New Opportunities and Approaches for Renewable Sources of Bioenergy, Biofuels, and Bioproducts?
While the extent to which lignin polymer composition and content can ultimately be modified and/or reduced is poorly understood, there are alternative biotechnological opportunities to produce renewable energy biofuels and specialty bioproducts, which have not been examined yet in detail. That is, there are other metabolic outcomes of the phenylpropanoid pathway from its entry point phenylalanine (1) (depending upon the species) that can include, for example, differential formation of coumarins, lignans, and flavonoids, as well as allyl/propenyl phenols (see Fig. 13.1). In particular, allylphenols and propenylphenols, which differ in the position of their side-chain double bonds, include the high-value liquids eugenol (33), estragole [(32), methylchavicol], and anethole (38) (Fig. 13.7). These natural products account for much of the aroma present in specialty ‘‘essential oils’’ of various plant species, such as cloves, tarragon, and anise, respectively, and are thought to be produced in planta mainly for defense against insects and parasites, as well as for attracting pollinators. In addition, most of these compounds are liquid at room temperature, and their relatively low degree of oxygenation grants them with high heats of combustion; these are potentially desirable characteristics when considering their possible utilization as fuels. Notably, lignins and most lignans, as well as the allyl/propenyl phenols, are all derived from the same monolignol precursors; thus an approach whereby the latter are differentially utilized could impact the production/accumulation of these diverse classes of compounds.Therefore, biotechnological manipulations of this pathway might be directed not only toward simply reducing lignin levels but also, perhaps, toward retargeting carbon toward related metabolic pathways, for example, through redirection of metabolic (carbon) flux to the production of related phenolic compounds in the main repositories for plant organic carbon
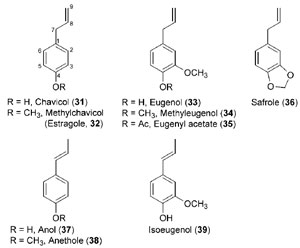 |
| FIGURE 13.7 Selected allylphenols and propenylphenols. |
storage. The latter could include either oilseed-bearing structures [e.g., flax (Linum usitatissimum) seed, Fig. 13.8A] (Ford et al., 2001; Teoh et al., 2003) or heartwood-forming tissues of trees [e.g., western red cedar (Thuja plicata), Fig. 13.8B] (Fujita et al., 1999; Kim et al., 2002). Indeed, it is these ‘‘repositories’’ that are largely used as plant renewable resources, whether as sources of (vegetable) oils or for lumber/pulp/paper products. In addition to the structural lignins, heartwood formation is often accompanied by massive deposition of non-structural low molecular weight molecules, such as the monolignol-derived lignan plicatic acid (30, see Fig. 13.1 for structure) and its congeners in western red cedar, whose amounts can be approximately 20% of the overall dry weight (Gardner et al., 1959, 1960, 1966). Rational optimization/modification of plant biomass could be done either directly for biofuel/bioenergy/bioproduct
 |
| FIGURE 13.8 (A) Flax (Linum usitatissimum) oilseed and (B) western red cedar (Thuja plicata) heartwood. (See Page 25 in Color Section.) |
generation in specific crops or indirectly as part of (heart)wood processing for pulp/paper, specialty chemicals, etc. Such considerations have not yet been explored, although the technologies are now available from previous research studies (Dinkova-Kostova et al., 1996; Fujita et al., 1999; Jiao et al., 1998; Kato et al., 1998; Teoh et al., 2003; Vassão et al., 2006b, 2007). For instance, recent studies have described the formation of some quite well-known phenylpropanoid pathway monomeric metabolites, namely, the liquid allyl/propenyl phenols, chavicol (31), eugenol (33), and their analogues (32, 34–39) (Fig. 13.7) (Koeduka et al., 2006; Vassão et al., 2006b).
Historically, such allyl/propenyl phenols have commonly been used throughout the world mainly as flavor/fragrance components present in spices, especially cloves, with these being largely imported from Tanzania, Madagascar, and Indonesia. Such material sources are imported simply because it is in these countries that the plant species accumulating these more unusual metabolites are cultivated. The recently described biochemical/biotechnological processes (Koeduka et al., 2006; Vassão et al., 2006a,b, 2007) thus offer yet another possibility of diversion of monolignols from either lignin and/or lignan formation in more commonly utilized woody/nonwoody plant species of, for example, North America, to afford instead the liquid allyl/propenyl phenol monomers.




