Moist Heat
It is possible to quantitate the response of microorganisms to heat by measuring the time required to kill them at different temperatures. The lowest temperature required to sterilize a standardized pure culture of bacteria within a given time (usually 10 minutes) can be called the thermal death point of that species, and, conversely, the time required to sterilize the culture at a stated temperature can be established as the thermal death time.| Purpose | To demonstrate destruction of microorganisms by moist heat applied under controlled conditions of time and temperature |
| Materials | Tubed nutrient broths (5-ml aliquots) Nutrient agar plates Sterile 1.0-ml pipettes 24-hour broth culture of Staphylococcus epidermidis Six-day-old broth culture of Bacillus subtilis |
Procedures
- Set up a beaker water bath and heat to boiling.
- Divide one nutrient agar plate in half by marking the bottom of the plate with a wax pencil or ink marker.
- Streak a loopful of the S. epidermidis culture onto one-half of the nutrient agar plate. Label the section of the plate with the name of the organism and the word Control.
- Repeat step 3 with the culture of B. subtilis, inoculating the second half of the plate.
- Place the “control” plate in the 35°C incubator for 24 hours.
- Divide two nutrient agar plates into 4 quadrants by marking the bottom of the plates with a wax pencil or ink marker. Label one plate S. epidermidis and the other B. subtilis. Label the 4 quadrants on each plate as follows: 5, 10, 15, 30 minutes.
- Take a pair of broth tubes and inoculate each, respectively, with 0.1 ml of S. epidermidis and B. subtilis. Place these tubes in the boiling water bath. Note the time.
- Leave the pair of broth cultures in boiling water for 5 minutes. Remove the tubes and cool them quickly under running cold tap water. Streak a loopful of each boiled culture onto the quadrant of nutrient agar labeled 5 minutes.
- Return the tubes to the boiling water bath for an additional 5 minutes. Begin timing when the water comes to a full boil. Cool the tubes as in step 8 then streak a loopful of each culture onto the quadrant of nutrient agar labeled 10 minutes.
- Repeat step 9 twice more, streaking loopfuls of culture onto the quadrants of the plates labeled 15 and 30 minutes, respectively.
- Incubate subcultures from boiled tubes at 35°C for 24 hours.
Results
- Read all plates for growth (+) or no growth (−). Record your results in the chart.
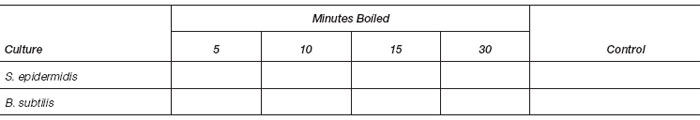
- State your interpretation of these results for each organism:
- S. epidermidis:
- B. subtilis:




