Role in Cell Walls
Calcium is a key element in the structure of primary cell walls. In the primary cell wall, cellulose microfibrils are linked together by cross-linking glycans, usually xyloglucan (XG) polymers but also glucoarabinoxylans in Poaceae (Gramineae) and other monocots (9). These interlocked microfibrils are embedded in a matrix, in which pectin is the most abundant class of macromolecule. Pectin is also abundant in the middle lamellae between cells.Pectin consists of rhamnogalacturonan (RG) and homogalacturonan (HG) domains. The HG domains are a linear polymer of (1→4)-α'-linked D-galacturonic acid, 100 to 200 residues long, and are deposited in the cell wall with 70 to 80% of the galacturonic acid residues methyl-esterified at the C6 position (9). The methyl-ester groups are removed by pectin methylesterases, allowing calcium ions to bind to the negative charges thus exposed and to form inter-polymer bridges that hold the backbones together (9). The whole structure can be thought of as resembling an eggbox (Figure 5.1).
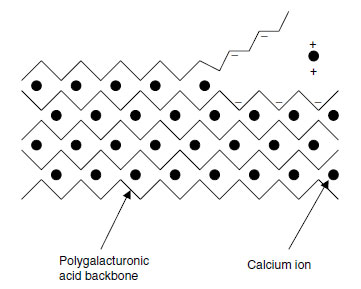 |
| FIGURE 5.1 The 'eggbox' model of calcium distribution in pectin. (Based on E.R. Morris et al., J. Mol. Biol. 155: 507-516, 1982.) |
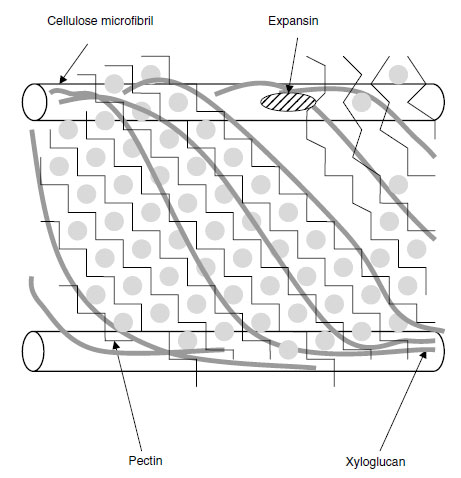 |
| FIGURE 5.2 Diagrammatic representation of the primary cell wall of dicotyledonous plants. (Based on E.R. Morris et al., J. Mol. Biol. 155:507-516, 1982; F.P.C. Blamey, Soil Sci. Plant Nutr. 49:775-783, 2003; N.C. Carpita and D.M. Gibeaut, Plant J. 3:1-30, 1993.) To the right of the figure, Ca2+ ions have been displaced from the HG domains by H+ ions, so that the pectin is no longer such an adhesive gel and slippage of the bonds between adjacent XG chains occurs and expansin is able to work on them. This loosens the structure and allows the cellulose microfibrils to be pushed further apart by cell turgor. |
Pectin is a highly hydrated gel containing pores; the smaller the size of these pores, the higher the Ca2+ concentration in the matrix and more cross-linking of chains occurs (11). This gel holds the XG molecules in position relative to each other, and these molecules in turn hold the cellulose microfibrils together (Figure 5.2). The presence of the calcium, therefore, gives some load-bearing strength to the cell wall (13). It is suggested that when a primary cell wall is expanding, localized accumulation of H+ ions may displace Ca2+ from the HG domains, thereby lowering the extent to which the pectin holds the XG strands together (11). In a root-tip cell, where the cellulose microfibrils are oriented transversely, slippage of the XG chains allows the cellulose microfibrils to move further apart from each other, giving cell expansion in a longitudinal direction.
Cell-to-cell adhesion may also be given by Ca2+ cross-linking between HG domains in the cell walls of adjacent cells, but this action is less certain as experimental removal of Ca2+ leads to cell separation in a only few cases (9). In the ripening of fruits, a loosening of the cells could possibly occur with loss of calcium. It has been postulated that decrease in apoplastic pH in ripening pome fruits may cause the release of Ca2+ ions from the pectin, allowing for its solubilization (14). However, in an experiment on tomato (Lycopersicon esculentum Mill.), the decline in apoplastic pH that occurred was not matched by a noticeable decrease in apoplastic Ca2+ concentration, and the concentration of the ion remained high enough to limit the solubilization of the pectin (15). It certainly seems that calcium inhibits the degradation of the pectates in the cell wall by inhibiting the formation of polygalacturonases (16), so the element has roles in possibly holding the pectic components together and in inhibiting the enzymes of their degradation. In a study on a ripening and a nonripening cultivar of tomato (Rutgers and rin, respectively), there was an increase in calcium concentration after anthesis in the rin cultivar, whereas in the Rutgers cultivar there was a noticeable fall in the concentration of bound calcium and an increase in polygalacturonase activity (17). In a study on calcium deficiency in potato (Solanum tuberosum L.), deficient plants had more than double the activity of polygalacturonase compared with normal plants (18).




