Secondary Metabolites
A plenty of literature is available on toxin production by a variety of micro-organisms. Roux and Yersin (1888) who first observed the presence of toxin in a disease caused by Corynebacterium diphtheriae. Gaumann (1954) stated "micro-organisms are| pathogenic only if they are toxigenic". He used the term "toxin" and "microbial poison" to denote all substances produced by the pathogens. Therefore, there is a correlation between toxin and development of disease, resulting in the death of hosts. Toxins interrupt the metabolic processes of the living host cells.
Toxins secreted by micro-organism lead to the development of infection in plants/animals, as the affected cells lose their resistance.
Toxins secreted by bacteria also play a key role in disease development in animals. In recent years use of toxin secreting micro-organisms in control of disease causing flies, has become a field of major interest in formulation of biological pesticides. However, screening programme for discovery of new secondary metabolite from Streptomyces having insecticidal activity has been carried out in Japan.
Chemical nature of the toxin
It is mentioned that generally toxins are proteinaceous in nature; they interrupt the metabolism of host cells. It differs from enzyme in action as the latter destroys structural integrity of the cell.
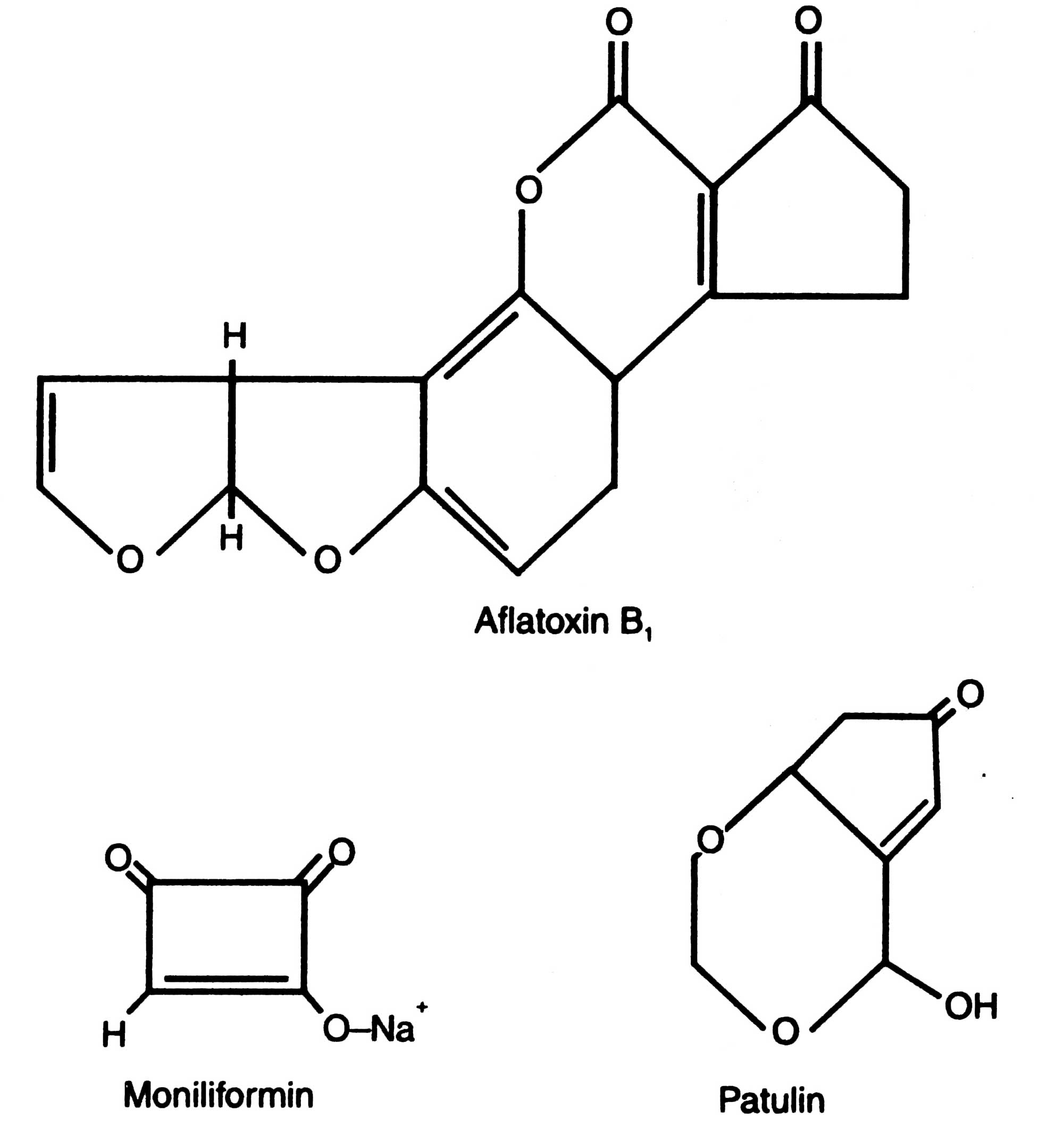
It is interesting to note that crystals of toxin secreted by B. thuringiensis are water soluble, and heat stable. This toxin has been termed as 6-exotoxin which is of molecular weight of 900. Fig. 13.5 shows that b-exotoxin is an adenine nucleotide with an unusual sugar derivative called allomucic acid.
Production of b-exotoxin on a large scale is easy. Pure culture of B. thuringiensis is grown on a nutrient medium (in submerged culture) supplemented with starch because this bacterium secretes amylolytic enzymes. When bacterial growth is over the culture fluid is filtered. The final product is obtained from the filtrate by vacuum drying.
Microbial insecticides
b-exotoxin is active against flies and mosquitos (diptera), beetles (coleoptera), termites (orthoptera), butterflies and moths (lepidoptera). Mammals including man are not affected because of secretion of acid in stomach and most probably degradation by pepsin to non-toxic form. Similarly, an anaerobic spore forming rod shaped bacterium, B. popilliae feeds upon the larvae of the Japanese bettle, Popillia japonica. A detailed account of microbial insecticides is given in Bacterial, viral and fungal pesticides.
Undoubtedly, fungi play a significant role in production of many products such as enzymes, organic acids, antibiotics, SCP, cheese, etc. However, the biotechnological hazards caused by them have also been realized in recent years. Mycotoxin refers to secondary metabolites of certain fungi which are toxic to human animals and plant health as well. Chemically, mycotoxin are non-antigenic compounds of low molecular weight. These toxins are generally detected in milk, cheese, corn, peanuts, cotton seeds, copra, almond, figs, spices and other foods and feeds. Depending on concentrations, these may be carcinogenic, mutagenic, teratogenic or oestrogenic with the result of reduced immune response and acute disease syndrome.
Mycotoxicosis is the poisoning by ingestion of mycotoxins through food contaminated by toxigenic fungi. Historically, mycotoxicosis has been known for hundreds of years ( 9th and 18th century) due to death of thousands of people in Europe caused by food-borne and a toxins (ergot) producing fungus (Qaviceps pupurea) in rye grains. In 1978, Ethiopian epidemic occurred due to consumption of grains contaminated with C. purpurea infected wild oats where about 50% infected people died and the remaining suffered from many side effects.
Though there is a large number of mycotoxins yet some of them which contaminate different food materials are aflatoxins, ochratoxins, zearalenone, citrinin, sterigmatocystin, trichothecenes, patulin, penicillic acid, etc. (Table 16.1; Fig. 16.1). In 1988, a new group of water soluble toxin, fumonisins, produced by certain stains of Fusarium moniliforme was discovered by Gelderblom (1988). These were associated with contaminated corn and commercial corn based food stuff. If contaminated corns are used in fermentation for ethanol production, a little degradation of toxin occurs. However, most of the fumonisin B1 could be recorded through distillation in spent grains, and stillage (Bothast et al, 1992). When spent grains are used as animal feed without detoxification, it causes serious problems. Similarly, patulin was found in detectable levels in wines, beers and fruit juices.
The toxigenic fungi
Mostly all the fungi are equipped with toxin producing ability depending on environmental conditions. However, a few of them associated with food and feed materials pose a great hazard, for example, species of Alternaria, Aspergillus, Claviceps, Fusarium, Penicillium, Boletus, Agaricus, Amantia, Myrothecium, Pithomyces, etc.
The biochemical pathways through which mycotoxins are produced are : (i) the polyketide pathway (aflatoxins), (ii) the terpene pathway (trichothecenes), (iii) the tricarboxylic acid pathway (rubratoxin), and (iv) the amino acid pathway (gliotoxin) (Turner, 1971). Discussion of these metabolic pathways in detail is not the aim of the author; therefore, these are not dealt with.
Mode of action involves the biochemical reactions of mycotoxins with molecular receptors in animal cells. The molecular receptors are DNA, RNA, functional proteins, enzyme co-factor and membrane constituents. The reactions between mycotoxins and their receptors may be either co-valent irreversible or non-covalent reversible. In the first reaction the reactive forms of mycotoxins conjugate with receptors to form adducts, whereas in the second reaction mycotoxin - receptor complexes get dissociated as the metabolic processes remove the toxin from the receptor sites (Hsieh, 1987).
After ingestion mycotoxins enter in human body and encounter various molecules. Toxins interact with gastrointestinal microflora, epithelial cells of intestine, liver, bile, blood, kidney, reproductive and nervous systems, skin and lungs. Some of the effects of mycotoxins are briefly described below:
(ii) Inhibition in synthesis of DNA, RNA, proteins and immune systems: Inhibition in the synthesis of these macromolecules lead to cell death. Aflatoxin B1 inhibits DNA synthesis in liver cells. This is caused due to covalent binding of aflatoxin B1 to DNA and proteins. Aflatoxin B1 is also known as to inhibit the synthesis of nuclear RNA in liver cells of rat. Since the mycotoxins inhibit DNA synthesis, the other products expressed by genes are also inhibited. Aflatoxin B{ causes delay in interferon production in turkeys. However, at high doses, it reduces IgG and IgA in chicks with the consequences of decreased acquired immunity. Aflatoxin B{ reduces the cell mediated immune response in animals. A reduced responses to phytohaemagglutinin stimulation in human lymphocytes has been observed in vitro (Savel et al, 1970).
Table 16.1. Natural occurrence of mycotoxins on different commodities, and mycotoxicoses.
| Mycotoxins/name | Mycotoxicoses | Commodities | |
| A. | ASPERGILLUS TOXINS | ||
| 1. | Aflatoxin B1, B2, G2, G2 | Liver cancer | Corn, peanuts, milk, treenuts |
| 2. | Sterigmatocystin | Carcinogenesis*, | Green coffee, grains, fruits |
| 3. | Ochratoxin | Renal tumour | Corn, coffee, wheatflour, bread |
| B. | FUSARIUM TOXINS | ||
| 1. | Moniliformin | Onylai diseases | Rice |
| 2. | Fumonisins | Leukoencephalo-malacia *, promote cancer * | Corn, wheat flour, corn flakes |
| 3. | Trichothecenes (T-2 toxin, Deoxyninalenol) | Dermatites, oesophagal cancer, digestive disorder | Corn, wheat, baby cereal, wine, commercial cattle feed |
| 4. | Zearelenone | Cervical cancer, abortion* | Corn meal, corn flakes, walnut |
| C. | PENICILLIUM TOXINS | ||
| 1. | Citreoviridin | Cardiac beriberi | Mouldy pecan fragments |
| 2. | Citrinin | Kidney damage* | Dry fruits, rice, corn |
| 3. | Cyclopiazonic acid | Kodua poisoning | Cheese crust, corn |
| 4. | Patulin | Capillary damage in vital organs | Cider, apple juice, wine, jam, grape juice, scented supari |
| 5. | Peaictllic acid | Edema* carcinogenesis* | Corn, bean, apple |
| 6. | Penitrem A | Bloody diarrhoea* death* | Mouldy cream cheese |
| 7. | Rubratoxin B | Liver disease * | Mouldy grains |
| D. | OTHER MYCOTOXINS | ||
| 1. | Agaricus toxins | Kwashiorkor | Agaricus bisporus, frozen, mushrooms |
| (i) | Agaritin, amotoxin | Mushroom poisoning | Mushrooms |
| (ii) | Amanitins | ||
| 2. | Ergot alkaloid | ||
| (i) | Ergosine, ergometrin, ergocristine | Ergotism | Flour of rye, wheat, triticale, wheat cereals, baby cereals. |
(iii) Effects on nervous systems. Mycotoxins are grouped into three on the basis of mode of action:
(a) Mycotoxins causing paralysis and inhibition in respiratory system e.g. citrioviridia. They kill nerve cells disrupting energy supply as they inhibit ATPase activity.
(b) Mycotoxins inducing trembling in animals e.g. fumitremorgin A, penitrem A. They alter functional states of neurotransmitters and disrupt nervous system.
(c) Mycotoxin causing vomiting in animals e.g. vomitoxins such as trichotheccnes. They act on chemoreceptor trigger zone in medula oblongata and change the biogenic amines.
(v) Carcinogenic effects. Aflatoxins, B1 G1, and M1 and sterigmatocystin, versicolorin A, luteoskyrin are known as carcinogenic mycotoxins. These are genotoxic also. The chemicals which cause gentle damage and initiate the carcinogenic process are known as genotoxic or initiators, whereas those which promote transformation of genetically modified cells to cancerous cells are known as promoters. Most of the chemicals are both genotoxic and promoters.
(vi) Effects on reproductive systems. Urogenital systems of swine cattle and poultry animals are known to be affected by zearalenone which at 1 ppm produces hyperastrogenism in pigs. In young male swine, it produces testicular atrophy, and mammary gland enlargement. It has also been observed that at high concentrations zearalenone is associated with infertility, teat enlargement and under secretion in cattle. Zearalenone causes embryonic death and inhibition of development in swines.
Ergot ingestion may causes abortion in animals. Moreover, ergot is also associated with reduced weight gain and milk production in animals.
Although total elimination of mycotoxin from human food and animal feed is far difficult, yet the hazards from high concentrations can be minimized in some cases by adopting certain precautionary steps.
(i) Residues of susceptible crops serves as a substrate for inoculum production of certain toxigenic fungi such as Claviceps, Aspergillus flavus, etc. Therefore, management of crop residues and use of resistant varieties should be done.
(ii) Crop rotation should be adopted to lower the primary inoculum of toxigenic fungi.
(iii) Since grain and oilseed crops are susceptible to fungal deterioration, these must be harvested when the crops have reached to their optimum maturity. At this time presence of low moisture content in grains will minimize the risk of field contamination. As most of the contaminants get associated from the field.
(v) Electronic or hand sorting of contaminated peanuts and other seeds, wherever possible, should be done.
(vi) Biological method of detoxification should be adopted by using certain strains of yeasts, molds or bacteria e.g. Flavobacterium aurantiacum (NRRL B-184) from the liquid medium. These catalyze the hydration of aflatoxins (Marth and Doyle, 1979).
(vii) Chemical detoxification is also one of the methods for inactivating the aflatoxins. Ammoniation (use of ammonia gas) resulted in significant reduction in levels of aflatoxins in contaminated peanuts, cotton seed meals, corn, etc. (Council for Agricultural Science and Technology, Report No. 116, 1989).