Metabolism
The two major carbon sources utilized by cells to synthesize organic molecules are(1) complex organic molecules, such as sugars and amino acids, and
(2) single-carbon compounds, such as CO2 or methane (CH4). Cells that use CO2 as their sole source of carbon are called autotrophs, and cells that require complex organic compounds are referred to as heterotrophs. Cells that can obtain energy from light are called phototrophs, and cells that require chemical energy are called chemotrophs.
Glycolysis is a nearly universal process in which the six carbon sugar glucose is anaerobically converted, through a series of enzymatically catalyzed steps in the cytosol, the fluid portion of the cytoplasm, into two molecules of the three carbon compound pyruvate. Two molecules of ATP are expended early on in glycolysis, but four more are generated later by substrate-level phosphorylation. Thus, there is a net production of two ATP molecules per molecule of glucose. In addition, two molecules of nicotinamide adenine dinucleotide (NAD) become reduced by gaining two electrons.
Either fermentation or respiration may follow glycolysis (Figure 1-3). Fermentation is an oxygen-independent process, occuring in the cytosol, which uses organic molecules as terminal electron acceptors. Fermentation regenerates the supply of NAD+ for glycolysis and results in the consumption of pyruvate and the release of molecules such as CO2 or H2 (gases); lactic, formic, acetic, succinic, butyric, or propionic acids; and ethanol, butanol, or propanol (alcohols). The final product depends on the species. No additional ATP is generated during fermentation.
Respiration involves the oxidation of molecules, the generation of high-energy molecules, such as ATP, by passing pairs of electrons (and hydrogen ions, or protons) through an electron transport system, and the donation of these electrons to an inorganic electron acceptor. If the terminal electron acceptor is oxygen, this process is termed aerobic respiration. Anerobic respiration occurs when the terminal electron acceptor is an inorganic molecule other than molecular oxygen (such as sulfate or nitrate). Organisms vary in their oxygen requirements; some are strict anaerobes and cannot survive in the presence of oxygen. Facultative anaerobes can respire aerobically or anaerobically, and obligate aerobes require oxygen for survival.
Pyruvate generated from glycolysis in the cytosol may enter the mitochondria and, if oxygen is available, be enzymatically converted to acetyl coenzyme α(acetyl CoA) and CO2. Within the matrix of the mitochondria or the cytosol of aerobic prokaryotes, the two-carbon acetyl CoA enters a circular set of enzymatic reactions known as the Krebs cycle, the tricarboxylic acid cycle (TCA), or the citric acid cycle (Figure 1-3).
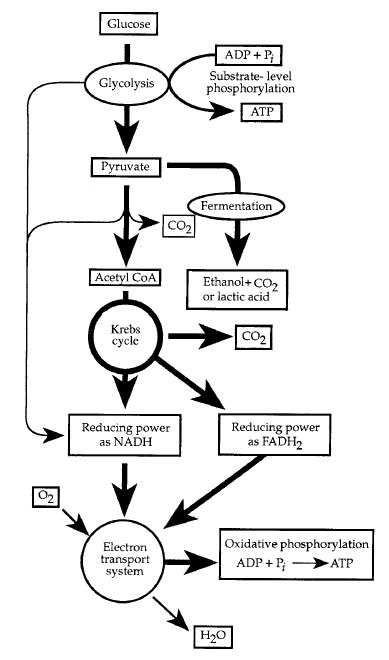 |
| Figure 1-3 Chemoheterotrophic metabolism. |
During oxidation of a substrate, two major electron carriers, NAD+ and FAD, become reduced to NADH and FADH2. One complete turn of the TCAproduces three molecules of NADH, two molecules of CO2, one molecule of FADH2, and one molecule of guanosine triphosphate (GTP). The electrons and H+ ions from NADH and FADH2 are transferred to the electron transport chains within the cristae of the mitochondria or the plasma membrane of prokaryotes. These chains consist of series of proteins that first serve as electron acceptors, then donors to the next complex in the chain. This series of coupled oxidations and reductions results in the terminal tranfer of electrons and H+s to oxygen, forming water as the end product.
The complete oxidation of glucose:
C6H12O6 + 6O2 → 6CO2 + 6H2O
ATP can be generated by three different mechanisms. It can be formed from adenosine diphosphate (ADP) by either substrate-level phosphorylation or oxidative phosphorylation. In substrate-level phosphorylation, an enzyme mediates the transfer of a phosphate group from a phosphorylated organic molecule to ADP. Oxidative phosphorylation occurs when molecules are oxidized and energy is extracted from the electrons by passing them through an electron transport system, where most of the resulting free enrgy is used to drive the phosphorylation of ADP, producing ATP. Photophosphorylation also synthesizes ATP, but uses the energy from sunlight rather than from the breakdown of organic molecules.




