Toxin
Toxins are all compounds that are either synthesized by the algae or formed by the composition of metabolic products and hence represent an intrinsic characteristic of the organism. Of the millions of species of microalgae those that produce specific toxins scarcely exceed a hundred. These occur in both salt and freshwaters, and while most are planktonics some are benthic or floating at water surface. Toxins can attract particular attention when they cause the death of livestock that has drunk water containing them or fish and shellfish in the sea, or humans that consume these. Toxins have been divided into different classes based on the syndromes associated with exposure to them, such as paralytic shellfish poisoning (PSP), diarrheic shellfish poisoning (DSP), neurotoxic shellfish poisoning (NSP), ciguatera fish poisoning (CFP), and amnesic shellfish poisoning (ASP).
Algae which seem to be directly producer of toxic substances mostly belong to three taxonomic groups: Cyanophyta, Haptophyta, and Dinophyta. In addition to these there are some groups which include one or two toxic members. Species of Chattonella and Heterosygma, belonging to the Raphidophyceae, form toxic red tides in Japanese waters and a few diatoms of the genus Peusdonitzschia produce domoic acid, a low molecular amino acid causing amnesic shellfish poisoning.
Among the 50 freshwater existing cyanobacteria genera, 12 are capable of producing toxins. While blue-green algae have significant taste and odor constituents, representing a moldy smell, their toxic metabolites have no taste, odor, or color. The risk of exposure to algal toxins may come from drinking water, recreational water, dietary supplements, or residue on produce irrigated with contaminated water and consumption of animal tissue. Avoiding cyanobacteria toxins is not as easy as avoiding a harmful algal bloom as toxins may be present in fish, shellfish and water even after the bloom has dissipated. Cyanobacterial toxins are responsible for a variety of health effects such as skin irritations, respiratory ailments, neurological effects, and carcinogenic effects.
The three major classes of these compounds are:
- Cyclic peptides (nodularins, microcystins). Nodularia, a well-known cyanobacterium, produce nodularins that are primarily a concern in marine and brackish waters thus creating a risk to recreational swimmers. The 65 variants of microcystins, however, are isolated from freshwaters worldwide and are produced by Microcystis (the most commonly identified cyanobacteria in human and animal poisonings), Anabaena, and other algae. They are very stable in the environment and resistant to heat, hydrolysis, and oxidation. Both toxins have an affinity for the liver. Other symptoms of exposure to microcystins may range from weakness, loss of appetite, vomiting, and diarrhea to cancer.
- Alkaloids (anatoxin, saxitoxin). Anatoxins may affect the nervous system, skin, liver, or gastrointestinal tract. These neurotoxins can cause symptoms of diarrhea, shortness of breath, convulsions and death, in high doses, due to respiratory failure. Saxitoxins are the cause of paralytic shellfish poisonings in humans consuming contaminated shellfish. There are no reports of similar poisonings via the drinking water route.
- Lipopolysaccharides (endotoxins). A similar cell wall toxin as found in Salmonella bacteria, but appears to be less toxic.
The physicochemical nature of the water source can have an effect on not only the growth but also the toxicity of the algal bloom. For example, some algae increase in toxicity when blooms are iron-deficient. In general, temperature, sunlight, and nutrient loads have a substantial impact on the proliferation of the bloom.
Haptophyta contain a few toxic species. Prymnesium parvum produces a potent toxin that causes extensive fish mortality in brackish water. Another flagellate Chrysochromulina polylepis produces and excretes glycolipids with hemolytic and ichtytoxic properties causing osmoregolatory failure similar to that brought about by P. parvum. Another widely distributed marine haptophyte Phaeocystis sp. is familiar to fishermen in the form of extensive blooms of mucilaginous colonies, which are avoided by herring. It produces large quantities of acrylic acid, which has strong bacteriocidal properties.
Already 70 years ago, P. pouchetii was suspected to cause avoidance of herring. Later it was demonstrated that copepods avoided gracing on healthy P. pouchetii colonies, food intake and growth were reduced in sea cage cultivated salmon during the spring bloom of P. pouchetii and water from P. pouchetii cultures acted toxic towards cod larvae.
Species belonging to the genus Phaeocystis are important in all oceans, and P. pouchetii is an important component of the spring bloom of phytoplankton in northern waters. Its life cycle is only partly resolved but is known to be polymorphic consisting of at least two solitary and one colonial stage.
P. pouchetii has been reported to produce a polyunsaturated aldehyde (PUA) as diatoms, namely the 2-trans-4-trans-decadienal. This compound is known to interfere with the proliferation of different cell types, both prokaryotic and eukaryotic. As mechanical stress is known to induce the release of PUAs in other phytoplankton species, the release of 2-trans-4-trans-decadienal has been suggested to be a mean of deterring grazers, for example, zooplankton or fish larvae. P. pouchetii is a common component of northern and temperate spring blooms; it is grazed by zooplankton at normal rates and can also be a diet preferable to diatoms. Copepods may avoid gracing on healthy colonies of P. pouchetii, thus, the production and excretion of PUAs seems to be depending on the state of the cells or on environmental factors, or both. It has been reported that this PUA can be released into the sea in the absence of grazers, indicating that it may serve as an allelochemical, that is, a compound which gives P. pouchetii a competitive advantage over phytoplankton species blooming at the same time by inhibiting their growth.
The Dinophyta includes about a dozen genera, with at least 30 species, producing water and lipid soluble, low molecular weight, neuroactive secondary metabolites that are among the most potent nonproteinaceous poisons known. In general, these toxins have been shown to block the influx of sodium through excitable nerve membranes, thus preventing the formation of action potentials. The toxins are accumulated and sometimes metabolized by the shellfish which feed upon these dinoflagellates, causing different types of shellfish poisoning, such as PSP, DSP, and NSP.
Ingestion of contaminated shellfish results in a wide variety of symptoms, depending on the toxins(s) present, their concentrations in the shellfish and the amount of contaminated shellfish consumed. In the case of PSP, caused by toxin of Alexandrium spp., the effects are predominantly neurological and include tingling, burning, numbness, drowsiness, incoherent speech, and respiratory paralysis. Less well characterized are the symptoms associated with DSP and NSP.
DSP is caused by okadaic acid, a diarrhoric shellfish toxin and tumor promoter found in many dinoflagellates of the genera Dinophysis and in Prorocentrum lima. DSP is primarily observed as a generally mild gastrointestinal disorder, that is, nausea, vomiting, diarrhoea, and abdominal pain accompanied by chills, headache, and fever.
Both gastrointestinal and neurological symptoms characterize NSP, including tingling and numbness of lips, tongue, and throat, muscular aches, dizziness, reversal of the sensations of hot and cold, diarrhea, and vomiting. The NSP toxins are produced by Gymnodinium breve, also denominated as Ptychodiscus brevis.
Another syndrome caused by dinoflagellate toxins is the ciguatera poisoning, connected with eating contaminated tropical reef fish. Ciguatoxins that cause ciguatera poisoning are actually produced by Gambierdiscus toxicus, a photosynthetic dinoflagellate that normally grows as an epiphyte and has a relatively slow growth rate of approximately one division every 3 days. In its coral reef habitat, G. toxicus is biflagellate and swims if disturbed, but is usually motionless and attached to certain macroalgae. The dinoflagellate may also be associated with macroalgal detritus on the sea floor. Some scientists believe that the diverse symptoms of CFP are a result of a combination of several toxins or their metabolites, produced by one or more dinoflagellates. However, G. toxicus, which is found on a variety of macroalgae eaten by herbivorous fish, is now widely considered the single-celled source of ciguatoxins and the potential cause of CFP.
G. toxicus produces two classes of polyether toxins, the ciguatoxins (CTX) and maitotoxins (MTX). The CTX are lipophilic and are accumulated in fish through food web transfer. More than 20 CTX congeners have been isolated; however, only a few have been fully characterized structurally. The maitotoxins are transfused ladder-like polyether toxins, but are somewhat more polar, due to the presence of multiple sulfate groups. MTX was originally identified as a water soluble toxin in the viscera of surgeonfishes, and later found to be the principal toxin produced by G. toxicus. MTXs have not been demonstrated to bioaccumulate is fish tissues, possibly due to their more polar structure. Thus, if MTX is involved in ciguatera poisoning, it may be implicated only in ciguatera poisonings derived from herbivorous fishes.
The toxic potency of MTX exceeds that of CTX (respectively 10 ng/kg and 50 ng/kg in mice). Its mode of action has not been fully elucidated. Its biological activity is strictly calcium dependent and causes both membrane depolarization and calcium influx in many different cell types.
These toxins become progressively concentrated as they move up the food chain from small fish to large fish that eat them, and reach particularly high concentrations in large predatory tropical reef fish. Barracuda are commonly associated with ciguatoxin poisoning, but eating grouper, sea bass, snapper, mullet, and a number of other fish that live in oceans between latitude 358N and 358S has caused the disease. These fish are typically caught by sport fishermen on reefs in Hawaii, Guam and other South Pacific islands, the Virgin Islands, and Puerto Rico. Ciguatoxin usually causes symptoms within a few minutes to 30 h after eating contaminated fish, and occasionally it may take up to 6 h. Common nonspecific symptoms include nausea, vomiting, diarrhea, cramps, excessive sweating, headache, and muscle aches. The sensation of burning or “pins-and-needles,” weakness, itching, and dizziness can occur. Patients may experience reversal of temperature sensation in their mouth (hot surfaces feeling cold and cold, hot), unusual taste sensations, nightmares, or hallucinations. Ciguatera poisoning is rarely fatal. Symptoms usually clear in 1–4 weeks.
In its typical form, CFP is characterized initially by the onset of intense vomiting, diarrhoea, and abdominal pain within hours of ingestion of toxic fish. Within 12–14 h of onset, a prominent neurological disturbance develops, characterized by intense paraesthesia (tingling, crawling, or burning sensation of the skin) and dysaesthesia (painful sensation) in the arms, legs, and perioral region, myalgia, muscle cramping, and weakness. During this stage of the illness, pruritus and sweating are commonly experienced.
Pseudonitzschia spp. are among the several other marine algae that can produce domoic acid, the cause of ASP. Domoic acid was first isolated in Japan from the macroalgae species Chondria armata in 1958, and was consequently called after the Japanese word for macroalgae, which is domoi. Its identification in 1987 as a neurotoxin was first treated with scepticism, because this water-soluble amino acid was known as a folk medicine in Japan to treat intestinal pinworm infestations when used in very small doses. Production of domoic acid by algae seems to be a genetic property for a secondary metabolite with no known function in defence or primary metabolism.
Domoic acid can enter the marine food chain via uptake by molluscan shellfish such as mussels that filter their food out of the water. This water can contain both diatoms and the toxin, which is released to the water column (although there is no evidence yet that the toxin can be taken up directly). The toxin accumulates in the digestive gland and certain tissues of shellfish, and it appears to have no effect on the animals. Domoic acid may be metabolized by bacteria (e.g., of the genera Alteromonas and Pseudomonas) present in tissue of blue mussels (Mytilus edulis). Scallops are reported not to contain these elimination bacteria. Anchovies can also contain domoic acid in their guts, by feeding on toxic Pseudonitzschia spp.; this toxin affects their behavior and survival. Effects are also seen in seals. In humans, consumption of contaminated seafood mostly affected the elderly or infirm. Heat does not destroy domoic acid, although shellfish toxicity can decrease during cooking or freezing via domoic acid transfer from the meats to the surrounding liquid. The sea otter is the only animal known to be able to avoid intoxication, probably recognizing toxic prey by their odor. The mechanism of domoic acid toxicity is explained by its structural similarity with the excitatory neurotransmitter glutamic acid and its analogs, but with a much stronger receptor affinity.
Domoic acid is three times more potent than its analog kainic acid and up to 100 times more potent than glutamic acid. After exposure, domoic acid binds predominately to N-methyl-D-aspartate (NMDA) receptors in the central nervous system, causing depolarization of the neurones. Subsequently, the intercellular calcium concentration increases, resulting in sustained activation of calcium-sensitive enzymes, eventually leading to depletion of energy, neuronal swelling, and cell death. The affected neurones are mainly located in the hippocampus, explaining the most striking effect of domoic acid poisoning, which is short-term memory loss, observed in 25% of the affected persons in the 1987 contaminated mussel event. Other symptoms are confusion, nausea, vomiting, gastroenteritis, cramps and diarrhea, all within 24 h. Neurological complaints, including ataxia, headaches, disorientation, difficulty in breathing and coma, develop 48 h after
consumption. Permanent brain damage can also be caused by domoic acid intoxication. Effects of chronic low level ingestion are unknown. Domoic acid from mussels is more neurotoxic than the chemically pure compound. This increase is due to domoic acid potentiation, caused by high concentrations of glutamic and aspartic acids present in mussel tissue. This neurotoxic synergism occurs through a reduction in the voltage-dependent Mg2 block at the receptor associated channel, following activation of non-NMDA receptors, in addition to the NMDA receptor activation by domoic acid itself.
Intensive research over the last years has revealed a new class of phytotoxins produced by diatoms with more subtle and less specific effects, a discovery that has drawn a lot of attention as the diatoms have traditionally been regarded a key component of the food chain. Three closely-related PUAs have been isolated from Thalassiosira rotula, Skeletonema costatum, and Pseudonitzschia delicatissima, namely 2-trans-4-cis-7-cis-decatrienal, 2-trans-4-trans-7-cis-decatrienal and 2-trans-4-trans-decadienal. In the same study these aldehydes were found to inhibit cleavage of sea urchin embryos, reduce growth of Caco-2 cells and hatching of copepod eggs. The structural element shared by these compounds, the unsaturated aldehyde group, is able to form adducts with nucleophiles and is thus capable of inducing reactions that are toxic to the cell. The harmful effects of PUAs have been demonstrated at the organism level as inducers of apoptosis in sea urchin embryos, at the cell level as cytotoxicity in human cell lines and at the protein level by deactivation of enzymes. Table 7.8 summarizes toxic algae and the corresponding metabolites.
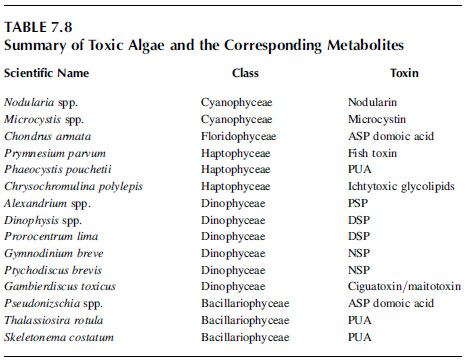
TABLE 7.8 Summary of Toxic Algae and the Corresponding Metabolites.





