Filamentous fungi
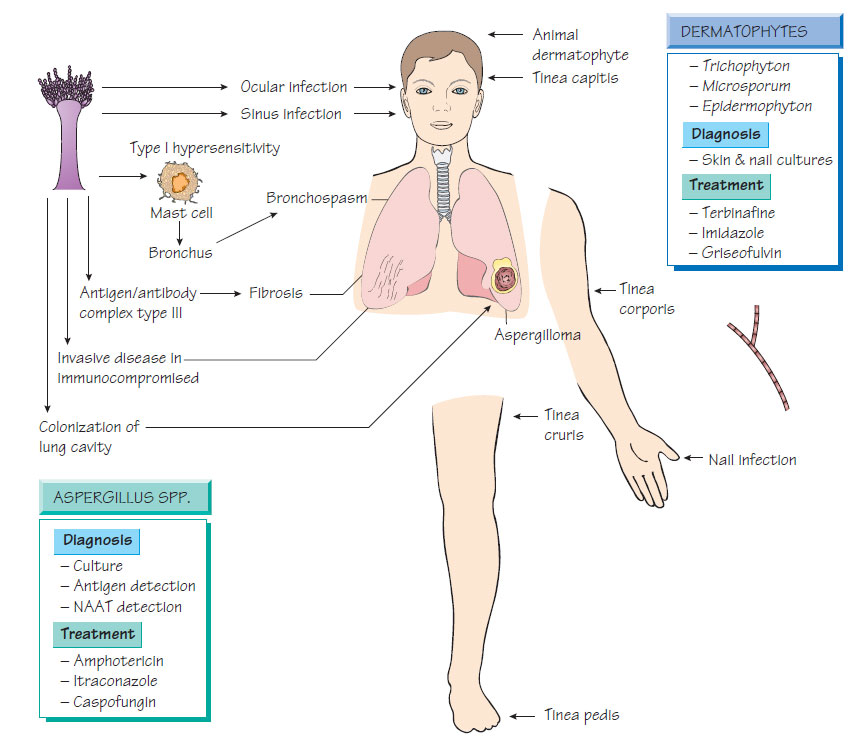
Aspergillus spp.Aspergillus spp. are ubiquitous, free-living, saprophytic organisms; A. fumigatus, A. niger, A. flavus and A. terreus are associated with human infection.
Pathology and Clinical features
Inhalation of Aspergillus spores can provoke a type III hypersensitivity reaction with fever, dyspnoea and progressive lung fibrosis (farmer's lung). Colonization by Aspergillus can provoke a type I hypersensitivity reaction that results in intermittent airway obstruction (bronchopulmonary allergic aspergillosis). Healed tuberculosis cavities or bronchiectasis can become colonized with Aspergillus, creating a 'fungus ball' or aspergilloma. In neutropenic patients, Aspergillus infection typically begins in the lungs and may be followed by fatal disseminated disease. Rarely the paranasal sinuses, skin, central nervous system and eye may become infected, often with a poor prognosis.
Sputum culture is of limited value. An isolate from bronchoalveolar lavage is diagnostic (98% specificity) but lacks sensitivity. Antibody detection may confirm bronchopulmonary aspergillosis and farmer's lung, but immunocompromised patients rarely produce an antibody response. Enzyme immunoassay (EIA) to detect galactomannan in serial samples is useful. Nucleic acid amplification tests (NAATs) are a useful adjunct to diagnosis.
Treatment and Prevention
Bronchopulmonary aspergillosis requires treatment of the airway obstruction with bronchodilators and steroids. Invasive aspergillosis requires treatment with amphotericin B. Itraconazole has activity against Aspergillus; voriconazole improves outcome in pulmonary aspergillosis. Surgery may be beneficial in some cases of pulmonary infection. Patients with farmer's lung should avoid further exposure. Neutropenic patients should be managed in rooms with filtered air and infection should be aggressively treated (see Intestinal protozoa).
Mucor, Rhizopus and Absidia may infect severely immunocompromised patients. Sinus infection may spread to the eyes and brain. Pulmonary disease may be complicated by dissemination. These infections are difficult to treat and have a poor prognosis.
Dermatophytes
Three genera of filamentous fungi are implicated in dermatophytosis: Epidermophyton, Microsporum and Trichophyton. Dermatophytes are also grouped according to their reservoir and host preference: anthrophilic (mainly human pathogens); zoophilic (mainly infect animals); and geophilic (found in soil and able to infect animals or humans). Anthrophilic species spread by close contact (e.g. within families, enclosed communities). Transmission of geophilic species is rare. Close contact with animals may give rise to zoophilic infection (e.g. pet owners, farmers and vets).
Dermatophyte infection in the form of ringworm presents as itchy, red, scaly, patch-like lesions that spread outwards leaving a pale, healed centre; chronic nail infection produces discoloration and thickening; scalp infection is often associated with hair loss and scarring. Clinical diagnostic labels are based on the site of infection (e.g. tinea capitis, head and scalp; tinea corporis, trunk lesions). Lesions are rarely painful, but zoophilic species produce an intense inflammatory reaction with pustular lesions or an inflamed swelling (kerion).
Laboratory Diagnosis
Infection of skin and hair by some species may demonstrate a characteristic fluorescence when examined under Wood's light. Skin scrapings, nail clippings and hair samples should be sent dry to the laboratory. Typical branching hyphal elements may be demonstrated by microscopy in a potassium hydroxide preparation. Dermatophytes take up to 4 weeks at 30°C to grow on Sabouraud's dextrose agar. Identification is based on colonial morphology, microscopic appearance (lactophenol blue mount), biochemical tests and sequencing of the 18S rRNA gene.
Dermatophyte infections may be treated topically with imidazoles (e.g. miconazole, clotrimazole, tioconazole or amorolfine). Some infections require oral terbinafine for several weeks.
Antifungal compounds
Terbinafine
Terbinafine inhibits squalene epoxidase with resultant accumulation of aberrant and toxic sterols in the cell wall. It is indicated for the oral treatment of superficial dermatophyte infections that have failed to respond to local therapy. Reported adverse effects include Stevens-Johnson syndrome, toxic epidermal necrolysis and hepatic toxicity. Treatment should be continued for up to 6 weeks for skin infections and 3 months or longer for nail infections.
Griseofulvin
Griseofulvin is active only against dermatophytes by inhibition of fungal mitosis. Given orally, it is incorporated into the stratum corneum or nail where it inhibits fungal invasion of new skin and nail. Treatment must be continued until uninfected tissue grows. It is now rarely used.
There are two polyene cyclic macrolides in clinical use, nystatin and amphotericin B, which are active against almost all fungi. Polyenes bind ergosterol in the fungal membrane forming a pore that leads to leakage of the intracellular contents and cell death. Resistance is rare.
Nystatin is used for topical treatment and the prevention of fungal infection in immunocompromised patients. It has no value for the treatment of dermatophyte infections. Amphotericin is given parenterally; liposomal formulations overcome much of the toxicity of earlier compounds enabling higher doses to be given safely.
Echinocandins
The echinocandins act by inhibiting the synthesis of 1,3β-glucan, a homopolysaccharide in the cell wall of many pathogenic fungi. They are active against both Candida and Aspergillus. New agents in this class, which are now entering clinical use (e.g. caspofungin), are well tolerated and offer a useful alternative for refractory infections.