Proton Transport: Mechanism of Photosynthetic Phosphorylation
The interplay of PSI and PSII leads to the transfer of electrons from H
2O to NADPH, and the concomitant generation of proton gradient across the thylakoid membrane for ATP synthesis. The thylakoid space becomes markedly acidic with pH approaching 4. The light-induced transmembrane proton gradient is about 3.5 pH units. The generation of these protons follows two routes:
- For the splitting of two water molecules and the release of one oxygen molecule four proton are released in the thylakoid space.
- The transport of four electrons through the cytochrome b6f complex leads to the translocation of eight protons from the stroma to the thylakoid space.
Therefore about 12 protons for each O
2 molecule released are translocated. The proton-motive force Δp, that is, the force created by the accumulation of hydrogen ions on one side of the thylakoid membrane, consists of a pH gradient contribution and a membrane-potential contribution. In chloroplasts, nearly all of Δp arises from the pH gradient, whereas in the mitochondria the contribution from the membrane potential is larger. This difference is due to the thylakoid membrane permeability to Cl
- and Mg
2+. The light-induced transfer of H
+ into the thylakoid space is accompanied by the transfer of either Cl
- in the same direction or Mg
2+ in the opposite direction (1 per 2H
+). Consequently, electrical neutrality is maintained and no membrane potential is generated. A pH of 3.5 units across the thylakoid membrane corresponds to Δp of 0.22 V or a ΔG (change in Gibbs free energy) of -4.8 kcal/mol. The change in Gibbs free energy associated with a chemical reaction is a useful indicator of whether the reaction will proceed spontaneously. This energy is called free energy because it is the energy that will be released or freed up to do work.
As the change in free energy is equal to the maximum useful work which can be accomplished by the reaction, then a negative ΔG associated with a reaction indicates that it can happen spontaneously. About three protons flow through the F
0F
1-ATPase complex per ATP synthesized, which corresponds to a free energy input of 14.4 kcal/mol of ATP, but in which only 7.3 kcal
are stored in the ATP molecule, a yield of about 50%. No ATP is synthesized if the pH gradient is less than two units because the gradient force is then too small. The newly synthesized ATP is released into the stromal space. Likewise, NADPH formed by PSI is released into the stromal space. Thus ATP and NADPH, the products of light reactions of photosynthesis, are appropriately positioned for the subsequent light-independent reactions, in which CO
2 is converted into carbohydrates. The overall reaction can be expressed as:
This equation implies that each H
2O is split in the thylakoids under the influence of the light to give off 1/2O
2 molecule, and that the two electrons so freed are then transferred to two molecules of NADP
+, along with H
+s, to produce the strong reducing agent NADPH. Two molecules of ATP can be simultaneously formed from two ADP and two inorganic phosphates (Pi) so that the energy is stored in high energy compounds. NADPH and ATP are the assimilatory power required to reduce CO
2 to carbohydrates in the light-independent phase. The generation of ATP following this route is termed non-cyclic phosphorylation because electrons are just transported from water to NADP
+ and do not come back.
An alternative pathway for ATP production is cyclic phosphorylation, in which electrons from PSI cycle in a closed system through the phosphorylation sites and ATP is the only product formed. Electron arising from P
700 are transferred to ferredoxin and then to the cytochrome
b6f complex. Protons are pumped by this complex as electrons return to the oxidized form of reaction center P
700 through plastocyanine. This cyclic phosphorylation takes place when NADP is unavailable to accept electrons from reduced ferredoxin because of a very high ratio of NADPH to NADP
+. The electrochemical potential of the proton gradient drives the synthesis of ATP through an ATP-synthase situated, as we have seen, anisotropically in the thylakoid membrane.
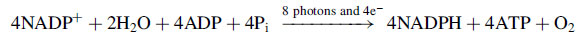 (3:3)
(3:3)



