Light Reactions
How the electromagnetic energy of light is converted to chemical energy in the form of reduced organic molecules is complex. Nonetheless, the first principles of energy conservation and conversions in photosynthesis may be simply depicted. All higher photosynthetic organisms contain two forms of the green pigment chlorophyll. More than 99% of the chlorophyll in chloroplasts, the organelles in which photosynthesis takes place, functions in a passive, purely physical manner. Organized in specific pigment– protein complexes within the photosynthetic membrane, these chlorophylls absorb visible light and transfer excitation energy to nearby chlorophylls with efficiencies very close to 100%. In a real sense, more than 99% of the chlorophylls function only to gather light and as such they are often referred to as light-harvesting chlorophylls.Within picoseconds of the harvesting, the excitation energy is transferred to specialized chlorophyll molecules called reaction center chlorophylls. These reaction center chlorophylls are identical to the majority of the lightharvesting chlorophylls.Yet, rather than acting in a passive manner when they are excited, the reaction center chlorophylls perform photochemistry. The two reaction center chlorophylls are termed P700 and P680. The “P” stands for pigment and the numbers refer to their absorption maxima, in nanometers, in the red region of the spectrum. The reaction center chlorophylls were first detected by lightinduced bleaching at 680 and 700 nm. When the reaction center chlorophylls are excited, either directly or by resonance energy transfer from excited light-harvesting chlorophylls, an electron is transferred from the reaction center chlorophyll ensemble to an electron acceptor. These light-driven oxidation–reduction reactions occur within picoseconds and can operate with a quantum efficiency that is close to 100%. The reactions may be written as follows:
| ⇒ Equation [6] | P700* + FeS → P700+ + FeS− |
| ⇒ Equation [7] | P680* + Q → P680+ + Q−, |
where the asterisks indicate the first excited singlet state of the reaction center chlorophyll, and FeS and Q are the redox active part of an iron–sulfur protein and a quinone, respectively, the first stable electron acceptors. P700+ and P680+ are chlorophyll cation radicals and Q− is a half reduced quinone and FeS− is a reduced iron-sulfur protein. The reactions shown in Eqs. (6) and (7) cannot take place, in the direction shown, in the dark when the reaction center chlorophylls are in the unexcited, ground state. The ΔG0' for both these reactions is approximately +24 kcal/mol. The excited reaction center chlorophylls are, however, much stronger reducing agents than the ground state chlorophylls are. The E0' of P700* is about 1.3 V more reducing than that of P700 in the ground state. These two electron transfer reactions are the only lightdriven reactions in photosynthesis and they set the entire process in motion. The electron transport chain of chloroplasts is illustrated in Fig. 10.
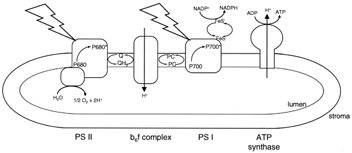 |
| Figure 10 Electron transport and ATP synthesis in chloroplasts. The jagged arrows represent light striking the two photosystems (PS I and PS II) in the thylakoid membrane. Other members of the electron transport chain shown are a quinone (Q), the cytochrome complex (b6f ), plastocyanin (PC), and an iron–sulfur protein (FeS). The chloroplast ATP synthase is shown making ATP at the expense of the electrochemical proton gradient generated by electron transport. |
Specific light-harvesting chlorophyll–protein complexes are associated with the reaction center chlorophyll– protein complexes in assemblies known as photosystems. Photosystem I (PS I) contains P700 and the FeS acceptor, and photosystem II (PS II), P680 and the quinone acceptor. Electron transfer in PS I generates a relatively weak oxidizing agent (P700+, E0'=+430 mV) and a strong reductant (FeS−, E0'=−600 mV). The primary reductant generated in photosynthesis is nicotinamide adenine dinucleotide phosphate (NADP+), which, as the name suggests, differs from NAD+ by a single phosphate. While the physical properties of NADP+ and NAD+ are very similar, enzymes that use these pyridine nucleotides as substrates can discriminate between them by at least a factor of 1000. In general NAD+ is used in catabolic metabolism as we have seen for glycolysis and the tricarboxylic acid cycle. The reduced form of NADP+, NADPH, is, in contrast, used in biosynthesis, or anabolic metabolism. The E0' of the NADP+−NADPH redox pair is −340 mV. Thus, electron transfer from the reduced iron–sulfur protein of PS I to NADP+ is energetically a very favorable spontaneous reaction. It is NADPH that provides the electrons for CO2 reduction. The ultimate electron donor is water.
Two water molecules are oxidized by PS II to yield four protons and molecular oxygen. Water is a very weak reducing agent. Thus, a strong oxidizing agent is needed for water oxidation. P680+ fits the bill. The midpoint potential of the P680+−P680 redox pair is on the order of +1 V. Since the water–oxygen redox couple has an E0' of +0.815 V, the oxidation of water by P680+ is an energetically spontaneous reaction. Water oxidation is catalyzed by a manganese-containing enzyme that is plugged into the energy-converting thylakoid membrane.
So far, we have seen that the reduced FeS protein of PS I is converted to its oxidized form by passing electrons eventually to NADP+. In PS II, P680+ is reduced to P680 with electrons extracted from water. For electron transport to continue, the electron acceptor of PS II, Q−, and the electron donor of PS I, P700+, must be oxidized and reduced, respectively. The redox potential of the Q−Q− couple is about +0.05 V, whereas that of P700+−P700 is near +0.450 V. Thus, electron transport from Q− to P700+ is energetically spontaneous with a free energy of 9.3 kcal/mol for each electron transferred.
Electron transport from Q− to P700+ is mediated by a quinone, iron–sulfur, and a cytochrome protein complex in the thylakoid membrane. This protein, the cytochrome b6f complex, is remarkably similar to the cytochrome bc1 complex of the mitochondrial electron transport chain.




