Requirements for In Vitro Cultures
For the tissue culture a good laboratory, similar to a microbiological laboratory, is required which must have facilities for: (i) nutrient medium preparation, sterilization, cleaning and storage of supplies, (ii) aseptic condition for working the living materials, (iii) a controlled environmental conditions for growth and development of cultures, (iv) observation and evaluation of culture as hoped, and (v) recording the observations made during the experiment (White, 1963).
Nutrient media could be prepared in a separate room where sufficient space is available for keeping and weighing the chemicals, and putting glassware. Vitamins and growth hormones are carefully weighed. Stock solutions of chemicals are kept in refrigerator to avoid contamination. For sterilization of glassware, working tables, nutrient media and plant materials different techniques are applied. Most common methods of sterilization are:
- Use of Chemicals : For example, chromic acid, mercuric chloride (0.1%), sodium hypochlorite or calcium hypochlorite (each 0.5%), alcohol (70%), etc. are used for sterilization of glassware, working table and living materials.
- Use of Dry Heat: Glassware and metal instruments are kept in oven. Sterilization is done by keeping high temperature between 200-300°C for 1h.
- Use of Wet heat or Steam: Distilled wafer, nutrient media, etc.are sterilized in an autoclave at 121°C for 30 min.
- Ultrafiltration : Only those chemicals e.g. vitamins, growth hormones, etc., which are unstable at high temperature are sterilized by Seitz filter, milipore membrane filter, etc.
- By Ultraviolet Light: It is used in the inoculation chamber or laminar flow (Fig. 8.1).
- Using Antibiotics: These are used only in case of animal cell culture, not for plant cells. Inoculation (transfer of sterilized biological material on sterile nutrient medium in an aseptic condition) of plant or animal material is done in an inoculation chamber/laminar flow where ultraviolet lamp, 70% ethyl alcohol, spirit lamp, inoculation needle, glassware, etc. are kept in advance.
The glassware (culture tubes or Erlenmeyer conical flasks) containing sterilized nutrient medium after inoculation with plant material, are plugged with non-absorbent sterile cotton, and finally kept in growth chamber. Environmental conditions such as temperature, moisture and light are controlled as needed. For cell suspension culture, orbital shaker or aeration instrument is placed in growth chamber. Generally temperature is set between 25--27°C. Environmental conditions may vary according to plant species and nature of experiment. Light intensity, quality and photoperiod (light and dark cycles) are also regulated. Other laboratory accessories are dissection microscope, compound microscope, centrifuge, first aid kit, etc. which are required during in vitro culture of plant material.
Nutrient Media : Composition and Preparation
Vital activity of a cell is the absorption of nutrients through cell membrance and rapid proliferation into innumerable cells. White (1934) observed the unlimited growth of isolated root tissues when provided with nutrient medium containing inorganic salts, sucrose, vitamins, growth hormone and a few amino acids. Composition of different nutrient media is given in Table 8.1.
Inorganic nutrients include macronutrients (e.g. nitrogen, phosphorus, potassium, calcium, magnesium and sulphur) in the form of salts in large amount and microelements (e.g. boron, molybdenum, copper, zinc, manganese, iron and chloride). A concentrated stock solution is prepared in advance and finally added to medium as required. To overcome the problem of solubility, the stock solution of iron is prepared in a chelated form as the sodium salt of ferric ethylenediamme tetra acetate (Fe-EDTA).
Growth Hormones
Several growth hormones are known which stimulate the biological activity in cultured materials. Table 8.1. Composition of nutrient media required for in vitro culture.
Chemicalconstituents |
MS medium *(mg/1) | ER medium*(mg/1) | B5 medium*(mg/1) | |
| 1. | Macronutrients | |||
| NH4NO3 | 1650 | 1200 | - | |
| KNO3 | 1900 | 1900 | 2500 | |
| CaCl2 2H2O | 440 | 440 | 150 | |
| MgSO4 .6H2O | 370 | 370 | 250 | |
| KH2PO4 | 170 | 340 | - | |
| (NH4)2 S04 | - | - | 134 | |
| NaH2PO4 | - | - | 150 | |
| 2. | Iron | |||
| Na2EDTA | 37.3 | 37.3 | 37.3 | |
| FeSO4. 7H2O | 27.8 | 27.8 | 27.8 | |
| 3. | Micronutrients | |||
| MnS04.4H2O | 22.3 | 2.3 | - | |
| MnSO4.H2O | - | - | 10.0 | |
| ZnSO4.4H2O | 8.6 | - | 2.0 | |
| Zn versanate | - | 15 | 2.0 | |
| Na2MO42H2O | 0.25 | 0.025 | 0.25 | |
| CuSO4.5H2O | 0.025 | 0.0025 | 0.025 | |
| CoCl2.6H2O | 0.025 | 0.0025 | 0.025 | |
| KI | 0.83 | - | 0.75 | |
| H3BO3 . | 6.2 | 0.63 | 3.0 | |
| 4. | Vitamins | |||
| Glycine | 2.0 | 2.0 | ||
| Nicotinic acid | 0.5 | 05 | 1.0 | |
| Pyrodoxine -HCL | 0.5 | 05 | 1.0 | |
| Thiamine - HCL | 0.1 | 0.5 | 10.0 | |
| 5. | Cytokinin | |||
| Kinetin | 0.04-10.0 | 0.02 | 0,1 | |
| Myo-inositol | 100.0 | - | 100.0 | |
| IAA | 1.0-30.0 | - | - | |
| NAA | - | 1.0 | - | |
| 2,4-D | - | - | 0.1-1.0 | |
| Sucrose (g) | 30.0 | 40.0 | 20.0 | |
| pH | 5.7 | 5.8 | 5.5 | |
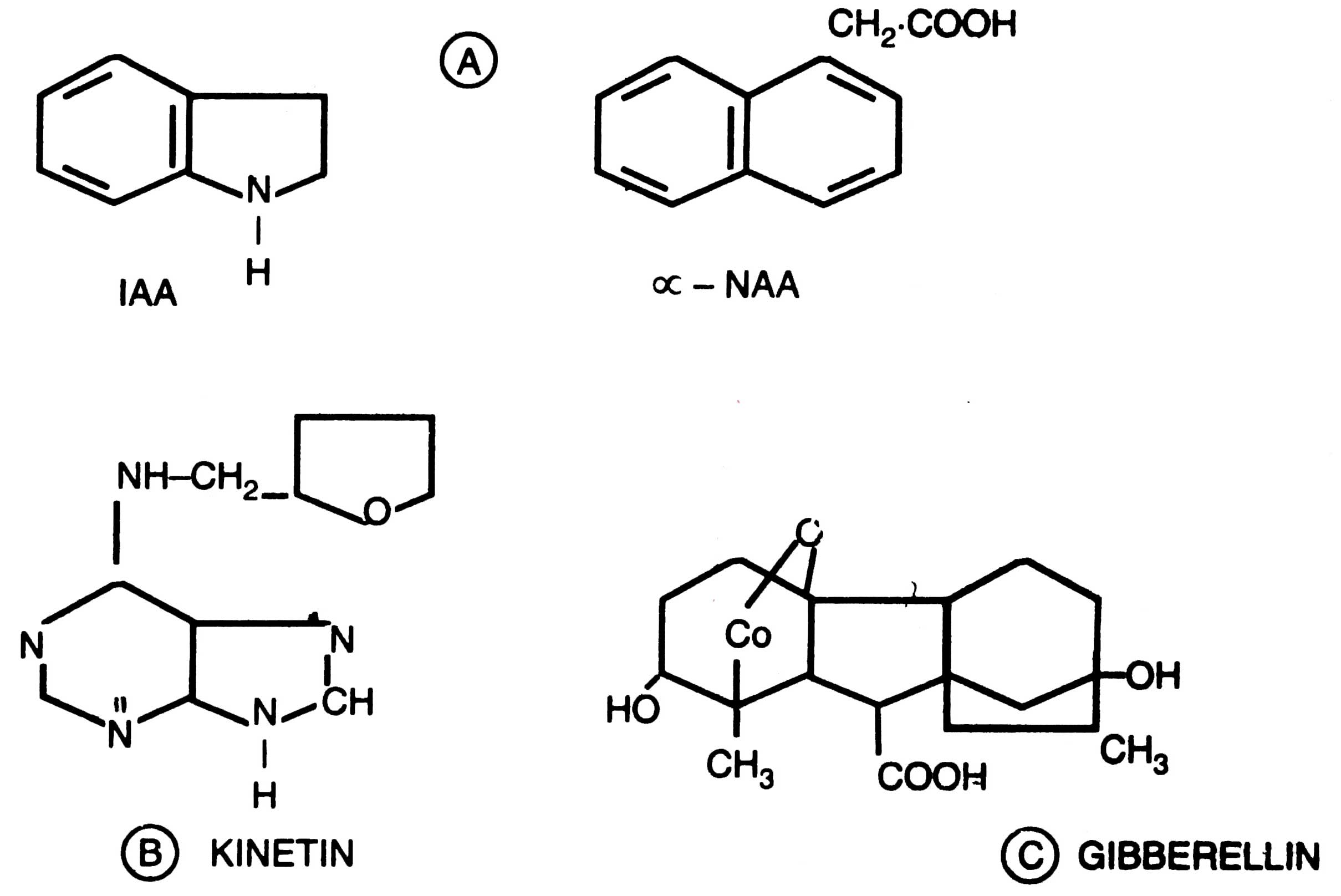
Cytokinins promote cell division and regulate growth and development similar to kinetin (6-furfuryl aminopurine). Auxin resembles indole acetic acid (IAA) and stimulates shoot elongation. Gibberellins are of less importance, however, GA3 is used in apical meristem (Morel and Muller, 1964). The most widely used cytokinins are adenin, kinetin, zeatin, benzyladenin; and auxins are IAA, NAA (a -Naphthalene acetic acid) 2,4-D. For the induction of callus the amount of kinetin should be 0.1 mg/liter. Structural formulae of cytokinins and auxins are given in Fig. 8.2.
Organic Constituents
The organic compounds serve a source of carbon and energy. They are used in high concentration, e.g. 20-30g/liter. Sucrose and D-glucose (carbohydrates) are commonly used; but glycerol and myoinosital are also the principal source of carbon. Other complex organic compounds are peptone, yeast extract, malt extract, coconut water, tomato juice, etc.
Vitamins
Vitamins are required in trace amount as they catalyze the enzyme system of the cells. Vitamin B{ (thiamine) is the most commonly used vitamin for all plant tissue cultures. Other groups of vitamins which stimulate growth are niacin (nicotinic acid) vitamin B2 (riboflavin) vitamin B6 (pyrodoxin) (Gamborg et al., 1976), vitamin C (ascorbic acid), vitamin H (biotin) and vitamin B12 (cyanocobalamin) (Huang and Murashige, 1977).
Although nitrogen sources are present in the inorganic salts, yet various amino acids and amides are used in plant tissue culture media as given by Huang and Murashige (1977). The most widely used amino acids are L-aspartic acid, L-asparagin, L-glutamic acid, L-glutamine and L-arginin.
A variety of media have been formulated by many workers, however, the composition of most commonly used nutrient media are given in Table 8.1.