Special Features of Diazotrophs
Many cyanobacteria capable of fixing nitrogen are filamentous and contain pale and thick walled cell (intercalary, lateral or terminal, single or in chains) called heterocyst. These are the sites of nitrogen fixation. Heterocysts are formed in the absence of utilizable combined nitrogen, such as ammonia, because it inhibits heteroyst differentiation and N2 fixing enzyme, the nitrogenase. As soon as cyanobacteria growing on ammonia-supplemented media are transferred to nitrogen medium, both heterocysts and nitrogenase develop parallelly. Besides, some strains, high light intensity inhibits N2 fixation. Heterocyst lacks oxygen evolving photosystem II, ribulose biphosphate carboxylase and may lack or have reduced amount of photosynthetic biliproteins (Wolk, 1982). Chlorophyll-a is present in the heterocysts. Wall of heterocyst contains O2 binding glycolipids which, together with respiratory consumption, maintain the anaerobic conditions (highly reduced atmosphere) necessary for N2 fixation (Bothe, 1982). In contrast, in vegetative cells adjacent to heterocysts, both photosystem I and II are present, therefore, oxygen evolution takes place by these cells.
Root nodules developed by Rhizobium (in leguminous plants) and Frankia (in non-leguminous plants) are the site of nitrogen fixation (see Symbiotic N2 fixation). However, lichens (a symbiotic structure formed by cyanobacteria and fungi) are the site of N2 fixation in lower group of microorganisms.
Presence of Nitrogenase and Reductants
All diazotrophs possess an enzyme nitrogenase which helps in conversion of N2 to NH3. Structure of nitrogenase is given in Fig. 11.1. It consists of two brown metalloproteins whose joint action is essential for reduction of N2 to IsfH3. Component I i.e. molybdo-ferro-protein (Mo-Fe-protein) which is also known as nitrogenase has a molecular weight of about 2.2 x 105 dalton. Nitrogenase contains molybdenum (2 atoms/mol), iron (32 atoms/mol) and a sulphur (30 atoms/mol). It is a larger unit than component II and non-sensitive to cold but losses activity at 0°C. On the other hand component II, nitrogenase reductase; is a smaller unit than the former one.

Fig. 11.1. Diagrammatic structure of nitrogenase. A, component I; B, component II; C, nitrogenase complex
It contains ferro-protein (Fe-protein) and has a molecular weight of about 5 x 104 dalton. It contains iron (4 atoms/ mol) and sulphur (4 atoms/mol), and is less stable than the component I (Sasson, 1984).
Thus, nitrogenase is an equilibrium mixture of Mo-fe-protein and Fe-protein in the ratio 1: 2 (Fig. 11.1).
Mo-Fe-Protein+2 (Fe-protein)
N2+12 Mg ATP + 6(H+ + e-)
In Rhodospirillum rubrum the physiological donors (e.g. ferredoxin I and II) of nitrogenase has been demonstrated to transfer reducing equivalent from the illuminated chloroplast to crude preparation of R. rubrum nitrogenase (Yoch and Arnon, 1975). Recently, it has been shown that pyruvate- 2-oxoglutartate and to a lesser extent, oxaloacetate supported nitrogenase activity in crude extract of R. rubrum (Ludden and Burris, 1981).
In addition to reduction of nitrogenase to ammonia, nitrogenase catalyses a number of other substrates. It has been found that nitrogenase isolated from R. rubrum catalyzed the reduction of acetylene, protons, cyanide, isocyanide and azide; all of which behaved as the substrates of nitrogenase from non-phototrophic bacteria (Munson and Burris, 1969).
On the basis of inhibitor studies, Hwang et al (1973) have proposed 5 sites for nitrogenase complex such as (i) N2 and N2 sites, (ii) acetylene site, (iii) azide, cyanide and methyl isocyanide sites, (iv) Co site, and (v) H+ site. However, the allocation of electrons to these substrates depends on their relative concentrations and the electron efflux through the complex. At low electron efflux, N2 competes poorly with acetylene and H+ as a substrate but at high electron efflux N2 becomes a very effective substrate as the dominant electron acceptor. The electron efflux can be altered by changing the concentration of Mg ATP or the ratio of the Fe-protein to Mo-Fe-protein (Hageman et al, 1980).
Presence of Hydrogenase
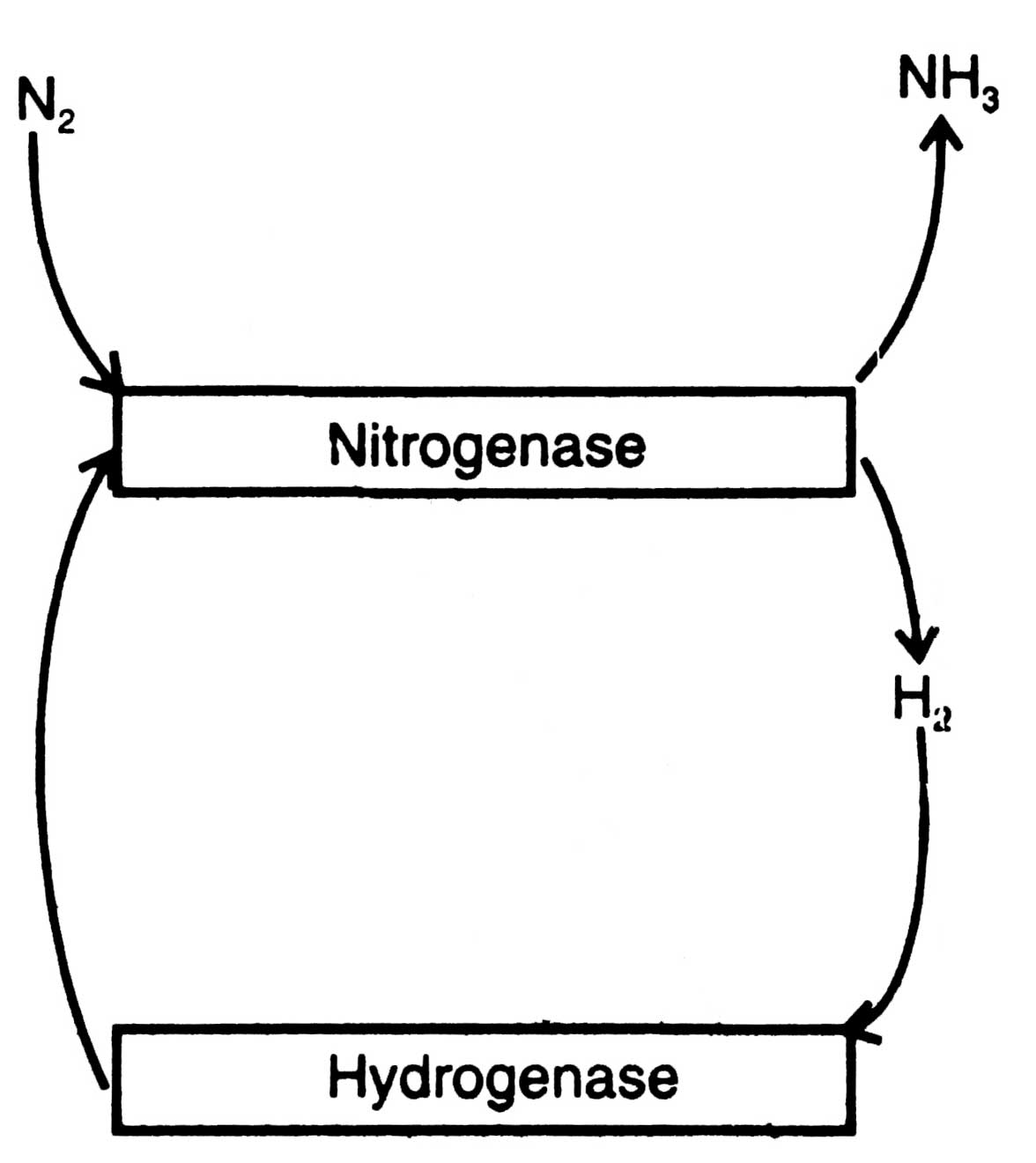
Many diazotrophs evolve hydrogen (H2) during N2 fixation which in turn inhibits N2 fixation reaction. To get protection from inhibition by H2 many diazotrophs possess, an enzyme hydrogenase to recycle H2 produced by nitrogenase (Fig. 11.2). Reutilization of H2 produces more ATP and improves the efficiency of N2 fixation (Johansson et al, 1983). For the first time, utilization of H2 by purple bacteria was discovered by Roelofsen (1935).
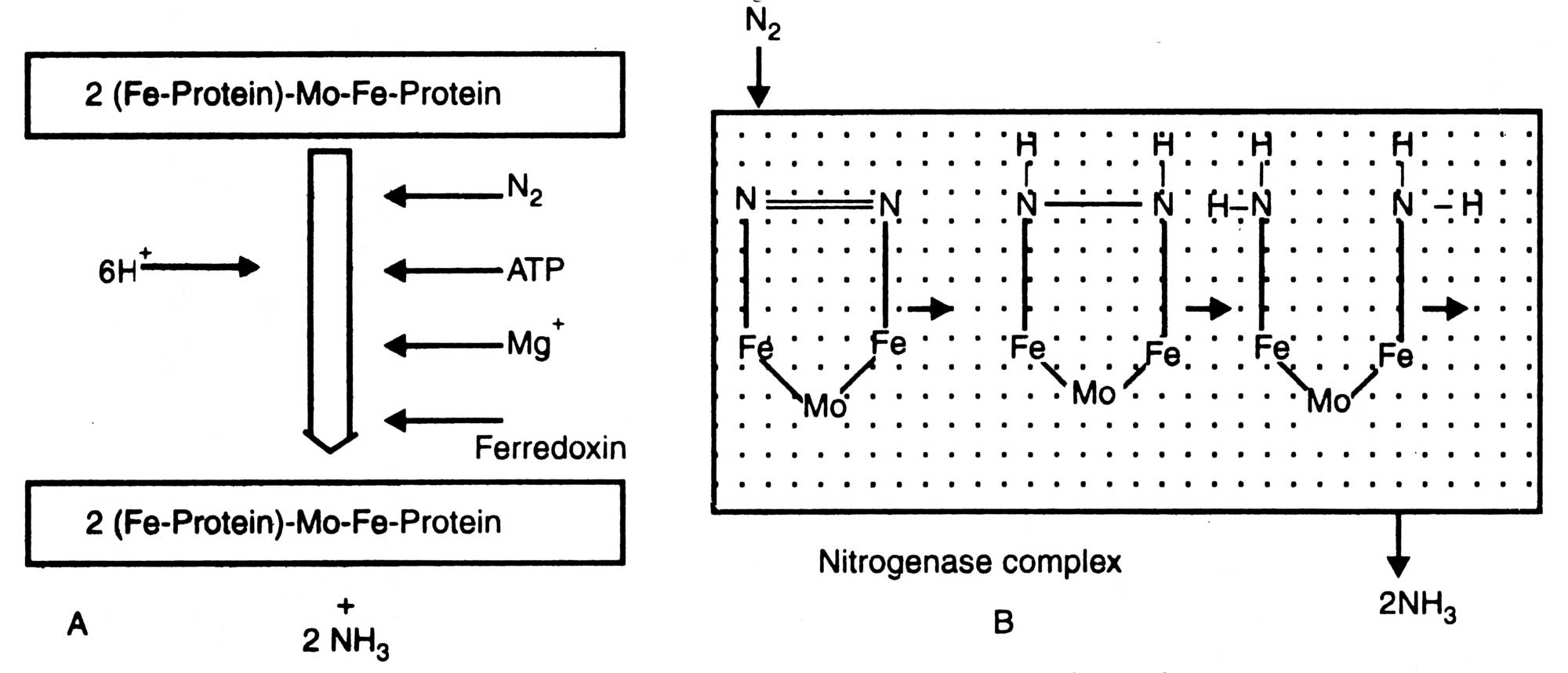
Fig. 11.3. Nitrogen fixation by nitrogenase complex (A) and mechanism of N2 conversion by component I (B).
Present knowledge indicates that the hydrogenase of purple bacteria is membrane-bound or at least membrane-associated.
It is cold labile and very sensitive to O2. The purified enzyme has a molecular weight of about 6,500 and most probably consists of a single polypeptide chain with 4 iron atoms and 4 acid labile sulphur atoms per molecule. It does not use NAD+ or NADP+ as electron acceptors.
Hydrogenase combines with H2 and reduces into a number of substrates (S) to form products (SH2) or liberates H2 from the reduced compounds as below :
| Hydrogenase | ||
| H2 + S | SH2 |
Ammonium salts are required for vegetative growth but in excess amount, it inhibits nitrogenase and declines N2 fixation process. It is obvious that ammonia is generated in microbial cells from N2 and at the same time is inhibitory to nitrogenase activity. Therefore, to continue N2 fixation process, the necessary step is to convert ammonia rapidly to organic nitrogen compounds or be removed from intracellular site of N2 metabolism (Alexander, 1977).
In diazotrophs nitrogenase activity is regulated by a complex mechanism developed in different microorganisms in different ways. Oxygen is required by aerobic bacteria and evolved by cyanobacteria. However, O2 inhibits nitrogenase activity. To carry out N2 fixation process efficiently, diazotrophs possess certain physiological adaptations as the defense mechanisms so that they can protect nitrogenase and carry out the N2 fixation process.
Azotobacters are among the most O2 tolerant free living diazotrophs. The molecular features .that allow this tolerance to O2, include an O2 excluding and protective protein that complexes with nitrogenase in vitro during the period of O2 stress (Haaker and Keeger, 1977; Robson, 1979). In Azotobacters protection is given to cell by their high respiratory rate. Due to which O2 is prevented from reaching the site of nitrogenase reaction.
In cyanobacteria, creation of O2 tension is remarkable as the photoautotrophs evolve O2 which inhibits nitrogenase activity. Hydrolysis of ATP in N2 fixing system provides an anhydrous atmosphere around the active site of nitrogenase and keeps enzyme in its most active form and activates electrons.
In heterocystous cyanobacteria, the compartmentation of photosynthesis and N2 fixation takes place. N2 is reduced only in heterocysts where photosynthetic systems (thylakoids) are in reduced amount or absent, therefore, nitrogenase is not inhibited by O2 evolved during photosynthesis.