient taken up by the plant as calcium cations. Generally a satisfactory pH level of a growing medium indicates suitable calcium levels (see liming). Gypsum (calcium sulphate) can be used where it is desirable to increase calcium levels in the soil without affecting soil pH. Deficiencies are infrequent and usually caused by lime being omitted from composts. Inadequate calcium in fruits is a more complex problem involving the distribution of calcium within the plant. Calcium nitrate or chloride solutions can be applied to apples to ensure adequate levels for safe storage (see plant tissue analysis). Sulphur Sulphur taken up as sulphate ions is a nutrient required in large quantities for satisfactory plant growth. It is not normally added specifically as a fertilizer because the soil reserves are replenished by re-circulated organic matter and a steady supply from winds off the sea in the form of dimethyl sulphide (DMS) that gives the distinctive smell of the seaside. Air pollution has added considerably to the supply reaching the land. Several fertilizers used to add other nutrients are in sulphate form, e.g. ammonium sulphate, superphosphate and potassium sulphate, and as such supply sulphur as well (see Table 21.2). However, as air pollution is reduced and fewer sulphate fertilizers are used, it is becoming necessary for growers in some parts of the world to take positive steps to include sulphur in their fertilizer programme. |
||||||||||||||||||||||||||||