Ascorbate
Ascorbic acid (vitamin C; Fig. 2) acts as a water-soluble free radical scavenger in both plant and animal tissues. Like phenolics, ascorbate (E°´= 282 mV) has a reduction potential below peroxyl radicals (E°´=1000 mV) and thus can inactivate peroxyl radicals. In addition, ascorbate’s reduction potential is lower than the α-tocopherol radical (E°´= 500 mV), meaning that ascorbate may have an additional role in the regeneration of oxidized α-tocopherol. Interactions between ascorbate and free radicals result in the formation of numerous oxidation products. Although ascorbate seems to primarily play an antioxidant role in living tissues, this is not always true in food systems. Ascorbate is a strong reducing agent especially at low pH. When transition metals are reduced, they become very active prooxidants that can decompose hydrogen and lipid peroxides into free radicals. Ascorbate also causes the release of protein-bound iron (e.g., ferritin), thus promoting oxidation. Therefore, ascorbate can potentially exhibit prooxidative activity in the presence of free transition metals or iron-binding proteins. This does not typically occur in living tissues due to the tight control of free metals by systems that prevent metal reduction and reactivity. However, in foods the typical control of metals can be lost by processing operations that cause protein denaturation. Thus in some foods, ascorbate my act as a prooxidant and accelerate oxidative reactions.Ascorbate is found in numerous plant foods including green vegetables, citrus fruits, tomatoes, berries, and potatoes. Ascorbate can be lost in foods due to heat processing and prolonged storage. Transition metals and exposure to air will also cause the degradation of ascorbic acid.
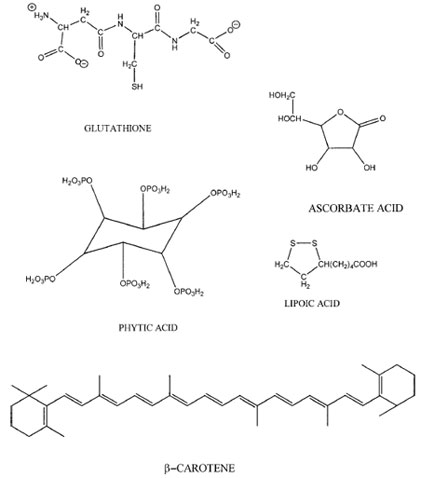 |
| FIGURE 2 Chemical structures of miscellaneous natural antioxidants. |




