Glycerol Spraying/Low-Angle Rotary Metal Shadowing
Heavy-metal replication (Williams and Wyckoff, 1945) is one of the oldest and yet most effective methods to provide enhanced contrast when imaging biological specimens in a transmission electron microscope (TEM) (for review, see Fowler and Aebi, 1983; Mould et al., 1985; Nermut and Eason, 1989). This technique requires efficient adsorption of the sample to a specimen support so that the biomolecules or particles to be visualized become well dispersed and distributed evenly. Moreover, to minimize shadow-casting artifacts produced by corrugations of the specimen support, it should ideally be smooth at atomic dimensions. Mica fulfills both these requirements and hence emerges as an ideal specimen support. For a good replica, the contrast-enhancing metal should produce a fine grain, detach easily from the specimen support, and be stable in the electron beam. An example of a metal that favorably meets these three criteria is platinum (Pt).
Glycerol spraying/low-angle rotary shadowing has been claimed to preserve native conformations comparably to cryotechniques (Mould et al., 1985; Nermut and Eason, 1989). It has been used time and again to characterize complex macromolecules at near-native conditions and especially also proteoglycans (Moergeli et al., 1985; Rodriguez et al., 1995; Scheel et al., 1999, Hofmann et al., 2000; Griffith et al., 2002). Indeed, a stabilization of the native confirmation by glycerol against temperature-, pH-, or organic solvent-induced changes was corroborated by electron spray mass spectrometry (Grandori et at., 2001).
Specimens that are suitable for glycerol spraying/ low-angle rotary metal shadowing range from single protein (e.g., myosin) and DNA molecules to relatively stable biopolymers or supramolecular assemblies such as intermediate filaments or protein/glycosaminoglycan complexes. The achievable resolution is a few nanometers and thus sufficient to reveal the overall size and shape and/or the principal domain architecture of many biomolecules.
II. MATERIALS AND INSTRUMENTATION
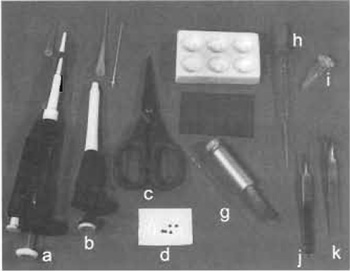
- Gilson Pipetman adjustable pipette P20, with tips (Fig. 1a), and Gilson Microman adjustable pipette M25, with tips (Fig. 1b) (http://www.gilson.com/)
- Vortex apparatus
- Pair of scissors (Fig. 1c).
- Double-sided adhesive Scotch tape
- Specimen support grids (Fig. 1d): For example, from Pelco International (http://www.pelcoint.com/) (typically, 400 mesh/in, copper grids, 3.05mm in diameter and 0.7mm thick)
- Spot plate (Fig. 1e): For example, from BAL-TEC (http://www.bal-tec.com/) (Cat. No. B 8010 030 83)
- Mica sheets (Fig. 1f): For example, from BALTEC (Cat. No. BU 006 027-T)
- 25-µl glass micropipettes with pipetter (Fig. 1 g)
- Pasteur pipettes (Fig. 1h)
- Eppendorf tubes (Fig. 1i)
- Precision forceps: For example from Electron Microscopy Sciences (http://www.emsdiasum.com/ ems/). A pair of fine, straight forceps (Fig. 1j) and a pair of fine, bent forceps (Fig. 1k).
- Compressed air at a pressure of 0.8 bar (80psi)
- Custom-built spray apparatus with air-pressure controller (Figs. 2d and 2e). The major elements or pieces to be manufactured or purchased include
A polyvinyl chloride (PVC) frame mounted onto an aluminum base
A Pasteur pipette connected to a compressed air supply via an air pressure/volume controller unit so that a set volume of a fine stream of air is produced at its nozzle by the push of the release button on the controller
A clamp mounted on a post to fasten a 25-µl glass micropipette containing the sample so that it points to the center of the stream of pressurized air
A metal sphere on a height-adjustable post so that it can be placed into the center of the fine air stream and disperse the sample drops evenly
A curved glass tube that traps the large sample droplets, aggregates, and other heavy particles
A square piece of freshly cleaved mica that is fastened with a piece of double-sided adhesive tape below the curved glass tube - Glow-discharge apparatus: For example, custom built as detailed by Aebi and Pollard (1987) (see also article by Baschong and Aebi) or SCD 005 (Cat. No. BU GO5 750 116-T) from BAL-TEC.
- High-vacuum evaporation unit: For example, BAE 080-T (Cat. No. BU P03 527) from BAL-TEC specifications:
- High-vacuum pumping unit with rotary vane pump
- Vacuum measuring device for rough and high vacuum
- Evaporation unit with controller, high current supply, and cross-shaped vacuum chamber
- Accessories for BAL-TEC high-vacuum evaporation unit:
- Cold trap for BAE 080-T (Cat. No. BB 176 942- W)
- Quick-release flange WF 206, including EK- 030 electron gun (Cat. No. BU 007 038-T)
- Quick-release flange WF 211, rotary table (Cat. No. BB 192 281-T)
- Quick-release flange WF 204, C-evaporator (Cat. No. BB 192 276-T)
- Digital quartz-crystal thickness meter QSG 060 (Cat. No. BU M01 259)
- Electron beam evaporation device EVM 030, (Cat. No. BU S04 000)
- Materials for BAL-TEC high-vacuum evaporation unit:
- Carbon rods for Pt/C evaporator (Cat. No. BU 006 101-T)
- Platinum inserts (Cat. No. BU 006 103-T)
- Carbon rods for C-evaporator (Cat. No. BU 006 115)
- Tungsten cathodes (Cat. No. BU 020 023-T)
- Liquid nitrogen
Solutions
- Glycerol 100% anhydrous: For example, from Fluka (http://www.sigmaaldrich.com/)(Cat. No. 49770)
- Sodium hypochlorite solution, 13-15%, technical grade: For example, from Siegfried
(http://www. siegfried.ch/) (Cat. No. 180550-01)
A. Spraying
Steps
- Hold a piece of mica with a pair of straight forceps and cut it into square pieces (5-7mm side length) with a pair of scissors (Fig. 2a).
- Dilute the sample to a concentration of about 10- 100nM (0.1-1.0 mg/ml for most proteins) and pipette 20µl into an Eppendorf tube.
- Add 8.6µl of 100% glycerol to the sample (i.e., 30% final glycerol concentration) using the Gilson Microman M25. Mix thoroughly with the pipette and vortex.
- Draw 10 µl of the glycerol-containing solution into a 25-µl glass micropipette and mount it on the spray apparatus (Figs. 2d and 2e).
- With two pairs of forceps, cleave the mica into two sheets (Figs. 2b and 2c).
- With its freshly cleaved side up, place the mica below the bent glass tube of the spray apparatus. During spraying, the mica sheet should either be fixed with a piece of double-sided adhesive tape as shown in Fig. 2e or held in position with a pair of forceps.
- Briefly (i.e., for about 1s) press the button of the airpressure control unit (depicted in Fig. 2d) to spray the sample suspension onto the freshly cleaved mica.
Note: The metal sphere placed in the spray path disperses the sample droplets evenly. The curved glass tube traps remaining large droplets, aggregates, and other heavy particles that will not follow the air flow when the spray path changes its direction by 90° . - Repeat spraying in the same way with another 10 µl of suspension onto the second piece of mica, etc.
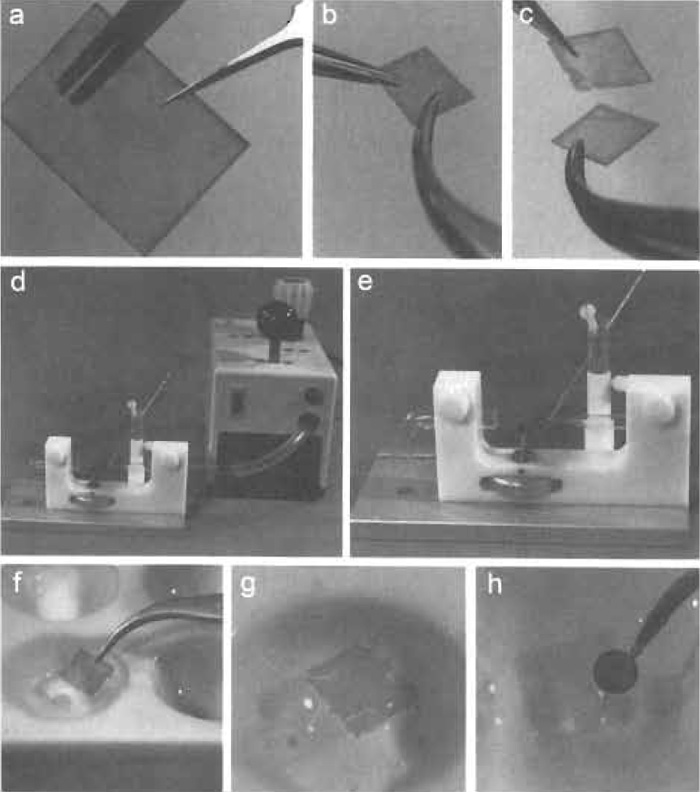 |
| FIGURE 2 Selected steps of the glycerol spraying/low-angle rotary metal shadowing technique. (a) A piece of mica held with a pair of straight forceps is cut into square pieces with a pair of scissors. (b and c) Using forceps, the mica is next cleaved into two sheets. (d and e) The glycerol-containing solution is drawn into a 25-µl glass micropipette that is mounted on the spray apparatus such that it points to the center of the stream of pressurized air that is focused by a Pasteur pipette. (f) The Pt/C-shadowed and C-backed mica is slowly immersed into water, and (g) the mica sheet is removed once the Pt/C+C-replica is floated off. (h) The replica is then placed on a specimen grid with a pair of forceps. |
Steps
- Switch on the thin film quartz monitor.
- Set up the electron gun for Pt/C evaporation and the carbon evaporation unit for C-evaporation according to the instruction manual of the supplier.
- The recommended working distance between the Pt/C tip in the electron gun and the middle of the table is 12 cm and the tilt angle of the table relative to the evaporation axis should be around 5° . For the C tips in the C evaporation unit, the recommended working distance is the same but the tilt angle should be 80-90° .
- Mount the mica with double-sided adhesive tape onto the table. Make sure that the sprayed side of the mica is up!
- Mount the table onto the rotating base in the vacuum chamber and start evacuating.
- After 30 min, the vacuum meter should read better than 2 × 10-5 mbar.
- Pour liquid nitrogen into the Meissner trap.
- After an additional 5min, the vacuum meter should read better than 6 × 10-6 mbar.
- Degas Pt/C tip according to the instruction manual of the supplier.
- Start the motor of the rotating base (set to about 120rpm).
- Set the thin film quartz monitor to zero.
- Start evaporating Pt/C according to the instruction manual. Open the manual shutter.
- Read the frequency on the thin film quartz monitor.
- After the frequency has changed by 300 Hz, close the manual shutter and stop evaporation.
- Evaporate C according to the instruction manual of the supplier.
- On a white reference the carbon layer should look deep brown (cf. Figs. 2f and 2g).
Note: It is advisable to calibrate current and evaporation times of the power supply so that standard settings can be used. - Turn the rotating table off, break the vacuum, and remove the rotating table from the vacuum chamber.
C. Floating off the PT/C+C Replica
Steps
- Fill the wells of the spot plate (Fig. le) with distilled water (Fig. 2f).
- As illustrated in Fig. 2f, slowly immerse the mica at an angle of ~45° holding it firmly with the forceps.
- Submerge the mica completely and remove it when the Pt/C+C-replica is floating on the water surface (Fig. 2g).
- With a pair of forceps, submerge a freshly glowdischarged specimen grid, place it underneath the Pt/C+C-replica, and use it as a sieve to collect the replica piece by piece.
- Dry the specimen grids by layering them with the film side up onto a piece of filter paper.
- The specimen grids are now ready for inspection in the EM.
D. Electron Microscopy
Steps
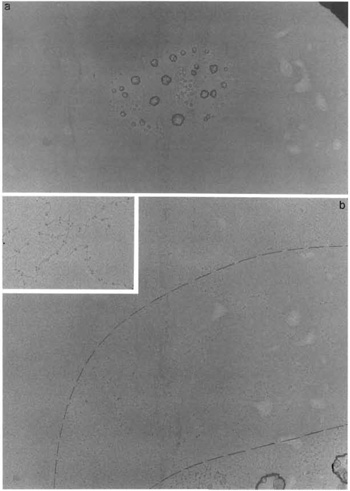
- Identify droplet centers as shown in Fig. 3a at a low magnification of, e.g., 5000×. Note: The droplet centers are easy to find if your sample is kept in nonvolatile buffers and/or contains salts. The droplet center size varies from 5 to 50 µm in diameter. Choose droplet centers with a size of about 5-10µm.
- Increase the magnification to about 20,000-50,000×. Set coarse focusing and then slowly move from the center toward the edge of the droplet.
- Dashed lines in Fig. 3b highlight the sharp transition from salt crystals to a clean background that is typically observed over increments of the droplet center distance by 0.5-2 µm. In this "clean belt" around the droplet center well-spread and well-preserved protein particles are usually found readily (Fig. 3b, inset).
IV. PITFALLS
- Replica resists detaching from the mica. Occasionally, the Pt/C+C replica sticks to the mica. The replica usually detaches more easily after the mica has been incubated in a moist atmosphere at 37°C for 30min. Alternatively, the replica may also be floated off onto 6% sodium hypochlorite rather than water; however, after this treatment, the replica has to be transferred with a platinum loop to distilled water and allowed to incubate for 10 min. One reason for frequent sticking of the Pt/C+C replica to the mica may be too good a vacuum during C evaporation. Breaking the vacuum after Pt/C evaporation, reevacuation to 7 × 10-5 mbar, and subsequent C evaporation may solve this problem.
- Preparation not satisfactory: (a) Specimen dependent: The specimen may not be optimal for glycerol spraying. Some specimens, including actin filaments, are relatively sensitive to shearing forces and may thus break into small pieces on spraying. (b) Buffer dependent: Many buffers and solutions can be used but detergents or high concentrations (i.e., ≥2M) of urea or guanidine hydrochloride cause problems through eutectic effects. In this case, the buffer should be changed or diluted.
- Phase separation after addition of glycerol. For good results, it is essential that the sample is a solution that contains glycerol. In case of phase separation, different buffers should be tried. If volatile buffers (e.g., ammonium acetate) are used, the evaporating unit should be evacuated for 2-3h rather than for 30min before metal evaporation (see earlier discussion).
This work was supported by the M. E. Mueller Foundation of Switzerland and the Canton Basel-Stadt.
References
Aebi, U., and Pollard, T. D. (1987). A glow discharge unit to render electron microscope grids and other surfaces hydrophilic. J. Electron. Microsc. Tech. 7, 29-33.
Fowler, W. E., and Aebi, U. (1983). Preparation of single molecules and supramolecular complexes for high-resolution metal shadowing. J. Ultrastruct. Res. 83, 319-334.
Fowler, W. E., and Erickson, H. P. (1983). Electron microscopy of fibrinogen, its plasmic fragments and small polymers. Ann. N. Y. Acad. Sci. 408, 146-163.
Grandori, R., Matecko, I., Mayr, P., and Mueller, N. (2001). Probing protein stabilization by glycerol using electrospray mass spectroscopy. J. Mass Spectrom. 36, 918-922.
Griffith, J. D., Lindsey-Boltz, L. A., and Sancar, A. (2002). Structures of the human Rad17-replication factor C and checkpoint Rad 9-1-1 complexes visualized by glycerol spray/low voltage microscopy. J. Biol. Chem. 277, 15233-15236.
Hall, C. E. (1956). Visualization of individual macromolecules with the electron microscope. Proc. Natl. Acad. Sci. USA 42, 801-806.
Hofmann, I., Mucke, N., Reed, J., Herrmann, H., and Langowski, J. (2000). Physical characterization of plakophilin 1 reconstituted with and without zinc. Eur J. Biochem. 267, 4381-4389.
Mould, A. P., Holmes, D. F., Kadler, K. E., and Chapman, J. A. (1985). Mica sandwich technique for preparing macromolecules for rotary shadowing. J. Ultrastruct. Res. 91, 66-76.
Moergelin, M., Paulsson, M., Malmstrom, A., and Heinegard, D. (1989). Shared and distinct structural features of interstitial proteoglycans from different bovine tissues revealed by electron microscopy. J. Biol. Chem. 264, 12080-12090.
Nermut, M. V., and Eason, P. (1989). Cryotechniques in macromolecular research. Scan. Microsc. Suppl. 3, 213-224; discussion 224-225.
Roughley, P. J., Rodriguez, E., and Lee, E. R. (1995). The interactions of "non-aggregating" proteoglycans. Osteoarthritis Cartilage 3, 239-348.
Scheel, J., Pierre, P., Rickard, J. E., Diamantopoulos, G. S., Valetti, C., van der Goot, F. G., Haener, M., Aebi, U., and Kreis, T. E. (1999). Purification and analysis of authentic CLIP-170 and recombinant fragments. J. Biol. Chem. 274(36), 25883-25891.
Tyler, J. M., and Branton, D. (1980). Rotary shadowing of extended molecules dried from glycerol. J. Ultrastruct. Res. 71, 95-102.
Williams, R. C., and Wyckoff, R. G. W. (1944). The thickness of electron microscopic objects. J. Appl. Phys. 15, 712-716.