Gene Cloning in Eukaryotes
Yeasts
Generally bacterial cultures are suitable for the production of polypeptides on a large scale, but not plant or animal cells, because of their slow growth rate. But certain limitations with bacteria have forced to develop its alternative and, to some extent, it has been overcome with the use of yeast {Saccharomyces cerevisiae). Yeast is an unicellular eukaryotic fungus containing a small well characterized genome. Unlike plant or animal cells, it has rather fast growth rate and itself is a non-pathogenic fungus. Most of its gene contain introns which are spliced during purification of mature mRNA.fr appears that introns found in yeast contain sequences for correct splicing as they are totally absent in higher eukaryotes. Moreover, yeasts can carry out post-translational modifications such as (removal of a signal sequence from a precursor polypeptide after the secretion of cells. This reveals a major advantage of yeasts over the bacteria.
Yeast cells contain their own plasmid (Begg, 1978) known as '2mm circle plasmid' which is used as vector for foreign genes. It is present in many stains in 50-100 copies per cell.
The foreign DNA taken up by yeast is integrated by a specific crossing over to yeast DNA containing a homologous sequence on chromosome recognized by the yeast as the origin of replication (ori). /In some cases segments of yeast DNA linked with foreign DNA have been demonstrated to transform very efficiently (about 104 cells) (Hsiao and Carbon 1979). The transforming molecules replicate autonomously within the cell DNA sequences of such property are known as Ars (autonomously replicating sequence) fragment.
Shuttle Vector
A shuttle vector is constructed by using bacterial origin of replication in a yeast plasmid which can be manipulated and cloned in bacteria. Finally they are transferred into yeast cells for the possible expression of eukaryotic genes (Fig. 4.8). The gene for B-lactamase is an example of a prokaryotic gene that is expressed in yeast. Kado and Tait (1983) have described the properties of gene shuttle vector as below :
- The vector must replicate in many organism (e.g. bacteria, yeast and plants) to facilitate the isolation and characterization of genes;
- The vector must be easily recognized by selectable markers;
- The vector should be small in size to accommodate DNA inserts;
- Cloned genes should be easily detected;
- Useful quantities of vector must be easily obtained;
- The vector must be stable, non-pathogenic and non-stress-inducing;
- The vector must effectively deliver genetic information for stable maintenance in alternate derived reciptients;
- The introduced genetic information should be stably maintained as a new heritable determinant.
Transformation in Filamentous Fungi
Transformation through plasmid of E. coli was first described in protoplasts of S. cerevisiae (Hinnen et al., 1978) and then in N. crassa. However, the situation in filmentous fungi is different from the others as there is no convincing evidence for autonomously replicating plasmids. But sequence having functions similar to ARS have been isolated.
Application of Transformation of Fungal Protoplasts
Mainly the transformation approaches are used (i) for the analysis of promoters and construction of expression vectors, and (ii) biotechnological application in the expression of foreign genes in fungi. Analysis of promoter involves the construction of vector which have different pieces of promoters. These can be cut by restriction enzymes and joined with DNA ligase. By this method changes in promoter region are also possible and this can be achieved through mutagenesis. Thus, a strain can be improved resulting in overproduction of the products (Peberdy, 1989).
Adopting t
Several companies in the U.S.A. and Europe have demonstrated the expression of foreign DNA introduced in Aspergillus sp. through transformation. Upshall et al. (1987) have constructed a plasmid pMl59 for the secretion of human tissue plasminogeil activater by A. nidulans. Yield of this protein was about 100mg/liter culture filtrate. Similarly, Gwynne et al. (1987) found the secretion of human interferon by transformed A. nidulans mycelia to about 1 mg/liter.
Agrobacterium plasmids
Several groups of plants have received attention as a means for DNA cloning and expression of foreign DNA in plant cells. The DNA viruses of plants are of two types: (i) double stranded DNA viruses, such as caulimoviruses, and (ii) single stranded DNA virus, for example, geminivirus. Although plant viruses have major scope for the development as cloning vector, yet this aspect has not been studied so far. A good deal of work is done on soil-borne bacteria, Agrobacterium tumifaciens causing crown gall disease and A. rhizogenes causing hairy root disease on the stems of numerous plants.
A. tumifaciens contains a large plasmid which induces tumour in plants, therefore, the plasmid is called as Ti-plasmid. The size of Ti-plasmid ranges between 180 - 250 Kb. It contains T-DNA region of about 23 to 25 Kb which is transferred into plant cells. Ti-plasmid can be grouped into three on the basis of opine types, for example, octopine, nopaline and agropine. Structural formulae and diagram of these are given in Figs. 4.10 and 4.11 respectively.
Left side of T-DNA is known as TL-T-DNA and right side of the same as TR-T-DNA. Both the essential (T-DNA) and non-essential regions of Ti-plasmids (both octopine and nopaline types) contain many genes. One or more unusual aminoacid derivatives encoded by DNA is known as opines. Opines are neither found normally in plants nor required by plants.
A. tumifaciens uses it as a source of carbon and nitrogen for their growth and multiplication. The genes responsible for the biosynthesis (ocs gene for octopine and nos for nopaline) and degradation (occ for octopine and noc for nopaline catabolism) of opines are located at the well defined regions. Arc genes are required for arginin catabolism. The plasmid also carries transfer (tra) gene to help the transfer T-DNA from one bacterium to other bacterial or plant cells, one gene for oncogenecity, ori gene for origin of replication and inc gene for incompatibility.
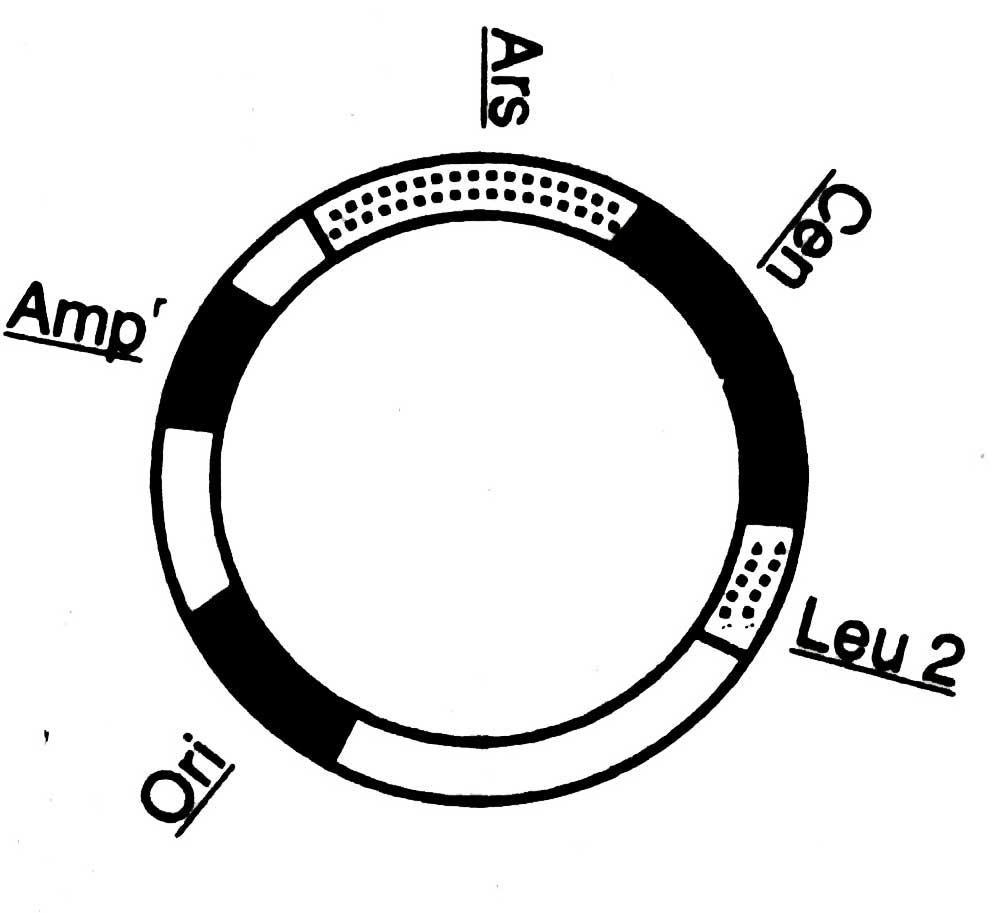
Fig. 4.8. Map of shuttle vector constructed by yeast and E.coli. Ars. autonomously replicating sequence; Cen, centromere of yeast; leu 2, complements of a defective gene encoding for leucin; on. origin of replication in prokaryote; Ampr, amphcillin resistance
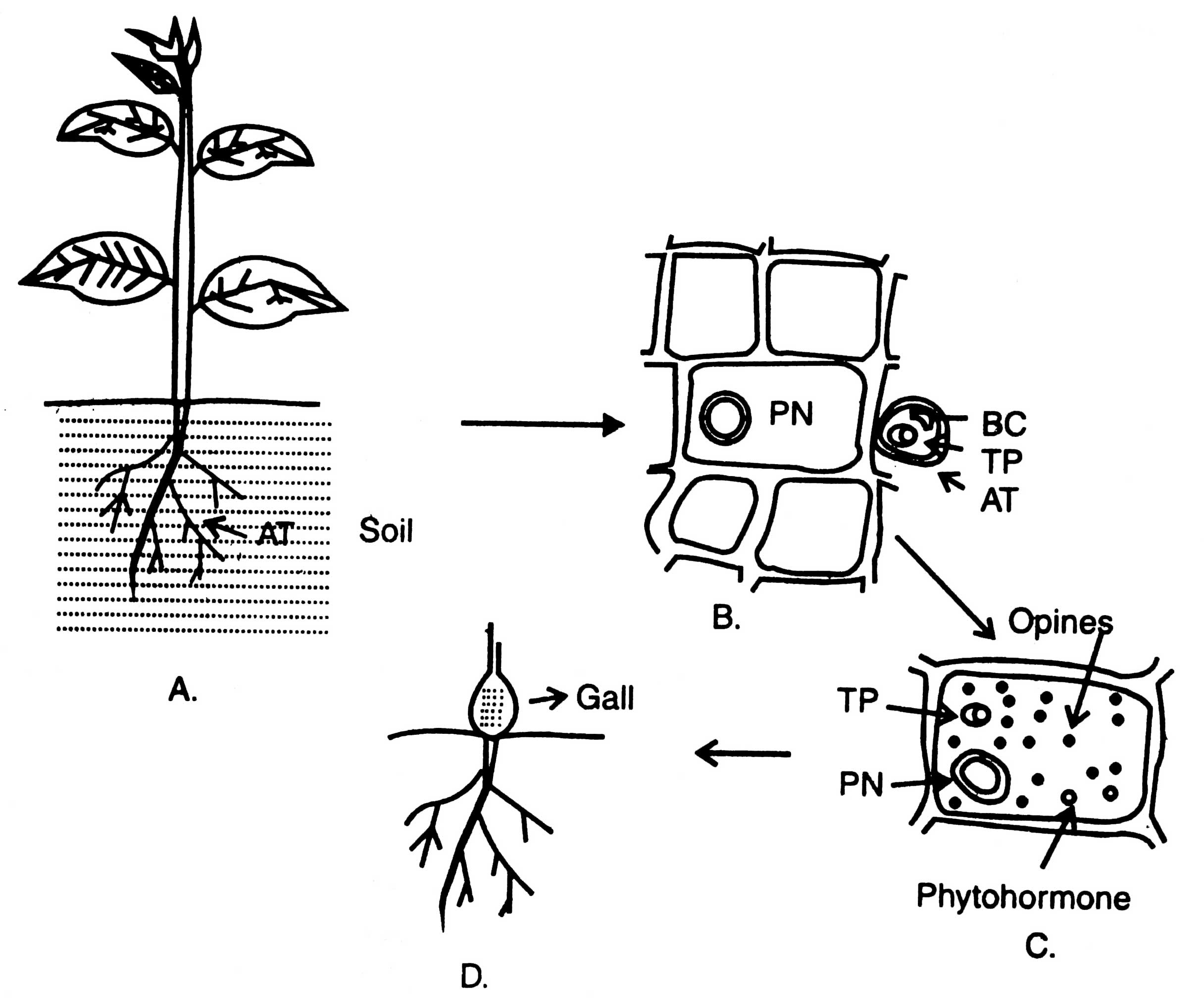
Fig. 4.9. Process of infection by Agrobacterium tumifaciens (AT) on a dicot plant and induction of tumour or gall (A-D) TP, T-DNA of plasmid, PN, plant nucleus; BC, bacterial chromosome.
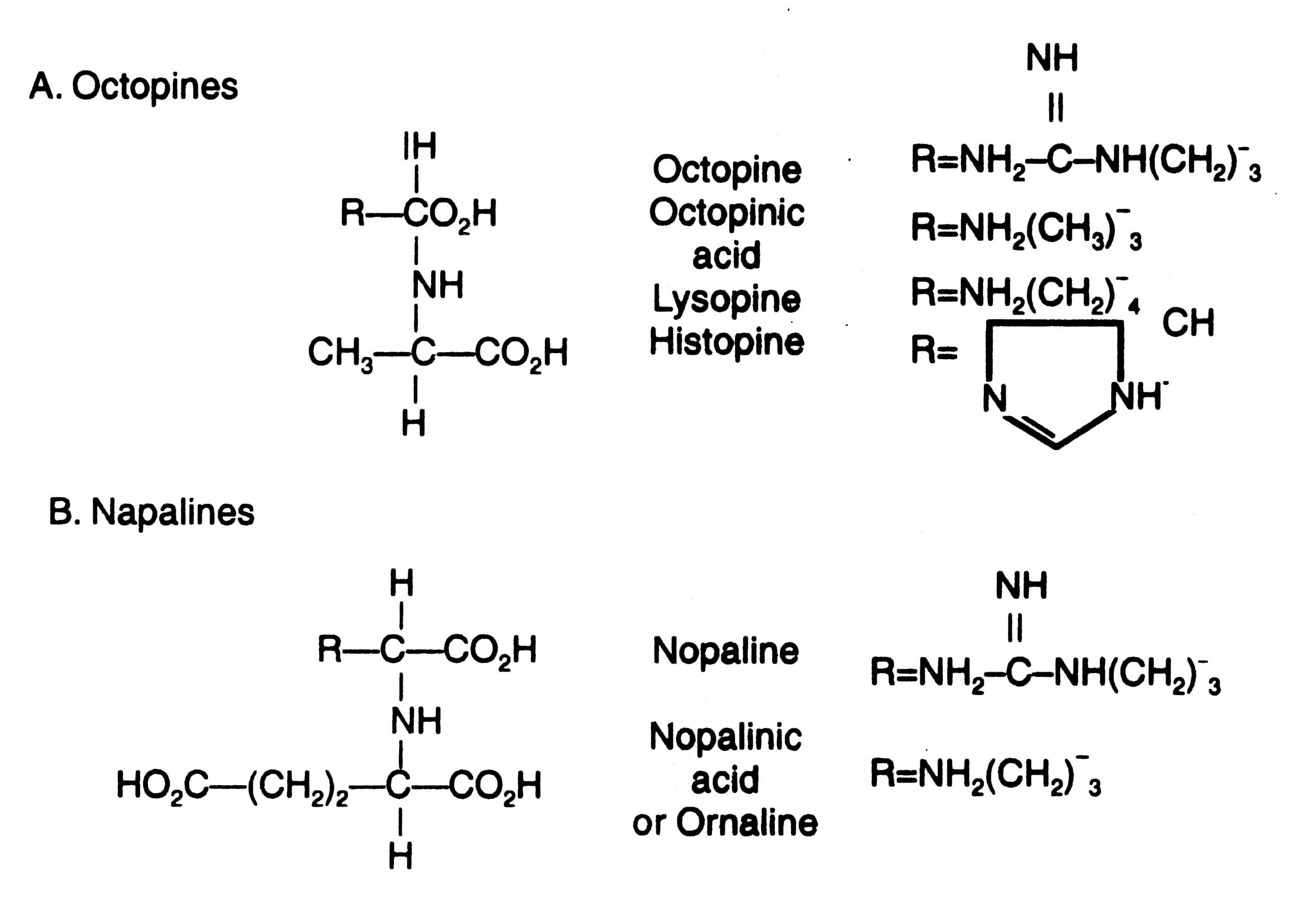
Fig. 4.10. Structural formulae of octopines (A) and nopalines (B).
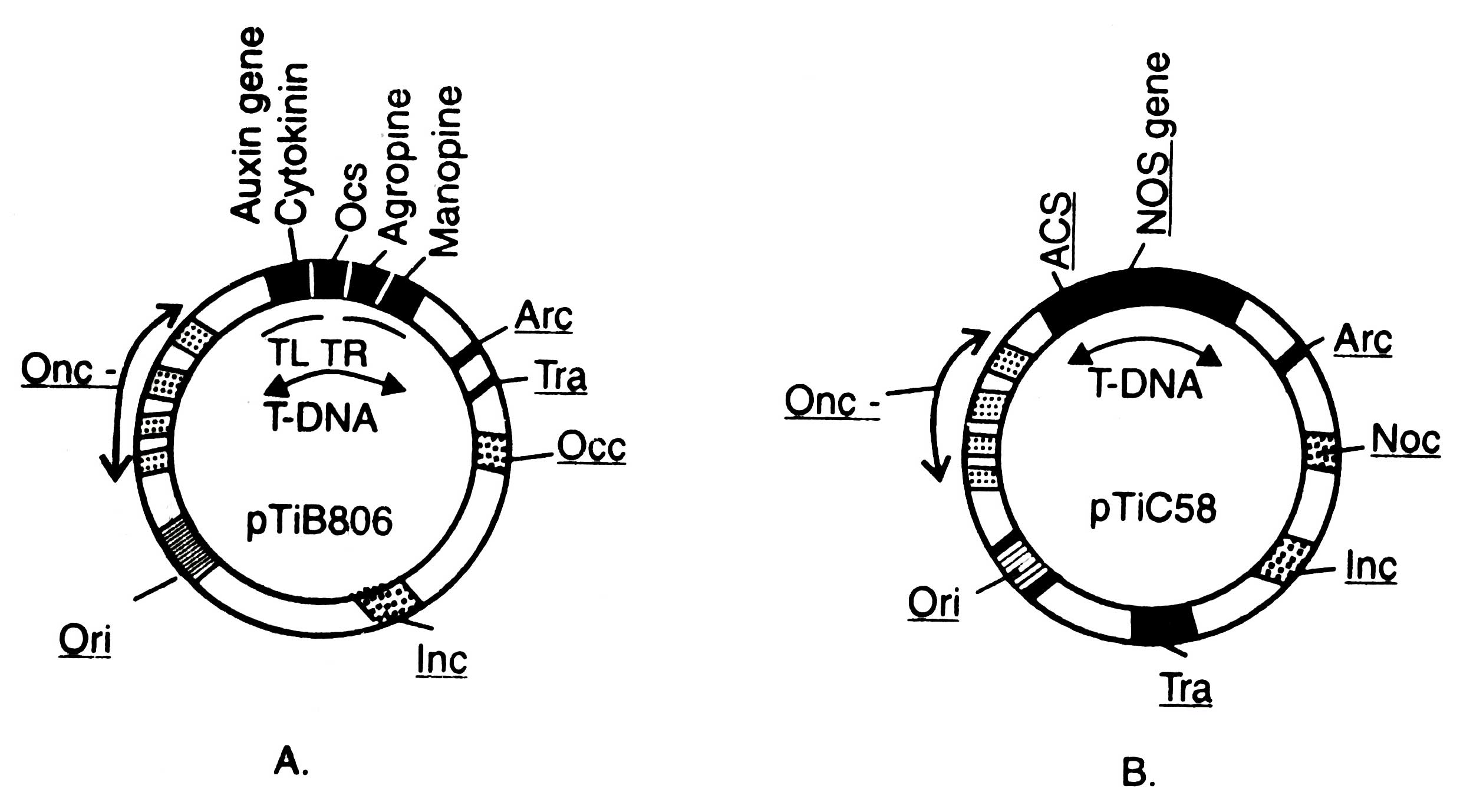
Fig. 4.11. A map of octopine plasmid (A) and nopaline plasmid (B).
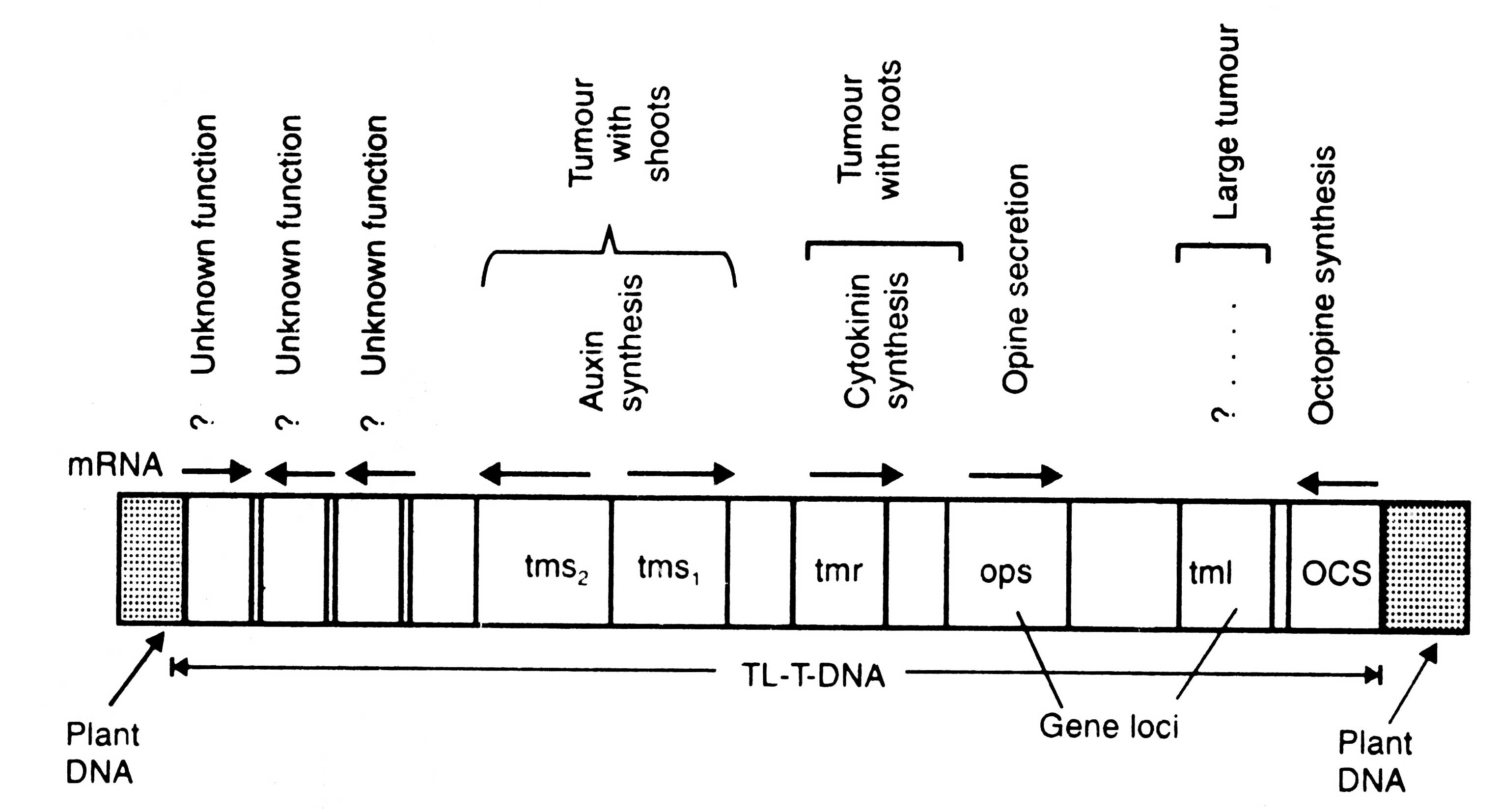
Fig. 4.12. Genetic map of octopine TL-DNA of Ti-plasmid controlling tumour morphology
(i) Mechanism of T-DNA transfer. A. tumifaciens lives in soil and attack many dicotyledonous plants (Fig. 4.9A) most probably at the level of soil surface. Formation of wound is necessary for establishment of bacterial plasmid into plant cells. Lipopolysaccharide, a compound secreted by bacterial cell wall, helps in their attachment with polygalacturonic acid fractions of plant cell wall. From the wounded cell walls of plants, a phenolic compound of low molecular weight (acetosyringone) is secreted which induce the vir gene of Ti-plasmids. Vir genes encode an enzyme which nicks the double stranded T-DNA on the same strand at two points, and produces single stranded DNA molecules. A cut is made on right border of T-DNA and a single stranded T-DNA fragment of 5' - 3' direction is generated. It is carried into plant cells (B).
T-DNA of Ti-plasmid is stably integrated with plant DNA. The sequencing of nucleic acid at the junction between plant DNA and T-DNA has been done. T-region in both octopine and nopaline plasmids is flanked by a direct repeat of 25 base pairs. Ops gene encodes enzymes for the synthesis of opines in transformed cells which are required for proliferation of infecting bacteria. TL-T-DNA encodes two enzymes that are involved in biosynthesis of two phytohormones, auxin and cytokinin (C) (Ream, 1989). This results in disorganized proliferation of cells commonly known as callus, gall or tumour (D). Therefore, the galls are colonized by the bacteria.
(ii) Tumour morphology. Several loci for controlling the morphology of tumour have been identified on TL-DNA of two octopine plasmids. These loci are grouped into three categories: tml (which causes larger tumour), tmr (which induce tumour with large number of roots) and tms (which causes tumour with large number of shoots) (Fig. 4.12). These effects are due to the products of loci of TL-DNA on the cytokinin/auxin ratios within the transformed tissues. The function of tml is not known so far.
(iii) Gene transfer in dicots by using T-DNA as vector. It is not possible to transfer Ti-plasmid into plant cell because of its large size, most probably upto 235 Kb. Therefore, T-DNA is excised and used as foreign DNA for expression in plant cells. From bacteriological point of view opine production and development of gall are not required for the successful integration and expression of DNA. Hence, replacement of nos and ops genes with a foreign DNA fragment retaining the opine synthase promoter for expression of inserted DNA fragment has no harm. Schell and Van Montague (1983) undertook some experiments where genes for resistance to kanamycin and methatrexate expressed in cultured callus cells. Generally it is easy to use opine promoters for the expression of foreign DNA. It has become possible that the inserted genes,
Tobean plant. Phaseolin gene isolated from french-bean was inserted into a Ti-plasmid which was again incorporated into A. tumifaciens cells. The transformed bacterial cell was allowed to infect a tobacco plant which developed crown gall. The cells from crown gall were cultured in vitro which developed into a fledgling plant. This plant was grafted onto stem of a developing tobacco plant. The mature grafted plant expressed its own genes and that transferred from the french bean. This new plant was, therefore, named as 'tobean'.
(iv) Gene transfer in monocots. The limitation of Ti-plasmid is that it is specific only to the dicot plants. A. tumifaciens does not induce tumour in monocot plants, however, most of the major crops including cereals are monocots. Hooykaas Van et al. (1984) discovered that A. tumifaciens could transfer T-DNA into certain monocots with the consequences of expression of opine gene within the plant cell without inducing the tumour. This discovery made it possible that T-DNA can be expressed into cells of monocot plants.
Agrobacterium rhizogenes causes formation of adventitious roots. This rhizogenicity has been correlated with the presence of a large plasmid, the Ri-plasmid, for root induction parallel to Ti-plasmid. A. rhizogenes does not induce enzymes for the biosynthesis of auxin or cytokinin. Plasmid for this species also contains a T-DNA which causes the development of hairy roots. It has also been reported to confer drought tolerance on apple seedlings (Moore et al, 1979).
The possible advantage of using Ri-plasmid of A. rhizogenes is the production of secondary root systems for (i) better anchorage of plants, (ii) ability to resist anoxia from flooding of the soil, (iii) increased drought resistance due to high root density, and (iv) greater chance of interaction with soil-borne mycorrhizal fungi (Smith and Walker, 1981). It may be applied to establish the secondary root systems in certain plants (fruit trees) by inoculation of soil as well as beneficial for those plants which are propagated through cutting and rooting as stimulants for root formation on cuttings.
Agrobacterium plasmids have been used for introduction of genes of desirable traits into plants. The properties of Ti- and Ri based plasmids are as below:
- They do not cause disease because of being disarmed by removing disease causing gene.
- They possess sites for insertion of foreign gene which needs to be introduced.
- They possess selectable markers i.e., genes which help in selecting the transformed cells.
The selectable marker is a powerful promoter e.g. 35S (for high level of expression of marker) derived from cauliflower mosaic virus (CaMV) and polyadenylation signal (for adding |poly| A to mRNA). The most commonly used selectable marker is neomycin phosphotrasferase (nptII) that confers resistance to neomycin and allows the cells to grow in medium containing neomycin. Thus, after inserting a foreign gene, a vector is designed. The vector is transferred to A. tumifaciens or A. rhizogenes depending on cases. The transformed bacterium is used to infect the designed host cell where the transformation of gene is to be brought about.
The explants are incubated or co-cultured with Agrobacterium containing vector with modified foreign gene. The transformed cell or colonies are selected du^ to the selectable marker and used for regeneration of whole plant which are expressed to be transgenic in nature. These regenerated plants can again be tested for the transfer of gene with the help of several scoreable or screenable markers called 'reporter genes' which is also transferred along with desired genes. The most commonly used reporter genes are nptII, cat, gus and lux (for detail see Transgenic plants).
Plant cell transformation
Plant cell suspension culture coupled with DNA transfer techniques has become a new field of gene manipulation in plants. Cell suspension cultures have been the traditional source of plant material for biochemical selection of plant cell mutants, such as numerous cell lines resistant to various amino analogs (Maliga, 1980). Suspension of protoplasts constitutes an identical plant material from one laboratory to other. The approaches made for plant protoplast transformation are :
- Introduction of DNA sequence via integration into T-DNA of Ti-plasmid of A. tumifaciens,
- Transfer of naked DNA into protoplast including bacterial spheroplasts and microinjection, and
- Transformation with plant viruses carrying inserts e.g. gemini and caulimoviruses.
Once DNA fragment is introduced into plant cells, it integrates with plant chromosomes and expresses from its signal. Transformation of plant cells with a gene for drug and antibiotic resistance linked to plant promoter has been possible. The gene could show expression in plant cell. Therefore, the transformed cells could be selected from the culture medium amended with drugs and antibiotics. The plants regenerated from cells show drug/antibiotics resistance.
Plant cell transformation by ultrasonication
Ultrasonication is done for various biological experiment. But in some plants this technique has been done for successful gene delivery. The Biotechnology Research Center, Beijing (China) has used this technique for gene transfer in plants like wheat, tobacco and sugarbeet. When the cultured explants were sonicated with plasmid DNA carrying market genes such as cat, nptll, and gus, and sonicated calli transferred to selective medium, shoots were grown successfully. The calli in control set of experiment which were not sonicated with plasmid did not grow on selective medium but they died. Transgenic tobacco plants were obtained at a frequency of 22 percent.
Liposomes are microscopically small sized particles. They contain a phospholipid bilayers which are concentric in nature. Liposomes enclose aqeuous chamber and can entrap water soluble molecules. Therefore, they are called lipid bags. Many plasmids are enclosed in them. By using polyethylene glycol (PEG) they may be stimulated to fuse with protoplast. In several plants like carrot, tobacco, petunia, etc. This technique has been used for successful transfer of genes. Due to endocyclosis of liposomes, DNA enters the protoplast. It gets adhered first to protoplast surface and get fused with protoplast at the site of surface. Consequently plasmids are released inside the cell. There are several advantages in using this technique such as (i) low toxicity, (ii) protection of DNA/RNA from nucleases that lyse them, (iii) long stable storage of DNA fragments in liposome, (iv) applicability in various cell types, and (v) high level of reproductivity.




