Polyhydroxybutyrate
PHB is the most widespread and thoroughly characterized PHA found in bacteria. A large part of our knowledge on PHB biosynthesis has been obtained from R. eutropha (Steinbü chel and Hein, 2001). In this bacterium, PHB is synthesized from acetyl-CoA by the sequential action of three enzymes (Fig. 8.4). The first enzyme of the pathway, 3-ketothiolase, encoded by the phbA gene, catalyzes the reversible condensation of two acetyl-CoA moieties to form acetoacetyl-CoA. Acetoacetyl-CoA reductase, encoded by the phbB gene, subsequently reduces acetoacetyl-CoA to R-(—)-3-hydroxybutyryl-CoA, which is then polymerized to PHB by the action of a PHA synthase encoded by the phaC gene. The PHA synthase of R. eutropha has been shown to accept the R-isomer of 3-hydroxybutyryl-CoA but not the S-isomer.PHAis typically produced as a polymer of 103 to 104monomers that accumulates as inclusions of 0.2 to 0.5 mmin diameter. In R. eutropha, PHB inclusions can typically accumulate to 80–85% of the dry weight (dwt) when bacteria are grown in media containing excess carbon, such as glucose, but limited in one essential nutrient, such as nitrogen or phosphate (Steinbüchel and Schlegel, 1991). Under these conditions, PHB synthesis acts as a carbon reserve and an electron sink.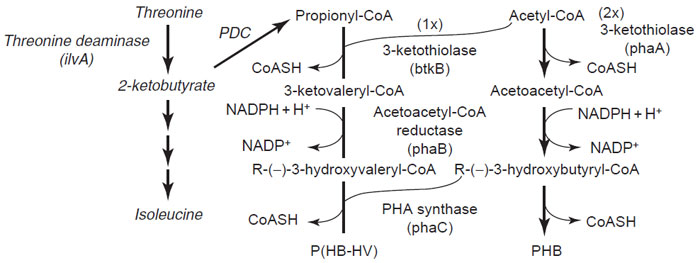 |
| FIGURE 8.4 Pathways of PHB and P(HB-HV) synthesis. The pathways common to bacteria and transgenic plants are shown in plain letters while the pathway specific to transgenic plants is shown in italics. PDC refers to the plant endogenous pyruvate dehydrogenase complex. |
PHB is a highly crystalline polymer and a stiff and relatively brittle thermoplastic (de Koning, 1995). Its melting point (Tm = 175 °C) is only slightly lower than the temperature at which it starts degrading to crotonic acid, making processing difficult. These properties seriously limit its use in a wide range of commodity products. PHB has good UV light resistance but relatively poor resistance to acids and bases. The polymer is water and air impermeable as well as relatively resistant to hydrolytic degradation, making it superior to starch-derived plastics, which are moisture sensitive.
Synthesis of PHB in the Cytoplasm
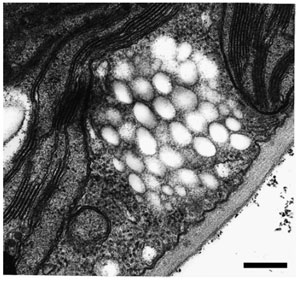 |
| FIGURE 8.5 Accumulation of PHA inclusions in the cytoplasm of transgenic A. thaliana cells expressing the PHB biosynthetic pathway. Bar = 1 µm. |
Despite its relatively poor physical properties as a thermoplastic, PHB was initially targeted for production in plants because the first bacterial PHA biosynthetic genes that were cloned were for PHB synthesis in the bacterium R. eutropha (Schubert et al., 1988; Slater et al., 1988). The cytoplasm was targeted as the first site for PHB synthesis because, in addition to containing acetyl-CoA, the building block for PHB, it also had the advantage that the bacterial enzymes could be directly expressed in this compartment without any modification of the proteins. Furthermore, an endogenous plant 3-ketothiolase is present in the cytoplasm as part of mevalonate pathway. Thus, creation of the PHB biosynthetic pathway in the cytoplasm was theoretically simpler, requiring only the expression of two additional enzymes, the reductase and synthase. The R. eutropha phaB and phbC genes, encoding, respectively, the acetoacetyl-CoA reductase and PHA synthase, were coexpressed in A. thaliana under the control of the cauliflower mosaic virus (CaMV) 35S promoter, allowing a relatively high expression of the enzymes in a broad range of tissues (Poirier et al., 1992a). The highest amount of PHB measured in the shoots of these plants was ~0.1% dwt (Poirier et al., 1992a). Detailed analysis of the PHB purified from A. thaliana confirmed that the polymer was isotactic poly ([R]-(—)-3-hydroxybutyrate) and that the thermal properties of plant PHB were similar to those of bacterial PHB (Poirier et al., 1995b). Furthermore, PHB accumulated in the form of granules that had a size and appearance very similar to bacterial PHB granules (Fig. 8.5) (Poirier et al., 1992a).
Plants expressing high level of acetoacetyl-CoA reductase in the cytoplasm have shown a strong reduction in growth, with the most affected plants being approximately five times smaller by fresh weight compared to wild-type plants (Poirier et al., 1992b). There was an overall good correlation between the extent of the growth reduction and the level of reductase enzyme activity. While no abnormal phenotype was observed in plants expressing only the PHB synthase (and not producing PHB), combination of the acetoacetyl-CoA reductase with the PHB synthase led to a further reduction in growth compared to plants expressing only the reductase (Poirier et al., 1992b). Although the reasons for the dwarf phenotype have not been unambiguously determined, it is likely that the diversion of cytoplasmic acetyl-CoA and acetoacetyl-CoA away from the endogenous isoprenoid and flavonoid pathways might lead to a depletion of essential metabolites, such as sterols, which may affect growth.
Synthesis of PHB in the cytoplasm of rape leaf cells gave results very similar to those in Arabidopsis (Poirier, 2002). Interestingly, overexpression of the bacterial 3-ketothiolase in plants expressing the reductase and PHB synthase did not lead to a significant increase in PHB accumulation, indicating that 3-ketothiolase activity was probably not limiting PHB synthesis in the cytoplasm, but that other factors, such as the low flux of acetyl-CoA, may be important.
PHB synthesis has also been demonstrated in the cytoplasm of cotton fiber cells (John and Keller, 1996). In this approach, PHA is not produced as a source of polyester to be extracted and used in the plastic industries, but rather as an intracellular agent that modifies the heat exchange properties of the fiber. The phaA, phaB, and phaC genes from R. eutropha were expressed in transgenic cotton under the control of a fiber-specific promoter (John and Keller, 1996). PHB accumulated in the cytoplasm to 0.3% dwt of the mature fiber, a level similar to PHB production in A. thaliana cell cytoplasm, while no deleterious effect on fiber development was reported.
Production of PHB has been reported in leaves of Nicotiana tabacum through the coexpression of the phaB gene from R. eutropha and the PHA synthase from Aeromonas caviae (Nakashita et al., 1999). Although the bacterial genes were expressed under the strong promoter CaMV35S, expression of both proteins was relatively low and the amount of PHB detected in leaves was only 10 µg/g fresh weight (fwt). Inhibition of the mevalonate pathway at the level of the 3-hydroxy-3-methylglutaryl-CoA reductase led to a twofold increase in PHB level in tobacco cell lines, indicating a link between PHB synthesis and availability of acetyl-CoA (Suzuki et al., 2002). Similar levels of PHB were obtained in potato expressing the phb enzymes in the cytosol.
Synthesis of PHB in the Plastid
The relatively limited supply of acetyl-CoA in the cytosol is thought to be responsible for the low accumulation of PHB as well as for the deleterious effects of transgene expression on plant growth observed in many plants. In this context, the plastid was viewed as a much better site for PHB synthesis, since this organelle has a larger flux of carbon through acetyl-CoA required for fatty acid biosynthesis. This is particularly true for the leucoplast of developing seeds of oil-accumulating plants, such as Arabidopsis and oilseed rape.
The phaA, phaB, and phaC proteins from R. eutropha were modified for plastid targeting by addition of the transit peptide of the small subunit of the ribulose bisphosphate carboxylase from pea (Nawrath et al., 1994). The modified bacterial genes were first expressed individually in A. thaliana under the control of the constitutive CaMV35S promoter, and later the transgenes were combined through crossings. Transgenic plants expressing only the plastid-targeted reductase and PHA synthase did not produce detectable PHB, providing further evidence that plastids do not have an endogenous 3-ketothiolase activity that could support PHB synthesis (Nawrath et al., 1994). However, plants expressing all three bacterial enzymes were shown to accumulate PHB inclusions exclusively in the plastids, with some organelle having a substantial portion of their volume filled with inclusions. The size and general appearance of these were similar to bacterial PHA inclusions (Nawrath et al., 1994). Interestingly, the quantity of PHB in these plants was found to gradually increase over time, with fully expanded presenescing leaves typically accumulating 10 times more PHB than do young expanding leaves of the same plant. The maximal amount of PHB detected in presenescing leaves was 10 mg/g fwt, representing ~14% dwt. In contrast to PHB synthesis in the cytoplasm, expression of the PHB biosynthetic enzymes in the plastid was not accompanied by a large reduction in growth of these plants. However, leaf chlorosis was observed in plants accumulating more than 3–5% dwt. These results indicated that although the plastid can accommodate a higher production of PHB with minimal impact on plant growth compared to the cytoplasm, there was nevertheless a limit above which alteration in some of the chloroplast functions could be detected (Nawrath et al., 1994).
In contrast to the individual expression of the R. eutropha phb genes in plants followed by stacking through crossing, an alternative strategy was devised where all three plastid-modified phb genes were cloned into a single binary vector. By this approach, a number of lines were identified which accumulated PHB between 3% and 40% dwt (Bohmert et al., 2000). While in a line accumulating 3% dwt most of the plastids contained some PHB inclusions, all plastids of mesophyll cells were packed with inclusion in the line containing PHB to 40% dwt. Interestingly, these transgenic plants showed a negative correlation between PHB accumulation and plant growth. While plants containing 3% dwt PHB showed only a relatively small reduction in growth, plants accumulating between 30% and 40% dwt PHB were dwarfed and produced no seeds (Bohmert et al., 2000). As previously observed by Nawrath and colleagues, all plants producing above 3% dwt PHB showed some chlorosis (Bohmert et al., 2000; Nawrath et al., 1994). Together, these experiments demonstrate that while it is possible to further increase PHB production in plastids by using new vectors, the approach of synthesizing PHB in the chloroplasts of shoots has its limits.
Since the production of PHA in the plastid typically requires the expression of several enzymes, strategies devised to simplify the number of individual genes that must be expressed could have advantages. In this respect, a novel fusion protein composed of the 3-ketothiolase and acetoacetyl-CoA reductase from R. eutropha was created (Kourtz et al., 2005). This was a challenging project since the native thiolase and reductase enzymes act as homotetramers in bacteria. Nevertheless, one fusion protein exhibited thiolase and reductase activities in crude extracts of recombinant Escherichia coli that were only threefold and ninefold less than those of the individually expressed thiolase and reductase enzymes, respectively. Expression of the plastid-targeted fusion enzyme, along with the PHA synthase, resulted in plants accumulating roughly half the amount of PHB synthesized in plants expressing the individual enzymes.
As a first step to bring the technology of PHA synthesis to the field, scientists at Monsanto have demonstrated the production of PHB in the plastids of corn leaves and stalk, as well as in the leucoplast of developing seeds of Brassica napus. In those experiments, the same R. eutropha genes modified for PHB production in the plastids of A. thaliana were used. Levels of PHB accumulation up to 5.7% dwt were reported (Poirier and Gruys, 2001). Similar to results obtained in A. thaliana, there was a progressive accumulation of PHB in corn shoots with time, with older leaves having more polymer than younger leaves. Furthermore, like in A. thaliana, there was a correlation between leaf chlorosis and higher amount of PHB (Poirier and Gruys, 2001). Perhaps one of the most striking observations made from the experiments in corn was the fact that while the leaf mesophyll cells showed few PHB granules, the bundle sheath cells associated with the vascular tissue were packed with granules (Poirier and Gruys, 2001). This unequal distribution of PHB was not due to the promoter used, since a similar pattern was seen for plants transformed with either the CaMV35S or the chlorophyll A/B binding protein promoters, the latter promoter being known to be a strong promoter in mesophyll cells. Interestingly, a similar observation had been made by the same group for A. thaliana plants transformed with the phb genes driven by the CaMV35S promoter; that is, significantly more granules were found in cells surrounding the vascular tissue and epidermal cells compared to mesophyll cells (Poirier and Gruys, 2001). These results suggest that the availability of plastidial acetyl-CoA for PHB synthesis may be quite different in various cell types, perhaps due to metabolic channeling.
For the creation of the PHB biosynthetic pathway in developing seeds of B. napus, the three modified bacterial genes phaA, phaB, and phaC were put under the control of the fatty acid hyroxylase promoter from Lesquerella fendeleri, enabling strong expression to the developing seed (Houmiel et al., 1999). PHB level up to 7.7% fwt of mature seeds was reported (Houmiel et al., 1999). Analysis of seeds by TEM revealed that PHB accumulated exclusively within the leucoplast and that apparently every visible plastid contained the polymer. Seeds accumulating nearly 8% dwt PHB appeared normal and germinated at the same rates as nontransformed seeds (Houmiel et al., 1999). These results demonstrate that at least in the range of 3–8% dwt PHB, the seed leucoplast appears a better production system than the leaf chloroplast. It is unknown at this point what is the upper limit of PHB accumulation in seeds and at what level PHB synthesis will start affecting the accumulation of lipids or proteins in the seed, two key factors that have a strong impact on the viability of this approach in the biotechnological production of PHA in oilseed crops.
Five additional crop plants have been investigated for PHA production through expression of the PHB pathway in the plastid. Transformation of alfalfa, tobacco, potato, and flax with the three R. eutropha phb genes modified for plastid targeting was shown to give transgenic plants producing PHB in their leaves to a maximum level of 0.18%, 0.32%, 0.009%, and 0.005% dwt (Bohmert et al., 2002; Saruul et al., 2002; Wróbel et al., 2004). Although the reasons behind the low level of PHB accumulation in these plants compared to either Arabidopsis or corn have not been fully elucidated, it has been demonstrated that constitutive expression of the bacterial 3-ketothiolase leads to a large decrease in the recovery of transgenic plants following transformation (Bohmert et al., 2002). The use of a construct where the bacterial 3-ketothiolase is expressed under the control of an inducible promoter led to an increased recovery of transgenic tobacco and potato producing PHB, although the amount of PHB produced remained relatively low at below 0.3% dwt (Bohmert et al., 2002). Transformation of in vitro cultured hairy roots of sugar beet with the same three R. eutropha genes modified for plastid targeting led to significantly higher amount of PHB, with a maximum of 5.5% dwt (Menzel et al., 2003). Thus, although accumulation of PHB in the plastid appears to be problematic for several plants, the success encountered with Arabidopsis, rape, corn, and roots of sugar beet indicate that there is no fundamental barrier to relatively high production of PHA in the plastids of plants.
As an alternative strategy to the transformation of the nuclear genome, transformation of the plastid genome with the phb gene has been examined. In theory, plastid transformation could lead to higher level of polymer production because of the much larger copy number of transplastome compared to the nuclear genome. However, transformation of tobacco plastome with the R. eutropha polycistronic operon containing the phbA, phbB, and phbC genes under the control of a bacterial promoter or of the plastid rRNA promoter (Prrn) has yielded plants synthesizing only low amount of PHB (<0.1% PHB dwt) (Arai et al., 2001, 2004; Nakashita et al., 2001). Expression of the R. eutropha polycistronic operon under the control of the plant psbA promoter and the psbA 5´ UTR improved PHB accumulation up to 1.7% dwt (Lö ssl et al., 2003). In these transgenic plants, a higher level of PHB was limited to the early stage of heterotrophic in vitro culture and decreased through autotrophic growth despite constant transcript levels. PHB amounts were also found to be highly variable in different tissues of the same plant. Furthermore, production of PHB in transplastomic tobacco was associated with growth retardation and male sterility (Lö ssl et al., 2003). Use of a transformation system where the plastidial polycistronic phb operon was under the control of an ethanol-inducible T7 RNA polymerase could solve the problem of growth retardation and sterility, but without further improvement in the yield of PHB (Lö ssl et al., 2005). Although further work is required to understand the factors limiting the stable production of PHB in transplastomic tobacco, it must be stressed that accumulation of PHA in tobacco and potato, either in the cytoplasm or in the plastid, has consistently been low compared to Arabidopsis or rape. In this context, it would be very interesting to know if the application of the transplastome approach to Arabidopsis and rape would give similar or higher amount of PHB compared to nuclear transformation.
PHB synthesized in plants is not thought to be degraded, since significant hydrolysis of PHA requires the presence of specialized bacterial enzymes, the PHA depolymerases (Jendrossek, 2002). PHA in plants is thus viewed as a final and largely unrecyclable carbon sink. This opens several interesting questions about how transgenic plants accumulating PHA can cope with a new carbon sink. For example, how does PHB synthesis in the plastids affect carbon flow to other compounds synthesized in the organelle, such as starch and fatty acids? How does the plant adjust, at the metabolic and genetic levels, to accommodate for the synthesis of this new sink? Why are plants producing high amount of PHB affected in their growth? Clearly, the tools of genomics, proteomics, and metabolic profiling could provide interesting answers to these questions and give general insights on plant biochemistry that would go well beyondPHAsynthesis in plants.
In a first small-scale study of metabolite profiling, over 60 metabolites were measured in transgenic A. thaliana lines producing high amount of PHB (Bohmert et al., 2000). Surprisingly, no changes in fatty acids were observed. There was, however, a correlation between an increase in PHB with a decrease in levels of isocitrate and fumarate, indicating a reduction in tricarboxylic acid cycle activity, leading perhaps to a reduction in pools of acetyl-CoA that may result in growth retardation. There was also a positive correlation between PHB accumulation and levels of several sugars such as mannitol, glucose, fructose, and sucrose. Together, these data indicate that a high amount of accumulation of PHB in chloroplasts has a negative and complex effect on plant metabolism that go beyond the chloroplast. At the gene expression level, no correlation could be found between level of expression of the three phb genes and PHB accumulation, leaving unresolved the question of what limits PHB synthesis in the plastids.
Synthesis of PHB in the Peroxisome
Acetyl-CoA is found not only in the cytoplasm and plastids but also in the mitochondria and peroxisomes, being primarily implicated in these organelles in the tricarboxylic acid and β-oxidation cycles, respectively. Although no conclusive demonstration of PHB in plant mitochondria has been reported, synthesis of PHB in the peroxisome was described in transgenic Black Mexican sweet corn suspension cell cultures (Hahn et al., 1999). In these experiments, the phaA, phaB, and phaC genes from R. eutropha were modified in order to add a peroxisomal targeting signal at the carboxy terminal end of each protein. Biolistic transformation of maize suspension culture with a mixture of all three genes led to the isolation of transformants expressing all three enzyme activities and accumulating PHB up to 2% dwt (Hahn et al., 1999). As no transgenic plants have been obtained from these transformed cells, it is difficult at this point to evaluate the potential effects of PHB synthesis in peroxisome on growth and metabolism.




