Soil Testing for Phosphorus
Crop response to phosphorus is correlated poorly to the total amount of phosphorus in a soil. Therefore, a successful soil test should represent some index of phosphorus availability. The development of a soil test requires selection of an extractant, development of studies that correlate the amount of nutrient extracted with phosphorus accumulation by crops, and calibration studies that determine a relationship between soil test results and amount of fertilizer required for optimal production.Over the past century, a number of soil-testing procedures have been proposed, and several excellent reviews on soil testing for phosphorus have been published (50-53).
This section focuses on historical developments, mode of action, and generalized interpretations of the major phosphorus soil tests utilized in the United States.
The major soil tests that have been used or proposed in the United States are summarized in Table 3.2. Most early soil tests were developed empirically and were based on simple correlations between extractant and some measure of crop response to fertilization with phosphorus. However, based on the phosphorus-fractionation method developed by Chang and Jackson (54), inferences have been made concerning the mode of action, or the forms of phosphorus extracted by various solutions. The inferred modes of action for various chemical extractant components are presented in Table 3.3. Generally, water or dilute salt solutions characterize phosphorus in the soil solution or the intensity factor, whereas acids, complexing solutions, or alkaline buffer solutions generally characterize the quantity factor. Tests based on water extraction often correlate well with phosphorus accumulation in shallowrooted, fast- growing vegetable crops. However, soil tests capable of better characterizing the labile fraction and capacity factor generally produce more reliable results for field and orchard crops.
An early soil test for phosphorus aimed at characterizing available phosphorus was the 1% citric acid test developed by Dyer (3). This test was adapted in England but was not used widely in the United States. A dilute acid test proposed by Truog (4) and a test based on a universal soil extracting solution proposed by Morgan (5) were among the earliest soil tests used in the United States.
 |
The test based on the Bray-I extractant was perhaps the first to be implemented widely in soil-testing laboratories in the United States, and it is still extensively used in the midwestern United States. This mild-acid solution has been shown reliably to predict crop response to phosphorus fertilization on neutral to acidic soils. However, the test is much less effective in basic soils, where the acid is neutralized quickly by the soil bases present and fluoride ions are precipitated by calcium (55).
In the southeastern United States, the Mehlich 1 (M-I) soil-test extractant is used commonly for simultaneous extraction of P, K, Ca, Mg, Cu, Mn, Fe, and Zn. The M-I soil test does not correlate with crop response on calcareous soils probably for the same reasons the Bray-I test does not. Consequently, the Mehlich 2 (M-II) test was introduced as an extractant that would allow simultaneous determinations of the same nutrients over a wide range of soil properties. However, the corrosive properties of the M-II in instruments discouraged wide acceptance of this extractant and prompted modifications that ultimately became the Mehlich 3 (M-III) extraction. The M-III has been shown to be reliable across a wide range of soil-crop production circumstances (56,57).
The sodium bicarbonate (NaHCO3) (58) soil test for phosphorus generally correlates well with crop response on calcareous soils in the western United States. The NH4HCO3-DPTA (diethylenetriaminepentaacetic acid) soil test also has been used for the simultaneous determination of P, K, Zn, Fe, Cu, and Mn (59,60) and performs similar to the NaHCO3 test with respect to phosphorus. Another test that shows good correlations on calcareous soils is the EDTA (ethylenediaminetetraacetic acid) soil test (61).
Isotopic dilution techniques (53) and phosphorus sorption isotherms (62) have been used not only to characterize the labile phosphorus fraction but also the phosphorus-buffering capacity of soils. However, these approaches are too tedious and costly to be used as routine soil tests.
Ultimately, soil-test phosphorus levels must be converted into phosphorus fertilizer recommendations for crops. A useful starting point is the determination of critical soil-test levels, that is the soil-test phosphorus level above which there is no response to phosphorus fertilizer. An example of a critical phosphorus soil-test level based on water extraction for celery is shown in Figure 3.5. Using the double calibration approach described by Thomas and Peaslee (50) information on how much fertilizer is required to achieve the critical concentration would result in a fertilizer recommendation. This approach is used for Histosols by the Soil Testing Laboratory at the University of Florida. An example of resulting fertilizer recommendations for several commodities is shown in Figure 3.6.
The laboratory mentioned above makes recommendations for Histosols over a limited geographical location. However, most soil-testing laboratories make recommendations over large geographical area and across more diverse soil types. Under most situations, quantitative information on how phosphorus fertilizer additions change with soil-test phosphorus levels across a range of soil types rarely exist. Owing to this uncertainty, most soil-testing laboratories make phosphorus fertilizer recommendations based on probability of response using class interval grouping such as low, medium, and high.
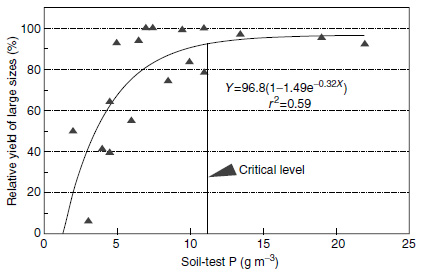 |
| FIGURE 3.5 Critical soil-test phosphorus levels for large, harvest-size celery on Florida Histosols. (Adapted from C.A. Sanchez et al., Soil Crop Sci. Soc. Fla. Proc. 29:69–72, 1989.) |
Crops produced on a soil scoring very low or low have a very high probability of responding to moderate to high rates of fertilization. Crops produced on soils classified as medium frequently respond to moderate rates of fertilization, and typically, crops produced on soils testing high for phosphorus would not respond to fertilization (Table 3.4). General soil-test phosphorus interpretations for mineral soils in California and Florida are shown in Tables 3.5 and 3.6 for comparative purposes. In California, only the probability of response to NaHCO3- phosphorus is indicated, and it is presumed that specific fertilizer recommendations are left to service laboratories, crop consultants, or the grower. In Florida, specific fertilizer recommendations for phosphorus are made for each level of M-I-extractable phosphorus. Furthermore, research aimed at validating and calibrating soil-test fertilizer recommendations for phosphorus in Florida is ongoing (63-65). It must be stressed that all fertilizer recommendations must be calibrated locally, and readers are advised to consult the cooperative extension service for recommendation guidelines specific to their region.
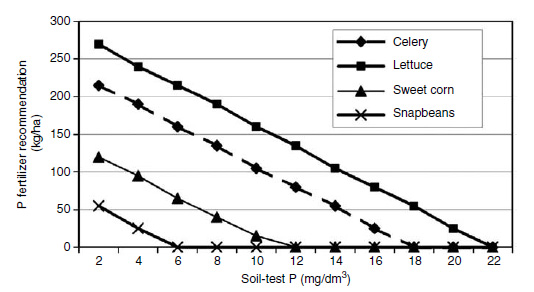 |
| FIGURE 3.6 Fertilizer phosphorus recommendations for selected crops on Everglades Histosols. (Adapted from C.A. Sanchez, Soil Testing and Fertilizer Recommendations for Crop Production on Organic Soils in Florida. University of Florida Agricultural Experiment Station Bulletin 876, Gainesville, 1990.) |
 |
 |




