Separation and Expansion of Human T Cells
For the characterization of specific cell types and the investigation of their functions, it is essential that the cells can be purified. Cell separation techniques based on the use of antibody-coated magnetic beads, e.g., Dynabeads (Ugelstad et al., 1980, 1994), are now widely used in research and clinical laboratories. Specific cells can, after binding to the magnetic beads, be selected by the use of a magnet and, following brief washing, high cell purity can be achieved. This technique continues to encompass new fields for the selective isolation of eukaryotic cells (Funderud et al., 1987; Luxembourg et al., 1998; Marquez et al., 1998; Soltys et al., 1999; Chang et al., 2002). The use of pure cell populations has also reached the field of therapy. Ex vivo expansion and manipulation of isolated cells have given promising possibilities in therapy, especially in immunotherapy. Dynabeads, having approximately the same size as eukaryotic cells, have proven to be very efficient in the ex vivo activation of T cells, the prime effectors of the acquired immune system (Garlie et al., 1999; Lure et al., 2001). An ex vivoexpanded population of T cells may be administrated to the patient, thereby helping to fight diseases such as cancer, HIV, and autoimmune disorders (Liebowitz et al., 1998; Levine et al., 1998, 2002; Thomas and June, 2001).
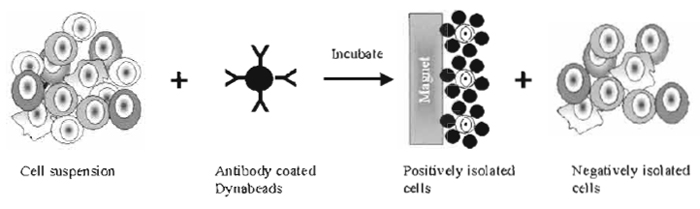 |
| FIGURE 1 Positive and negative isolation of cells using antibody-coated Dynabeads. By positive selection, a specific cellular subset is isolated directly from a complex mixture of cells based on the expression of a distinct surface antigen. The resulting immune complexes of beads and target cells are collected using a magnet. By negative selection, all unwanted cell types are removed from the sample by the magnetic beads. |
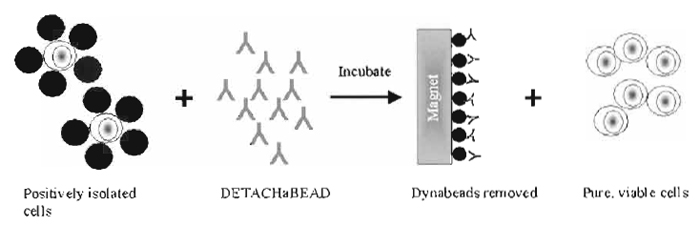 |
| FIGURE 2 Detachment of cells from Dynabeads using DETACHaBEAD. Cells are detached from the beads by a polyclonal antibody (DETACHaBEAD) that binds the Fab-region of the cell-specific monoclonal antibody, thereby altering its affinity for the antigen. The cells are left without antibody on their surface. |
Lymphoprep 6 × 500 ml (Prod. No. 1114547), density 1.077 ± 0.001g/ml, osmolality 290 ± 15mOsm from Axis-Shield Poc is stable for 3 years if stored at room temperature in the dark. Human serum, AB 100ml (Part Code US14-490E) from BioWhittaker is stable for 3 years if stored at -20°C. Human serum, AB should be heat inactivated (56°C, 30min) before use. All human blood-based products should be handled in accordance with currently acceptable biosafety practices and guidelines for the prevention of blood-borne viral infections. Fetal bovine serum, premium, US origin 500ml (Part Code US14-501F) from BioWhittaker is stable for 3 years if stored at -20°C. Fetal bovine serum should be heat inactivated (56°C, 30 min) before use. Bovine serum albumin (BSA) >96% by electrophoresis, 500g (Prod. No. A-4503) is from Sigma. DPBS, without Ca2+ or Mg2+ 500ml (Part Code BE17- 512F) from BioWhittaker is stable for 2 years if stored at 15-30°C. RPMI 1640, with L-glutamine 500ml (Part Code BE12-702F) from BioWhittaker is stable for 2 years if stored at 15-30°C. Proleukin interleukin-2 (recombinant IL-2) 22 × 106 IU from Chiron B.V. was reconstituted in sterile H2O at 6 × 105 IU/ml and stored at -20°C for up to 1 year. CD4-FITC 100 tests/2 ml (Prod. No. 3021) from Diatec is a mouse monoclonal IgG2a/κ, clone EDU-2 recommended for flow cytometry. CD4-FITC should be stored at 2-8°C (short term) or -20°C (long term). CD25-PE 100 tests (Cat. No. 341010) from BD Biosciences Pharmingen is a mouse monoclonal IgG1, clone 2A3 recommended for flow cytometry. CD25-PE should be stored at 2-8°C. CD8- PC5 100 tests/2ml (Part No. IM2638) from Beckman Coulter is a mouse monoclonal IgG1, clone B9.11 recommended for flow cytometry. CD8-PC5 should be stored at 2-8°C. Dynal CD4 positive isolation kit, 2 ml (Prod. No. 113.03), Dynabeads CD25, 2ml (Prod. No. 111.33), DETACHaBEAD CD4/CD8, 5ml (Prod. No. 125.04), Dynal CD4 negative isolation kit, 5 ml (Prod. No. 113.17), Dynal CD8 negative isolation kit, 5ml (Prod. No. 113.19), Dynabeads TcapTM EBV/BMLF-1 (Prod. No. 103.01), and Dynabeads CD3/CD28 T-cell expander, 2ml (Prod. No. 111.31) are from Dynal Biotech. These products, which contain magnetic beads (2.8-4.5µm) and/or antibody cocktails (mouse monoclonal antibodies) and/or release agents (polyclonal sheep antimouse antibodies), are stable for 12-36 months when stored at 2-8°C. Magnets: Dynal MPC (Prod. No. 120.01, 120.20, and 120.21) and from Dynal Biotech. Sodium citrate dihydrate pro analysis, >99% pure, l kg (Cat. No. 1.06448.1000) is produced by Merck and sold by VWR International. rHLAA2/ GLC-PE MHC tetramers 50 tests (code T2A-G) from ProImmune are stable for >6 months when stored at 2-8°C. Centrifuge: Rotanta 460 R from Hettich was delivered by Nerliens. Sample mixer: Dynal MX1 (Prod. No. 159.07) with 12-tube mixing wheel (Prod. No. 159.03) is from Dynal Biotech ASA. Flow cytometer: BD LSR II with 488- and 633-nm lasers from BD Biosciences was delivered by Laborel. Laminar flow bench: Biowizard Kojair KR 200 from Kojair Tech Oy was delivered by Houm AS. 37°C CO2 incubator: Forma Scientific Model 3548 (water-jacked incubator) was delivered by Houm AS. Pipettes: Finnpipette 0.5-10 µl, 5-50 µl, 20-200 µl, and 200-1000 µl from Thermo Labsystem were delivered by VWR International.
This article covers procedures for some basic principles of cell isolation and cell stimulation using Dynabeads.
The isolation of CD4+CD25+ regulatory T cells (Shevach, 2002) demonstrates the different isolation techniques. The isolation is performed in two steps; the first step involves isolation of CD4+ cells by positive selection (protocol A) or negative selection (protocol B). The second step involves isolation of CD25+ cells from the CD4+ cell population (protocol C).
Isolation of antigen-specific CD8+ T cells using recombinant HLA molecules on Dynabeads demonstrates enrichment of rare cells from a complex cell sample (protocol D). Expansion of these antigen-specific CD8+ T cells demonstrates ex vivo cell stimulation and manipulation (protocol E). For further questions, contact the supplier at: techcentre@dynalbiotech.com.
A. Positive Selection of CD4+ T Cells (Direct Technique)
CD4+ T cells are isolated from buffy coat by positive selection using the Dynal CD4 positive isolation kit. The isolated CD4+ cells are then ready for further studies, e.g., isolation of the CD4+CD25+ regulatory Tcell subpopulation (protocol C).
- Phosphate-buffered saline (PBS) pH 7.4: Dulbecco's PBS without Ca2+ and Mg2+
- PBS/citrate: PBS with 0.6% (w/v) sodium citrate (to prevent microcoagulation)
- Heat-inactivated fetal calf serum (FCS)
- PBS/BSA or PBS/FCS: PBS with 0.1% (w/v) bovine serum albumin or PBS with 2% (v/v) FCS.
- Culture medium: RPMI 1640 with 1% (v/v) FCS.
Steps for Cell Isolation
- Dynabeads required: 2 × 107 beads/ml × 40ml = 8× 108 beads (2.0 ml).
- Wash Dynabeads; mix vial, transfer beads to a 50-ml tube, add 10ml PBS/BSA, collect beads on a magnet for I min, remove supernatant, and replace with 2ml PBS/BSA; cool to 2-8°C.
- Dilute 15ml buffy coat in 25ml PBS/citrate; cool to 2-8°C.
- Add 40ml of cell suspension to the Dynabeads (2ml) directly into the 50-ml tube. Mix gently by tilting and rotation for 30min at 2-8°C.
- Isolate the cells that are attached to beads (rosetted cells) by placing the tube in the magnet for 2min. Discard the supernatant while the rosetted cells are held at the tube wall by the magnet.
- Wash cells; remove the tube from the magnet and resuspend the rosetted cells gently in 10ml PBS/citrate. Repeat steps 5 and 6 four to five times.
- Resuspend the rosetted cells in 1 ml of culture medium in a 5-ml tube. The cells are now ready for the removal of beads.
- Add 10 µl DETACHaBEAD per 107-108 beads used for cell capture.
- Mix gently by tilting and rotation for 45-60min at room temperature.
- Pipette gently five to six times to resuspend the cell/beads suspension and place the tube in a magnet for 2min.
- Collect the supernatant while the beads are held at the tube wall by the magnet.
- To obtain residual cells, resuspend the beads in culture medium and repeat steps 3 and 4 twice.
- Combine the supernatants, and wash the cells twice with PBS/BSA to remove DETACHaBEAD.
- The cells are now ready for further analysis or isolation of subpopulations (protocol C).
B. Negative Selection of CD4+ T Cells (Indirect Technique)
CD4+ T cells are isolated from peripheral blood mononuclear cell (PBMC) by negative selection using the Dynal CD4 negative isolation kit. Unwanted cells are removed, and the CD4+ T cells are then ready for further studies, e.g., isolation of the CD4+CD25+ regulatory T-cell subpopulation (protocol C).
Solution
PBS/citrate, PBS/BSA, FCS, and culture medium are prepared as in protocol A.
- Dynabeads required" 4 beads/PBMC × 108 PBMC = 4 × 108 beads (1.0ml).
- Wash Dynabeads as described in protocol A, resuspend in 1 ml.
- Isolate PBMC with low platelet content; add 35 ml solution (10ml buffy coat +25ml of PBS/citrate) on top of 15 ml of Lymphoprep at room temperature. Centrifuge for 20min at 160g at 20°C. Remove 20 ml of supernatant by suction to eliminate platelets. Centrifuge for 20min at 350g at 20°C. Recover PBMC from the plasma/Lymphoprep interface. Wash PBMC three times in PBS/BSA (centrifuge for 8min at 500g the first time and then at 225g) and resuspend the PBMC at 107 cells per 100-200 µl in PBS/BSA. Keep cells at 2-8°C.
- Incubate 108 PBMC with the 200-µl antibody mix and 200µl FCS for 10min at 2-8°C.
- Wash cells; add 5-10ml PBS/BSA and centrifuge for 8min at 500g at 2-8°C. Resuspend cells in 9ml PBS/BSA at room temperature.
- Add 9ml of PBMC to the depletion Dynabeads (1 ml) directly into the 50-ml tube. Mix gently by tilting and rotation for 15 min at room temperature.
- Pipette gently five to six times to resuspend the rosetted cells and double the sample volume with PBS/BSA. Place the tube in a magnet for 2min. Collect the supernatant containing CD4+ T cells. The cells are now ready for further analysis or isolation of subpopulations (protocol C).
C. Positive Selection of CD4+CD25+ Regulatory T Cells (Fig. 3)
CD4+ T cells have been isolated by either positive (protocol A) or negative (protocol B) selection. From these cells the CD4+CD25+ regulatory T-cell subpopulation can be isolated using Dynabeads CD25, and the beads are removed with DETACHaBEAD.
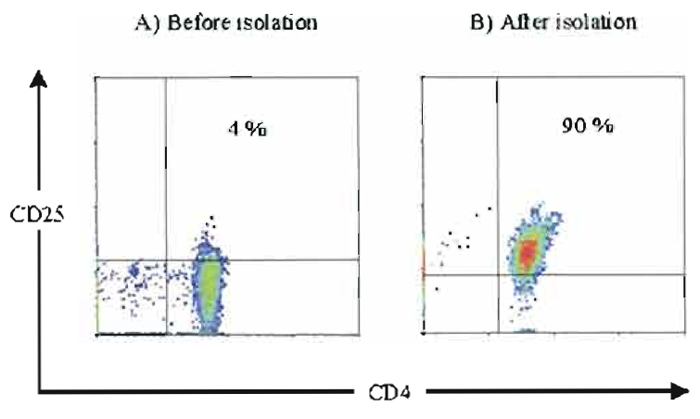 |
| FIGURE 3 Isolation of CD4+CD25+ regulatory T cells. CD4+CD25+ regulatory T cells were isolated according to protocol C. Diatec CD4-FITC and Pharmingen CD25-PE (clone 2A3) were used for cell staining. (A) Staining of CD25+ T-cells in the CD4+ T-cell population. (B) Staining of CD25+ T-cells after positive isolation from the CD4+ T-cell population. |
- PBS, PBS/BSA, FCS, and culture medium are prepared as in protocol A
- PBS/10% FCS: PBS with 10% (v/v) FCS
Steps for Cell Isolation
- Dynabeads required: 4 beads/CD4+ cell × 5 × 107 CD4+ cells = 2 × 108 beads (500 µl).
- Wash Dynabeads as described in protocol A, but in a 15-ml tube, resuspend in 500µl.
- Prepare cells by protocol A or B, resuspend at 107 cells/ml in PBS/BSA, and cool to 2-8°C.
- Add 5 ml of cells to the Dynabeads (500µl) directly into the 15-ml tube. Mix gently by tilting and rotation for 20rain at 2-8°C.
- Isolate and wash the rosetted cells as described in protocol A (steps 5 and 6), but use 2 ml PBS/BSA for washing.
- Use precoated tubes (10min at room temperature using PBS/10% FCS) from this step to avoid cell loss. Resuspend the rosetted cells in 200 µl of culture medium in a 1.5-ml tube. The cells are now ready for removal of beads.
- Add 10 µl DETACHaBEAD per 107 beads used for cell capture.
- Incubate, resuspend, collect, and wash cells as described in protocol A; use precoated tubes. The cells are now ready for further analysis and functional studies.
D. Isolation of Antigen-Specific CD8+ T Cells (Fig. 4)
Dynabeads coupled with recombinant HLA (rHLA) class I molecules loaded with the relevant peptide can be used to enrich antigen-specific CD8+ T cells (Garboczi et al., 1992; Luxembourg et al., 1998; Ostergaard Pedersen et al., 2001). The starting material is PBMC or preferably negatively isolated CD8+ T cells (protocol B, using Dynal CD8 negative isolation kit). Antigen-specific CD8+ T cells are isolated using Dynabeads TcapTM EBV/BMLF-1.
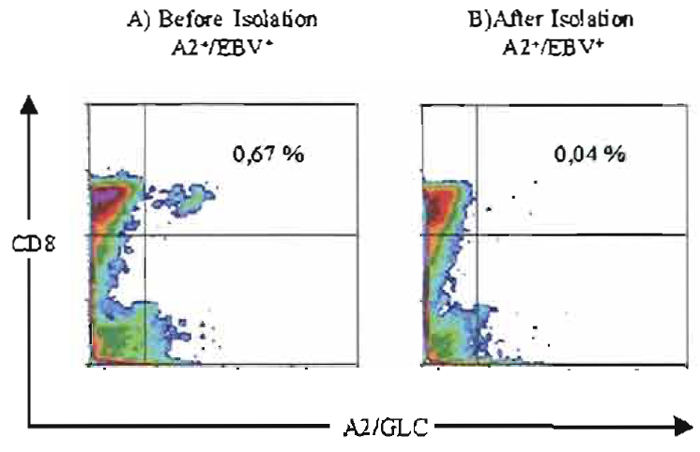 |
| FIGURE 4 Isolation of rHLA-A2/GLC positive T cells from PBMC. (A) PBMC from a HLA-A2/GLC positive donor stained with rHLA-A2/GLC tetramers. (B) PBMC after removal of rHLAA2/ GLC tetramer positive T cells by rHLA-A2/GLC coated Dynabeads, i.e., Dynabeads HLA-A2 EBV/BMLF-1 (see protocol D). The rHLA-A2 was produced and loaded with the peptide GLCTLVAML according to Ostergaard Pedersen et al. (2001). |
Solutions
- PBS/BSA and FCS are prepared as in protocol A.
- Culture medium: RPMI 1640 with 5% (v/v) FCS or 5% (v/v) AB serum.
- Dynabeads required: 107 beads/ml × 1 ml = 107 beads (25 µl).
- Wash Dynabeads; mix vial, transfer beads to a 1.5- ml tube, add 0.5ml PBS/BSA, collect beads on a magnet for 1 rain, remove supernatant, and replace with 100µl PBS/BSA; cool to 2-8°C.
- Prepare cells by protocol B (preferably negatively isolated CD8+ T cells), resuspend at 5 × 107 cells/ml in PBS/BSA, and cool at 2-8°C.
- Add 1 ml of cells to the Dynabeads directly into the 1.5-ml tube. Mix gently by tilting and rotation for 30min at 2-8°C.
- Isolate and wash the rosetted cells at 2-8°C as described in protocol A (steps 5 and 6), but use 1 ml PBS/BSA for washing. Resuspend the rosetted cells in 100µl of culture medium (2-8°C). The cells are now ready for further analysis or expansion (protocol E).
E. Activation and Expansion of T Cells Using Dynabeads
T-cell activation in vivo is initiated by the binding of T-cell receptors on its surface to appropriate peptide- HLA molecules on the surface of antigen-presenting cells (APC). However, activation of highly pure T cells in vitro is difficult to perform without the presence of APC. The Dynabeads CD3/CD28 T-cell expander can mimic the APC and therefore be used to activate pure T cells. Some T cells are CD28 negative and will require other costimulatory signals than through CD28, e.g., through CD137 (Maus et al., 2002).
- FCS is prepared as in protocol A.
- Culture medium: RPMI 1640 with 5% (v/v) FCS or 5% (v/v) AB serum
- Culture medium/rlL-2: culture medium with 20-100 IU/ml recombinant IL-2
Steps for in vitro Cell Expansion
- Dynabeads required: 4 beads/T cell × 107 cells = 4 × 107 beads (1.0ml).
- Starting material may be monocyte-depleted PBMC, pure CD3+ CD4+ or CD8+ T cells (e.g., prepared according to protocol A or B). Resuspend at 106 cells/ml in culture medium and add 1 ml of this cell suspension to each well in a 24-well culture plate. If rare cells are isolated and beads are still attached to the cells (e.g., protocol D), each well should contain a maximum of 106 beads (used for isolation), regardless of the number of cells isolated.
- Add the Dynabeads CD3/CD28 T-cell expander directly to the cells at a ratio of four beads per cell. Mix gently by pipetting.
- Incubate cells for 3 days at 37°C, resuspend, and count the cells. Dilute to 0.5 × 106 cells/ml with culture medium/rIL-2. Split cells every second to third day with culture medium/rIL-2 to keep the cell concentration at 0.5-1 × 106 cells/ml.
- Cells can be expanded for 14 days without adding extra beads, and cell expansion rates are usually between 100- and 1000-fold, depending on the donor.
Immunomagnetic cell isolation offers rapid and direct access to target cells from whole blood and bone marrow without cell loss or damage. If necessary, unwanted elements such as erythrocytes, free DNA, fat, or serum proteins may be removed before cell isolation to improve the performance of the beads. Buffy coat, a concentrate of white blood cells, has the advantage of high target cell concentration. However, the quality of buffy coat preparations may vary considerably. Density gradient isolation of cells provides removal of possible interfering elements and the ability to manipulate target cell concentration to perform cost-efficient cell isolation. Drawbacks include cell losses during centrifugations and negative effects on the cells due to contact with the density gradient medium. Generally the concentration of nucleated cells should be 108 cells/ml when performing immunomagnetic cell isolation.
By positive selection the cells of interest are isolated for analysis. General isolation parameters are ≥1 × 107 beads/ml, bead:cell ratio 4:1-10:1, and 10-30min of incubation. Typically, 95-100% purity and viability are achieved with 60-95% yield. For some downstream applications the beads can remain attached to the cells (e.g., mRNA or DNA isolation). By negative selection (= depletion), unwanted cells are removed prior to analysis of the remaining population. General isolation parameters are ≥2 × 107 beads/ml, bead:cell ratio ≥4:1, and 20-60min of incubation. Typically, 95-99% depletion of unwanted cells is achieved. Two successive depletion cycles may result in higher purity for small cell populations. The direct technique offers fast cell isolation with antibody-coated beads. Cell handling is minimized, reducing the risk for cell damage and loss. The indirect technique is especially useful when the affinity/avidity of the primary antibody is low or when the epitope density on the target cell is limited. The disadvantage of the indirect technique is cell handling (centrifugation).
Primary-coated Dynabeads are ready-to-use products for a wide variety of cell surface markers. In addition, secondary-coated Dynabeads offer an excellent possibility to make beads with the reactivity of choice using mouse, rat, or rabbit antibodies directly from culture supernatant, ascites, or polyclonal sera (without the need of purification). This is especially useful when only small amounts of nonpurified antibody are available. However, affinity-purified antibodies are preferred.
- Prolonged incubation (>60min) and increased bead concentration (>1 × 108 beads/ml)will rarely improve the cell selection efficiency. However, nonspecific binding may increase, damage of cells from sheer forces of the beads may occur, and risk of cell trapping increases.
- A soluble form of cell surface antigens or other serum components can reduce the efficiency of immunomagnetic cell isolation. One or two washing steps will overcome this problem.
- Nonspecific binding. Genomic DNA from lysed cells (e.g., present in buffy coat, PBMC, or after freezing/ thawing of cells) will induce non-specific binding of cells to beads. DNase treatment of the cell suspension prior to cell selection will prevent this problem without harming intact cells. Some sample tubes (e.g., glass or polystyrene) tend to bind cells nonspecifically, which can be a major problem when working with minor cell populations (e.g., rare, circulating tumour cells). Precoating of sample tubes with a protein solution before use or the use of low-binding plastic tubes is recommended.
- Phagocyte cells (e.g., monocytes) will bind and engulf beads if incubation is performed at temperatures above 2-8°C.
Chang, C. C., Ciubotariu, R., Manavalan, J. S., Yuan, J., Colovai, A. I., Piazza, E, Lederman, S., Colonna, M., Cortesini, R., Dalla- Favera, R., and Suciu-Foca, N. (2002). Tolerization of dendritic cells by T(S) cells: The crucial role of inhibitory receptors ILT3 and ILT4. Nature lmmunol. 3, 237-243.
Funderud, S., Nustad, K., Lea, T., Vartdal, E, Gaudernack, G., Stenstad, P., and Ugelstad, J. (1987). Fractionation of lymphocytes by immunomagnetic beads. In "Lymphocytes: A Practical Approach" (G. G. B. Klaus, ed.), pp. 55-65. IRL Press, Oxford.
Garboczi, D. N., Hung, D. T., and Wiley, D. C. (1992). HLA-A2- peptide complexes: Refolding and crystallization of molecules expressed in Escherichia coli and complexed with single antigenic peptides. Proc. Natl. Acad. Sci. USA 89, 3429-3433.
Garlie, N. K., LeFever, A. V., Siebenlist, R. E., Levine, B. L., June, C. H., and Lum, L. G. (1999). T cells coactivated with immobilized anti-CD3 and anti-CD28 as potential immunotherapy for cancer. J. Immunother. 22, 336-345.
Levine, B. L., Bernstein, W. B., Aronson, N. E., Schlienger, K., Cotte, J., Perfetto, S., Humphries, M. J., Ratto-Kim, S., Birx, D. L., Steffens, C., Landay, A., Carroll, R. G., and June, C. H. (2002). Adoptive transfer of costimulated CD4+ T cells induces expansion of peripheral T cells and decreased CCR5 expression in HIV infection. Nature Med. 8, 47-53.
Levine, B. L., Cotte, J., Small, C. C., Carroll, R. G., Riley, J. L., Bernstein, W. B., Van Epps, D. E., Hardwick, R. A., and June, C. H. (1998). Large-scale production of CD4+ T cells from HIV- 1-infected donors after CD3/CD28 costimulation. J. Hematother. 7, 437-448.
Lum, L. G., LeFever, A. V., Treisman, J. S., Garlie, N. K., and Hanson, J. P. Jr. (2001). Immune modulation in cancer patients after adoptive transfer of anti-CD3/anti-CD28-costimulated T cells-phase I clinical trial. J. Immunother. 24, 408-419.
Luxembourg, A. T., Borrow, P., Teyton, L., Brunmark, A. B., Peterson, P. A., and Jackson, M. R. (1998). Biomagnetic isolation of antigen-specific CD8+ T cells usable in immunotherapy. Nature Biotech. 16, 281-285.
Marquez, C., Trigueros, C., Franco, J. M., Ramiro, A. R., Carrasco, Y. R., Lopez-Botet, M., and Toribio, M. L. (1998). Identification of a common developmental pathway for thymic natural killer cells and dendritic cells. Blood 91, 2760-2771.
Maus, M. V., Thomas, A. K., Leonard, D. G., Allman, D., Addya, K., Schlienger, K., Riley, J. L., and June, C. H. (2002). Ex vivo expansion of polyclonal and antigen-specific cytotoxic T lymphocytes by artificial APCs expressing ligands for the T-cell receptor, CD28 and 4-1BB. Nature Biotechnol. 20, 143-148.
Rasmussen, A.-M., Smeland, E., Eriksten, B. K., Calgnault, L., and Funderud, S. (1992). A new method for detachment of Dynabeads from positively selected B lymphocytes. J. Immunol. Methods 146, 195-202.
Shevach, E. M. (2002). CD4+CD25+ suppressor T cells: More questions than answers. Nature Rev. 2, 389-400.
Soltys, J., Swain, S. D., Sipes, K. M., Nelson, L. K., Hanson, A. J., Kantele, J. M., Jutila M. A., and Quinn, M. T. (1999). Isolation of bovine neutrophils with biomagnetic beads: Comparison with standard Percoll density gradient isolation methods. J. Immunol. Methods 226, 71-84.
Thomas, A. K., and June, C. H. (2001). The promise of T-lymphocyte immunotherapy for the treatment of malignant disease. Cancer J. 7, S67-S75.
Ugelstad, J., Kilaas, L., Aune, O., Bjørgum, J., Herje, R., Schmid, R., Stenstad, P., and Berge, A. (1994). Monodisperse polymer particles. In "Advances of Biomagnetic Separation" (M. Uhlén, E. Hornes, and Ø. Olsvik, eds.), pp 1-20, Eaton Publ. Comp., Natick, MA.
Ugelstad, J., Mork, P. C., Herder Kaggerud, K., Ellingsen, T., and Berge, A. (1980). Swelling of oligomer particles: New methods of preparation of emulsions and polymer dispersions. Adv. Colloid Interface Sci. 13, 101.
Werther, K., Normark, M., Hansen, B. E, Brunner, N., and Nielsen H. J. (2000). The use of the CELLection kit in the isolation of carcinoma cells from mononuclear cell suspensions. J. Immunol. Methods 238, 133-141.




