Aluminum Absorption and Transport within Plants
Phytotoxic Species
The most phytotoxic form of aluminum is Al3+ (more correctly, Al(H2O)63+), which predominates in solutions below pH 4.5 (31-33) (Figure 16.1). Possibly, hydroxyl-aluminum (AlOH2+ and Al(OH)2+) ions are also phytotoxic, particularly to dicotyledonous plants (31,34). However, as pointed out by many researchers (35,36), these aluminum species are interrelated along with the pH variable, so it is difficult to rank their relative toxicity.In contrast, Al-F, Al-SO4, and Al-P species are much less toxic or even nontoxic to plants (34,37). Barley (Hordeum vulgare L.) roots were unaffected by aluminum when 2.5 to 10 µM F- was added to nutrient solution containing up to 8 µM total soluble aluminum (37). Also using nutrient solution, Kinraide and Parker (38) positively demonstrated the nontoxic nature of Al-SO4 complexes (AlSO4+ and Al(SO4)2-) for wheat (Triticum aestivum L.) and red clover (Trifolium pratense L.). Soybean had longer root growth when increasing amounts of phosphorus were added to nutrient solutions having constant total aluminum concentrations (39).
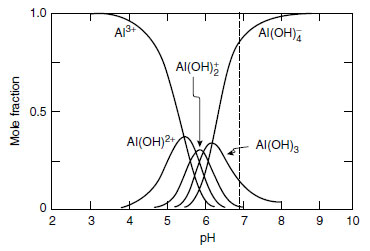 |
FIGURE 16.1 Speciation of aluminum as affected by solution pH. (From R.B. Martin. Fe3+ and Al3+ hydrolysis
equilibria. Cooperativity in Al3+ hydrolysis reactions. J. Inorg. Biochem. 44:141-147, 1991.) |
Absorption
Since aluminum is a trivalent cation in its phytotoxic form in the external medium, it does not easily cross the plasma membrane. Akeson and Munns (40) calculated that the endocytosis of Al3+ could contribute to its absorption. Alternatively, it is possible that Al3+ could be absorbed through calcium channels (41) or nonspecific cation channels.Our understanding of aluminum absorption across plant membranes has been limited by the complex speciation of Al, its binding to cell walls, lack of an affordable and available isotope, and lack of sensitive analytical techniques to measure low levels of aluminum in subcellular compartments (42). Aluminum absorption by excised roots of wheat, cabbage (Brassica oleracea L.), lettuce (Lactuca sativa L.), and kikuyu grass (Pennisetum clandestinum Hochst. ex Chiov.), and by cell suspensions of snapbean (Phaseolus vulgaris L.) followed biphasic kinetics (43-45). A rapid, nonlinear, nonmetabolic phase of uptake occurred during the first 20 to 30 min. This nonsaturable phase was thought to be accumulation in the apoplastic compartment due to polymerization or precipitation of aluminum or binding to exchange sites in cell walls (44). A linear, metabolic phase of uptake was superimposed over the nonlinear phase and thought to be accumulation in the symplasmic compartment (i.e., within the plasma membrane). Using the rare 26Al isotope and accelerator mass spectrometry on giant algal cells of Chara corallina Klein ex Willd., Taylor et al. (42) provided the first unequivocal evidence that aluminum rapidly crosses the plasma membrane into the symplasm. Accumulation of 26Al in the cell wall was nonsaturable during 3 h of aluminum exposure and accounted for most of aluminum uptake. Absorption of aluminum into the protoplasm occurred immediately but accounted for less than 0.05% of the total accumulation (42). Accumulation in the vacuole occurred after a 30-min lag period (42).




