Transfilter Cell Invasion Assays
The ability to invade surrounding extracellular matrices and tissues is an important phenotype of malignant tumor cells. To determine the invasive properties of malignant cells, several invasion assays have been developed. For example, organ fragments (Nicolson et al., 1985), reconstituted tissue spheroids (Mareel et al., 1988), membranous tissues (Nabeshima et al., 1988; Yagel et al., 1989), cultured cell monolayers (Kramer and Nicolson, 1979; Waller et al., 1986), or extracellular matrices (Albini et al., 1987; Schor et al., 1980) have been used as tissue or matrix for cell invasion studies. Commercially available Matrigel, a mouse EHS tumor extract consisting of major basement membrane components polymerized into a gel, has been the most commonly used material to determine the invasiveness of various types of cells (Albini et al., 1987). The apparatus of choice for measuring cell invasion has been a modified Boyden chamber or Transwell. Briefly, the apparatus contains two chambers (upper and lower) that are separated by a microporous polycarbonate filter, the upper surface of which is coated with a thin layer of Matrigel. Tumor cells are placed into the upper chamber where they settle by gravity onto the Matrigel layer. In the lower chamber a chemotactic agent can be placed to stimulate directional cell migration of cells that invade the Matrigel layer. The invasive ability of these cells can be expressed as the number of cells invading through the Matrigel layer and filter with time. For the most part, the invading cells are found on the lower surface of the filter and are not released into the fluid of the lower chamber. Although there are some exceptions to this (Simon et al., 1992), the invasive abilities of tumor cells in the transfilter invasion assay are usually related to their in vivo invasion behavior (Albini et al., 1987; Repesh, 1989; Hendrix et al., 1987). This article describes the basic protocol of the Matrigel transfilter invasion assay.
Culture medium DME/FI2 (Cat. No. 11330-032), fetal bovine serum (FBS), phosphate-buffered saline (PBS) (Cat. No. 14040-133), and trypsin (2.5%, Cat No. 15090-046) are from GIBCO-BRL. Matrigel (Cat. No. 40234) and fibronectin (Cat. No. 40008) are from Collaborative Biomedical Products. Bovine serum albumin (BSA) (Cat. No. 810661, fraction V) is from ICN. EDTA (Cat. No. 423-384) is from CMS. Hematoxylin (Cat. No. GHS-1-16), eosin (Cat. No. HTIIO-3- 16), and 10% neutralized formaldehyde solution (Cat. No. HT50-1-128) are from Sigma.
Transwells (Cat. No. 3421, 6.5-mm diameter, 5-µm pore size) and 24-well culture plates (Cat. No. 25820) are from Corning Costar Corporation. Additional equipment includes general tissue culture supply and equipment, forceps, cotton swabs, a reticle (1/10-mm measurement), and a cell-counting device (Coulter counter, hemocytometer, or equivalent equipment).
III. PROCEDURES
A. Matrigel Coating on the Microporous Transwell Filter
Solutions
1:30 diluted Matrigel solution (sterile): To make 900 µl Matrigel solution for 12 Transwells (50 µl X the number of Transwells), thaw Matrigel at 4°C overnight and mix 30µl with 870µl of ice-cold PBS. Keep ice cold until the solution is applied to the Transwell. Aliquot the remaining Matrigel (e.g., 1 ml each), freeze, and store at -80°C.
1:100 diluted Matrigel solution (sterile): To make 2 ml, mix 20µl of Matrigel with 2ml of ice-cold PBS. Keep solution ice-cold.
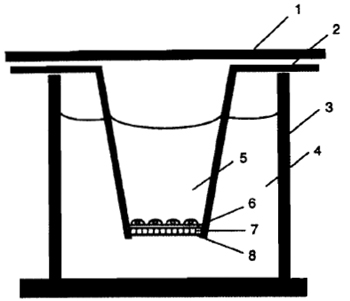 |
| FIGURE 1 Transwell invasion chamber system: (1) lid, (2) Transwell, (3) 24-well culture plate, (4) lower chamber (800µl of 10µg/ml fibronectin in invasion buffer), (5) upper chamber (200µl), (6) Matrigel layer, (7) microporous polycarbonate filter (5-µm pore size), and (8) lower surface of the filter coated with a thin layer of Matrigel. |
Steps
- Place 500 µl of 1: 100 diluted Matrigel solution in a small culture dish (i.e., 30mm diameter) on an icecold plate.
- Use a forceps for the handling of Transwells. The Transwell invasion chamber system is shown in Fig. 1. Soak the lower surface of the polycarbonate filter of each Transwell in the Matrigel solution. After briefly removing any excessive amount of the solution, place the Transwell into a 24-well culture plate and allow it to dry in a hood overnight at room temperature.
- Pour 50µl of the 1:30 diluted Matrigel solution into the upper chamber of the Transwell.
- Carefully overlay 200µl of sterilized, doubledistilled H2O to each filter and allow it to dry completely in a hood at room temperature under occasional ultraviolet light. This usually takes about 2 or 3 days.
- Proceed to the next step or store the coated Transwell sets in a scaled 24-well culture plate at 4°C.
B. Setting up the Invasion Assay
Solutions
2% BSA stock solution: To make 100ml, slowly dissolve 2 g of BSA in ice-cold PBS for 20min and sterilize by filtration through a 0.22-µm filter.
Invasion buffer, 0.1% BSA in DME/F12: To make 40ml, mix 2ml of 2% BSA stock solution with 38 ml of DME/FI2 medium.
EDTA stock solution (200 mM): To make 100 ml, dissolve 7.44g EDTA with double-distilled H2O and adjust the pH to 7.4.
Trypsin-EDTA (0.25%): To make 100 ml, add 10ml of 2.5% trypsin solution and 1 ml of EDTA stock solution into 89ml of PBS.
10% FBS-DME: To make 20 ml, mix 2 ml of FBS with 18 ml of DME/FI2 medium.
Fibronectin solution (10µg/ml): To make 10ml, mix 100µl of fibronectin stock solution (1 mg/ml) with 10ml of invasion buffer. In the example here, fibronectin is used as a chemoattractant.
- These procedures should be performed under sterile conditions. Culture (4 × 100-mm-diameter dishes) B16-F10 cells in DME/FI2 medium supplemented with 5% FBS at 37°C in an atmosphere of 5% CO2-95% air and grow to about 80% confluency.
- Prewarm the Matrigel-coated Transwells in the 24-well culture plate to room temperature, and rehydrate the Matrigel with 200 µl of invasion buffer for 1 h.
- Wash the B16-F10 culture dishes with 10 ml of DME/FI2 medium and incubate with 3ml each of trypsin-EDTA for 5 min. Place in a 50-ml tube and sediment cells by centifugation at 1000rpm for 5 min.
- Wash the cells with 10ml of 10% FBS-DME medium by centrifugation at 1000rpm for 5 min and resuspend the cells in 10 ml of 10% FBS-DME medium. Let the cells incubate for 15 min at room temperature.
- Wash the cells with 40ml of DME/FI2 medium twice by centrifugation at 1000rpm for 10min. Resuspend the cells in 5 ml of invasion buffer.
- Prepare 2.5 × 105 cells/ml in invasion buffer by counting the cells with a Coulter counter, hemocytometer, or equivalent equipment.
- Carefully discard the buffer in the upper chambers of the Transwells, wash the chambers again with 200 µl of invasion buffer, and carefully pour 800 µl of the fibronectin solution (chemoattractant solution) through the slit of the Transwell into the lower chamber.
- Add 200µl of the cell suspension into the upper chamber. Avoid making bubbles in the upper chamber so that the cells can settle down evenly on the filter. Carefully put the Transwell invasion chamber system into an incubator and incubate for 48h at 37°C in 5% CO2-95% air.
Steps
- Prepare a 24-well culture plate with 400 µl of 10% formaldehyde, hematoxylin, or eosin in each well and fill four jars (about 500ml each) with distilled water.
- Place the Transwell in the 10% formadehyde solution for 10min and then rinse the Transwell by submerging it in a jar containing distilled water.
- Transfer the Transwell to the hematoxylin solution for 10min and then rinse with water. Place the Transwell into the well with warm distilled water (about 40°C) for 10 min.
- Transfer the Transwell to the eosin well for 5 min and then place it into a jar containing distilled water. Gently rub the cells off the upper side of the filter using a cotton swab and rinse the Transwell with distilled water.
D. Counting Invading Cells Using a Phase- Contrast Microscope (or Coulter Counter)
Steps 1. Place the Transwell into a 24-well culture place. Make sure that there is moisture on the inside of the culture plate. Count the cells in the 16 fields indicated in Fig. 2 at 200 × magnification. 2. Using a reticle with 1/10-mm guides (Bausch & Comb Inc.), place the cell suspension onto the stage of a phase-contrast microscope, and measure the diameter of the field (r; mm) with the magnification at 40×. The total number of invading cells (1) can be calculated with the formula
| I = c × (5 × R/r)2, |
where c is the average number of invading cells per ×200 magnification field and R is the diameter of the filter (6.5 mm). For example, the value of r of a Diaphot (Nikon) is 4.45. Therefore, (5 × R/r)2 = 53.3. If the cells are too dense to count the entire field, a photo as shown in Fig. 2 can be used. Using a Diaphot (Nikon), the areas of a, b, and c in Fig. 2 are 47, 17, and 4.3% of the total area of the field, respectively.
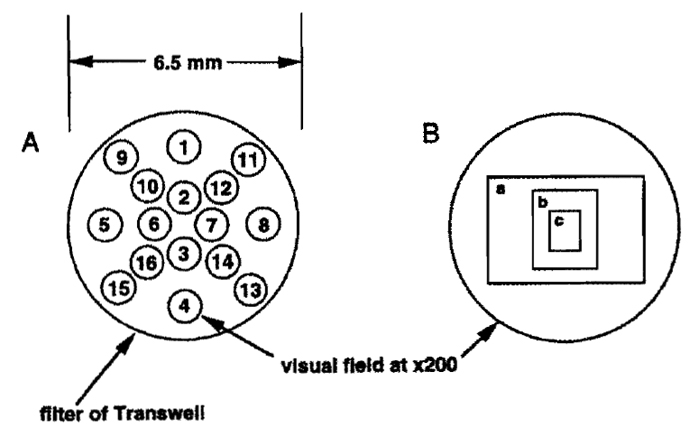 |
| FIGURE 2 A Transwell filter in a microscopic field. (A) Spots used to count the cells on the filter are shown as numbered circles. The numbers of the spots are an example of the sequence recommended for cell counting. (B) Photo frames of the Diaphot (Nikon). Relative sizes of the frames to the field are 47 (a), 17 (b), and 4.3% (c). |
Steps
- After counting invading cells, wipe the cells off the lower surface of the filter using a cotton swab and soak the Transwells in a detergent solution (e.g., 1% Contrad 70) for at least 1 day.
- Wipe both surfaces of the filter gently and thoroughly using a cotton swab and place the Transwells into the 24-well culture plate.
- Fill the entire Transwell plate with distilled water, cover the plate with a lid, shake it several times, and discard the water.
- Repeat step 3 at least three times. After the final wash, let the Transwell plate sit for 1 h filled with distilled water.
- Wash the plate with double-distilled water several times and rinse with 70% ethanol as in step 3. Dry the Transwells in a tissue culture hood overnight under ultraviolet light.
IV. COMMENTS
Although tumor cells in vivo usually invade connective or parenchymal tissue, a number of reports indicate that the invasive and metastatic phenotypes of cells correlate with their in vitro Matrigel invasion capacity (Albini et al., 1987; Repesh, 1989; Hen&ix et al., 1987). This may be due to the fact that invasion in vivo into connective or parenchymal tissue is limited by the capability of tumor cells to undergo basement membrane invasion, a key event in malignant progression. The invasive process involves at least three events (adhesion, degradation, and migration) (Liotta, 1986); for tumor cells to be invasive, they must be at least somewhat proficient in each step (Nicolson, 1989).
The incubation time for the invasion assay is usually 24 to 72h. We feel that shorter incubation times are better as long as a significant number of cells invade the filter. Using longer incubation times, other parameters, such as the growth of the cells on the Matrigel, loss of cell viability, or the detachment of cells from the underside of the filter, must be taken into account. The incubation time can be varied by changing the amount of Matrigel used in coating the filter, usually in the range of 5 to 50µg (or 10µg in our case).
The very thin Matrigel coating on the lower surface of the filter should be completely dried before adding the 1:30 Matrigel solution in the upper chamber. This will avoid leaks in the filter.
The overlay of 200 µl of double-distilled H2O on the Matrigel solution is necessary for an even coating of Matrigel on the filer.
The number of cells applied onto the filter of the Transwell should not exceed the number that will yield 100% confluency on the filter. Usually the cells are 50-80% confluent on the filter after settling.
The cell suspension volume in the upper chamber should be 200µl instead of 100µl to reduce the meniscus effect that can cause uneven settling of the cells onto the Matrigel layer. Thus the volume of the lower chamber should be increased to 800 btl (from the usual 600µl).
Several other methods can be used to count the invading cells. For example, cells can contain fluorogenic substrates (Garrido et al., 1995), radioisotopes (Garrido et al., 1995; Muir et al., 1993), or dyes (Imamura et al., 1994). These labels are suitable when dealing with a large number of samples; however, visual cell counting is still necessary for optimizing the methods or for checking reproducibility of the methods. When the number of invading cells is large enough (>1000), the cells can be counted using a Coulter counter after collecting the invading cells by trypsinization of the filter.
Transwells are normally reusable four or five times or until their polycarbonate filters become cracked.
References
Albini, A., Iwamoto, Y., Kleinman, H. K., Martin, C. R., Aaronson, S. A., Kozlowski, J. M., and McEwan, R. N. (1987). A rapid in vitro assay for quantieating the invasive potential of tumor cells. Cancer Res. 47, 3239-3245.
Garrido, T., Riese, H. H., Quesada, A. R., Barbacid, M. M., and Aracil, M. (1995). Quantitative assay for cell invasion using the fluorogenic substrate 2'. 7'-bis(2-carboxyethyl)-5(and-6)-carboxyfluorescein acetoxymethylester. Anal. Biochem. 235, 234-236.
Hendrix, M. J. C., Seftor, E. A., Seftor, R. E. B., and Fidler, L. J. (1987). A simple quantitative assay for studying the invasive potential of high and low human metastatic variants. Cancer Lett. 38, 137-147.
Kramer R. H., and Nicolson, C. L. (1979). Interactions of tumor cells with vascular endothelial cell monolayers: A model for metastatic invasion. Proc. Natl. Acad. Sci. USA 76, 5704-5708.
Liotta, L A. (1986). Tumor invasion and metastatic-role of extracellular matrix: Rhoads memorial award lecture. Cancer Res. 46,1-7.
Mareel, M., Dragonetti, C., Travernier J., and Fiers, W. (1988). Tumorselective cytotoxic effects of murine necrosis factor (TNF) and interferon-gamma in organ culture of BIG melanoma cells and heart tissue. Int. J. Cancer 42, 470-473.
Muir, D., Sukhu, L., Johnson, J., Lahorra, M. A., and Maria, B. L. (1993). Quantitative methods for scoring cell migration and invasion in filter-based assays. Anal. Biochem. 215, 104-199.
Nabeshirna, K., Kataoka, H., Koita, H., Murayama, T., and Koono, M. (1988). A new long-term in vitro invasion assay using fibrous connective tissue matrices maintaining architectural characteristics of connective tissue. Invas. Metast. 8, 301-316.
Nicolson, G. L., Dulski, K., Basson, C., and Welch, D. R. (1985). Preferential organ attachment and invasion in vitro by B16 melanoma cells selected for differing metastatic colonization and invasive properties. Invas. Metast. 5, 144-158.
Repesh, L. A. (1989). A new in vitro assay for quantitating tumor cell invasion. Invas. Metast. 9, 192-208.
Schor, S. L., Allen, T. D., and Harrison, C. J. (1980). Cell migration through three-dimensional gels of native collagen fibres: Collagenolytic activity is not required for the migration of two permanent cell lines. J. Cell Sci. 46, 171-186.
Simon, N., N6el, A., and Foidart, J.-M. (1992). Evaluation of in vitro reconstituted basement membrane assay to assess the invasiveness of tumor cells. Invas. Metast. 12, 156-167.
Waller, C. A., Braun, M., and Schirrrnacher, V. (1986). Quantitative analysis of cancer invasion in vitro: Comparison of two new assays and of tumour sublines with different metastatic capacity. Clin. Exp. Metast. 4, 73-89.
Yagel, S., Khokha, R., Denhardt, D. T., Kerbel, R. S., Parhar, R. S., and Lala, P. K. (1989). Mechanisms of cellular invasiveness: A comparison of amnion invasion in vitro and metastatic behavior in vivo. JNCI 81, 768-775.




