Nutrient cycles
ContentAll the plant nutrients are in continuous circulation between plants, animals, the soil and the air. The processes contributing to the production of simpler inorganic substances, such as ammonia, nitrites, nitrates, sulphates and phosphates, are sometimes referred to as mineralization. Mineralization yields chemicals that are readily taken up by plants from the soil solution. The formation of humus, organic residues of a resistant nature, is known as humification. Both mineralization and humification are intimately tied up in the same decomposition process, but the terms help identify the end product being studied. Likewise it is possible to follow the circulation of carbon in the carbon cycle and nitrogen in the nitrogen cycle, although these nutrient cycles along with all the others are interrelated.
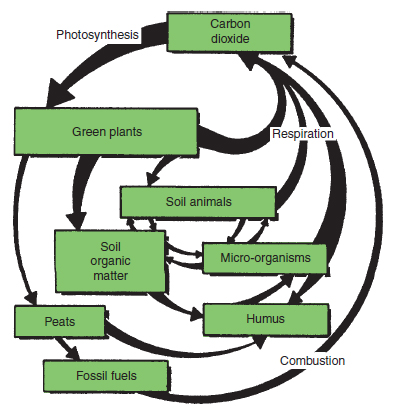
The carbon cycle Green plants obtain their carbon from the carbon dioxide in the atmosphere and, during the process of photosynthesis, are able to fix the carbon, converting it into sugar. Some carbon is returned to the atmosphere by the green plants themselves during respiration, but most is incorporated into plant tissue as carbohydrates, proteins, fats, etc. The carbon incorporated into the plant structure is recycled and eventually released as carbon dioxide, as illustrated in Figure 18.4. All living organisms in this food web release carbon dioxide as they respire. The sugars, cellulose, starch and proteins of succulent plant tissue, as found in young plants, are rapidly decomposed to yield plant nutrients and have only a short-term effect. In contrast, the lignified tissue of older plants rots more slowly. Besides the release of nutrients, humus is formed from this fibrous and woody material, which has a long-term effect on the soil. Plants grown in the vicinity of vigorously decomposing vegetation, e.g. cucumbers in straw bales, live in a carbon dioxide enriched atmosphere. Carbon dioxide is also released on combustion of all organic matter, including the fossil fuels such as coal and oil. Organic materials such as paraffin or propane, which do not produce harmful gases when burned cleanly, are used in protected culture for carbon dioxide enrichment. The nitrogen cycle The nitrogen cycle similarly follows the fate of nitrogen in its many forms in the plant, the soil and the atmosphere. The sulphur cycle Sulphur is an essential constituent of plants that accumulates in the soil in organic forms. This sulphur does not become available to plants until aerobic micro-organisms mineralize the organic form to produce soluble sulphates. Under anaerobic conditions there are micro-organisms which utilize organic sulphur and produce hydrogen sulphide, which has a characteristic smell of bad eggs often evident in waterlogged soils in warm conditions. Carbon to nitrogen ratio (C:N) All nutrients play a part in all nutrient cycles simply because all organisms need the same range of nutrients to be active. Normally there are adequate quantities of nutrients, with the exception of carbon or nitrogen, which are needed in relatively large quantities. A shortage of nitrogenous material would lead to a hold-up in the nitrogen cycle, but would also slow down the carbon cycle, i.e. the decomposition of organic matter is slowed because the micro-organisms concerned suffer a shortage of one of their essential nutrients. A useful way of expressing the relative amounts of the two important plant foods is in the carbon to nitrogen (C:N) ratio. Plant material has relatively wide C:N ratios, but those of microorganisms are much narrower. This is because micro-organisms utilize about three quarters of the carbon in plants during decomposition as an energy source. The carbon utilized this way is released as carbon dioxide, whereas, usually, all the nitrogen is incorporated in the microbial body protein. This concentrates the nitrogen in the new organism that is living on the plant material. Sometimes the C:N ratio is so wide that some nitrogen is drawn from the soil and ‘locked up’ in the microbial tissue. This is what happens when straw (and similar fibrous or woody material such as wood chips and bark) with a ratio of 60:1 is dug into the soil (see Table 18.1). For example, if one thousand 12 kg bales of straw are dug into one hectare of land then the addition to the soil will be 12 000 kg of straw containing 4800 kg of carbon and 80 kg of nitrogen. Three-quarters of the carbon (3600 kg) is utilized for energy and lost as carbon dioxide and a quarter (1200 kg) is incorporated over several months into microbial tissue. Microbial tissue has a C:N ratio of about 8:1, which means that by the time the straw is used up some 150 kg of nitrogen is locked up with the 1200 kg of carbon in the micro-organisms. Since there was only 80 kg of nitrogen in the straw put on the land, the other 70 kg has been ‘robbed’ from the soil. This nitrogen is rendered unavailable to plants (‘locked up’) until the micro-organisms die and decompose. To ensure rapid decomposition or to prevent a detrimental effect on crops the addition of straw must be accompanied by the addition of nitrogen. Nitrogen is released during decomposition if the organic material has a C:N ratio narrower than 30:1, such as young plant material, or with nitrogen-supplemented plant material such as farmyard manure (FYM). |