Important Information
- This laboratory manual is for reference and use for those using the environmental science/engineering laboratory.
- Discussion after each experiment should be based on the following points:
- Limit prescribed for that constituent in drinking water standards.
- The suitability of the sample for drinking purpose with respect to that particular constituent.
- Users may refer the following for writing the discussion after each experiment:
- “Standard Methods for the Examination of Water and Waste Water”, American Public Health Association, 1015, 15th Street, N.W., Washington D.C., 2005.
- “Chemistry for Environmental Engineers”, Sawyer and McCarty, Tata Mc-Graw Hill.
- “Manual of Standards of Quality for Drinking Water Supplies”, Indian Council of Medical Research, New Delhi.
- “International Standards for Drinking Water” — World Health Organisation.
- “IS 2490 - 1981, IS 3306 - 1974, IS 3307 - 1977, IS 7968 - 1976, IS 2296 - 1974”, Bureau of Indian Standards, New Delhi.
- Do thoroughly clean the glassware before and after use.
- Do handle the glassware carefully.
- Do not handle chemicals with bare hands.
- Do not blow out the last drop from the pipette. When the liquid has drained out completely, touch the tip of the pipette to the inner surface of the vessel.
- Do not add water to acids. Do always add acid to water.
- Do use large volumes of water, when a person is splashed with acid to prevent serious burns.
- Do weigh the articles in a balance only at room temperature.
- Do use different pipette for different reagents.
- Do not pipette out acids and other toxic reagents by mouth.
- Do read the level of the curve (meniscus), in all volumetric glassware, with the eye at approximately the same level as the curve of solution.
In water and wastewater analysis, the results are usually reported in terms of mg/L of some particular ion, element or compound. It is most convenient to have the standard titrating agent of such strength, that 1mL is equivalent to 1mg of material being measured. Thus 1 litre of the standard solution is usually equivalent to 1g of the standard substance.
Normality
The desired normality of the titrant is obtained by the relationship of 1 to the equivalent weight of the measured material. Thus normality of acid solution to measure ammonia, ammonia nitrogen, and alkalinity as CaCO3
Ammonia N — 1/eq. wt. = 1/14 = N/14 = 0.0715N
Alkalinity — 1/eq. wt. = 1/50 = N/50 = 0.020N
The normality of basic solution to measure mineral acidity as CaCO3 is:
Acidity — 1/eq. wt. = 1/50 = N/50 = 0.020N
The normality of silver nitrate to measure chloride and sodium chloride is:
Chloride — 1/eq. wt. = 1/35.45 = N/35.45 = 0.0282N
Sodium chloride — 1/eq. wt. = 1/58.44 = N/58.44 = 0.071N
Thus the substance measured is calculated as follows:
= mL of titrant used x 1,000 ÷ mL of samplemg/L
Most materials subjected to the analysis of water and wastewater fall in the realm of dilute solutions i.e., a few mg in a litre. So the results are normally expressed in mg/L or ppm. Parts per million (ppm) is a weight ratio; but mg/L is a weight by volume ratio. The relationship is given as follows:
ppm = mg / L ÷ Sp.gr.
If concentrations are less than 0.1 mg /L, express them in µg/L (micrograms per litre). If concentrations are more than 10,000 mg/L, they are expressed in percentages.
Rules listed by Worthing and Geffner are to be followed while plotting graphs. They are:
- The independent and dependent variables should be plotted on abscissa and ordinate respectively.
- The scale should be so chosen that the value of either coordinate could be found quickly and easily.
- The curve should cover as much of the graph sheet as possible.
- The scales should be so chosen that the slope of the curve approach unity as nearly as possible.
- The variables should be chosen to give a plot that will be as nearly a straight line as possible.
Classification of Procedures
Laboratory analytical procedures are classified to quantify the chemical substances as follows:
- Toxic chemical substances: e.g., lead, arsenic, selenium, hexavalent chromium, cyanide.
- Chemical substances affecting health: e.g., fluoride, nitrate.
- Chemical substances affecting potability: e.g., residue, turbidity, colour, taste and odour, iron, manganese, copper, zinc, calcium, magnesium, sulphate, chloride, pH and phenolic compounds.
- Chemical substances indicative of pollution: e.g., total organic matter, BOD, Kjeldahl nitrogen (total organic nitrogen), albuminoid nitrogen, nitrite nitrogen and phosphate.
- Residual chlorine.
Standards of water quality are presented as follows:
Bacteriological Quality
- Treated water: In 90% of the samples examined throughout the year, the coliform bacteria shall not be detected or the MPN index shall be less than 10. None of the samples shall have an MPN index of coliform bacteria in excess of 10. An MPN index of 8-10 shall not occur in consecutive samples.
- Untreated water: In 90% of the samples examined throughout the year, the MPN index of coliform organisms should not be less than 10. None of the samples should show an MPN index greater than 20. An MPN index of 15 or more should not be permitted in consecutive samples.
Chemical and Physical Quality
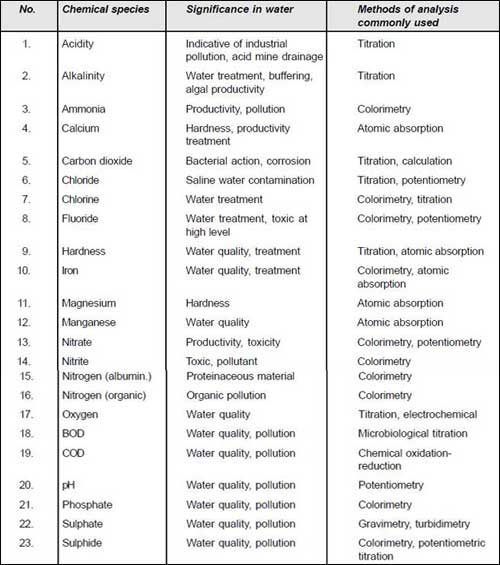
Significance and Determination of Chemical ParametersChemical parameters and their significance are presented as follows. The methods of the analysis adopted are also presented. However, only simple methods will be dealt within this manual.
IV. Introduction
A characteristic, which has set Homo sapiens apart from other species, has been their ability to control many aspects of their environment. Throughout recorded history people have continually struggled to manage their natural environment in order to improve their health and well-being. In recent years environmental sanitation in many parts of the world has led to large reduction or virtual elimination of diseases spread via the environment. Continuous environmental vigilance is necessary to keep away these weeds from the garden of humanity from increasing out of proportion among a large part of the earth's population.
The public has been more exacting in their demands as time has passed, and today water engineers are expected to produce finished waters that are free of colour, turbidity, taste, odour and harmful metal ions. In addition, the public desires water, which is low in hardness and total solids, non-corrosive, and non-scale forming. To meet with such stringent standards, chemists, biologists and engineers must combine their efforts and talents together and hence the need for analytical testing of water and waste becomes necessary.
Quantitative analysis serves as the keystone of engineering practice. Environmental engineering is perhaps most demanding in this respect, for it requires the use of not only the conventional measuring devices employed by engineers but, in addition many of the techniques and methods of measurement used by chemists, physicists and some of those used by biologists.
Every problem in environmental engineering must be approached initially in a manner that will define the problem. This approach necessitates the use of analytical methods and procedures in the field and laboratory, which have proved to yield reliable results. Once the problem has been defined quantitatively, the engineer is usually in a position to design facilities that will provide a satisfactory solution.
Most problems in environmental engineering practice involve relationships between living organisms and their environment. Because of this, the analytical procedures needed to obtain quantitative information are in often a strange mixture of chemical and biochemical methods, and interpretation of the data is usually related to the effect on microorganisms or human beings. Also, many of the determination used fall into the realm of microanalysis because of the small amounts of contaminants present in the samples. Ordinarily, the amounts determined are a few milligrams per litre and often they are found only in few micrograms.
Standard Methods of Analysis
Concurrent with the evaluation of environmental engineering practice, analytical methods have been developed to obtain the factual information required for the resolution and solution of problems. In many cases different methods have been proposed for the same determination, and many of them were modified in some manner. As a result, analytical data obtained by analysis were often in disagreement.
"Standard Methods" as published today is the product of the untiring effort of hundreds of individuals who serve on committees, testing and improving analytical procedures for the purpose of selecting those best suited for inclusion in "Standard Methods", which is now available as "Standard methods for the examination of water and waste water".