Classes of Molluscs
Classes of Molluscs
For more than 50 years five classes of living molluscs were recognized: Amphineura, Gastropoda, Scaphopoda, Bivalvia (also called Pelecypoda), and Cephalopoda. Discovery of Neopilina in the 1950s added another class (Monoplacophora), and Hyman* contended that
solenogasters and chitons
make up separate classes (Polyplacophora
and Aplacophora), lapsing the
name Amphineura. Recognition of
important differences between organisms
such as Chaetoderma and other
solenogasters has separated Aplacophora
into the sister groups Caudofoveata
and Solenogastres.†
Class Caudofoveata
Members of class Caudofoveata are wormlike, marine organisms ranging from 2 to 140 mm in length (see Figure 16-41). They are mostly burrowers and orient themselves vertically, with the terminal mantle cavity and gills at the entrance of the burrow. They feed on microorganisms and detritus. They have no shell, but their bodies are covered with calcareous scales. There are no spicules or scales on the oral pedal shield, an organ apparently associated with food selection and intake. A radula is present, although reduced in some, and sexes are separate. This little group has fewer than 70 species; however, its features may be closer to those of the common ancestor of molluscs than any other living molluscs.
Class Solenogastres
Solenogasters (see Figure 16-41) and caudofoveates were formerly united in class Aplacophora, and some zoologists retain the name Aplacophora for solenogasters, excluding caudofoveates. Both caudofoveates and solenogasters are marine, wormlike, shell-less, with calcareous scales or spicules in their integument, with reduced head, and without nephridia. Solenogasters, however, usually have no radula and no gills (although secondary respiratory structures may be present). Their foot is represented by a midventral, narrow furrow, the pedal groove. They are hermaphroditic. Rather than burrowing, solenogasters live free on the bottom, and they often live and feed on cnidarians. Solenogasters are also a small group, numbering about 250 species.
Class Monoplacophora
Until 1952 it was thought that Monoplacophora were extinct; they were known only from Paleozoic shells. However, in that year living specimens of Neopilina (Gr. neo, new, + pilos, felt cap) were dredged up from the ocean bottom near the west coast of Costa Rica. Nearly a dozen species of monoplacophorans are now known. These molluscs are small and have a low, rounded shell and a creeping foot (Figure 16-8). They have superficial resemblance to limpets, but unlike most other molluscs, a number of organs are serially repeated. Such serial repetition occurs to a more limited extent in chitons. Neopilina has five pairs of gills, two pairs of auricles, six pairs of nephridia, one or two pairs of gonads, and a ladderlike nervous system with 10 pairs of pedal nerves. The mouth bears the characteristic radula.
Class Polyplacophora: Chitons
Chitons (Gr. coat of mail, tunic) (Figures 16-9 and 16-10) represent a somewhat more diverse molluscan group. They are rather flattened dorsoventrally and have a convex dorsal surface that bears eight articulating limy plates, or valves, hence their name Polyplacophora (“many plate bearers”). The plates overlap posteriorly and are usually dull colored to match the rocks to which chitons cling. Their head and cephalic sensory organs are reduced, but photosensitive structures (esthetes), which have the form of eyes in some chitons, pierce the plates.
Most chitons are small (2 to 5 cm);
the largest, Cryptochiton (Gr. crypto,
hidden, + chiton, coat of mail), rarely
exceeds 30 cm. They prefer rocky surfaces
in intertidal regions, although
some live at great depths. Most chitons
are stay-at-home organisms, straying
only very short distances for feeding.
In feeding, the radula projects from the
mouth to scrape algae from the rocks.
The radula is reinforced with the ironcontaining
mineral, magnetite. A chiton
clings tenaciously to its rock with
its broad, flat foot. If detached, it can
roll up like an armadillo for protection.
The mantle forms a girdle around the margin of the plates, and in some species mantle folds cover part or all of the plates. Compared with the primitive condition, the mantle cavity has been extended along the side of the foot, and the gills have been increased in number. Thus the gills are suspended from the roof of the mantle cavity along each side of the broad ventral foot. With the foot and the mantle margin adhering tightly to the substrate, these grooves become closed chambers, open only at the ends. Water enters the grooves anteriorly, flows across the gills, and leaves posteriorly, bringing a continuous supply of oxygen to the gills. At low tide the margins of the mantle can be tightly pressed to the substratum to diminish water loss, but in some circumstances, the mantle margins can be held open for limited air breathing. A pair of osphradia (sense organs for sampling water) are found in the mantle grooves near the anus of many chitons.
Blood pumped by the threechambered heart reaches the gills by way of an aorta and sinuses. A pair of kidneys (metanephridia) carries waste from the pericardial cavity to the exterior. Two pairs of longitudinal nerve cords are connected in the buccal region.
Sexes are separate in most chitons, and trochophore larvae metamorphose directly into juveniles, without an intervening veliger stage.
Class Scaphopoda
Scaphopoda, commonly called tusk shells or tooth shells, are benthic marine molluscs found from the subtidal zone to over 6000 m depth. They have a slender body covered with a mantle and a tubular shell open at both ends. In scaphopods the molluscan body plan has taken a new direction, with the mantle wrapped around the viscera and fused to form a tube. Most scaphopods are 2.5 to 5 cm long, although they range from 4 mm to 25 cm long. Dentalium (L. dentis, tooth) is a common Atlantic genus.
The foot, which protrudes through the larger end of the shell, is used to burrow into mud or sand, always leaving the small end of the shell exposed to the water above (Figure 16-11). Respiratory water circulates through the mantle cavity both by movements of the foot and ciliary action (Figure 16-11). Gaseous exchange occurs in the mantle, for gills are absent. Most food is detritus and protozoa from the substratum. It is caught on cilia of the foot or on the mucus-covered, ciliated knobs of the long tentacles extending from the head (captacula) and is conveyed to the nearby mouth. A radula carries food to a crushing gizzard. The captacula may serve some sensory function, but eyes, tentacles, and osphradia typical of many other molluscs are lacking. Sexes are separate, and the larva is a trochophore.
Class Gastropoda
Among molluscs class Gastropoda is by far the largest and most diverse, containing about 40,000 living and 15,000 fossil species. It contains so much diversity that there is no single general term in our language that can apply to them as a whole. They include snails, limpets, slugs, whelks, conchs, periwinkles, sea slugs, sea hares, and sea butterflies. They range from marine molluscs with many primitive characters to highly evolved terrestrial, air-breathing snails and slugs. These animals are basically bilaterally symmetrical, but because of torsion, a twisting process that occurs in the veliger stage, their visceral mass has become asymmetrical.
The shell, when present, is always of one piece (univalve) and may be coiled or uncoiled. Starting at the apex, which contains the oldest and smallest whorl, the whorls become successively larger and spiral around the central axis, or columella (Figure 16-12). The shell may be right handed (dextral) or left handed (sinistral), depending on the direction of coiling. Dextral shells are far more common. Direction of coiling is genetically controlled.
Gastropods range from microscopic forms to giant marine forms such as Pleuroploca gigantea, a snail with a shell up to 60 cm long, and sea hares Aplysia (see Figure 16-21), some species of which reach 1 m in length. Most of them, however, are between 1 and 8 cm in length. Some fossil gastropods are as much as 2 m long.
The range of gastropod habitats is large. In the sea gastropods are common both in littoral zones and at great depths, and some are even pelagic. Some are adapted to brackish water and others to fresh water. On land they are restricted by such factors as mineral content of the soil and extremes of temperature, dryness, and acidity. Even so, they are widespread, and some have been found at great altitudes and some even in polar regions. Snails occupy all kinds of habitats: in small pools or large bodies of water, in woodlands, in pastures, under rocks, in mosses, on cliffs, in trees, underground, and on the bodies of other animals. They have successfully undertaken every mode of life except aerial locomotion.
Gastropods are usually sluggish, sedentary animals because most of them have heavy shells and slow locomotion. Some are specialized for climbing, swimming, or burrowing. Shells are their chief defense, although they are also protected by coloration and by secretive habits. Many snails have an operculum, a horny plate that covers the shell aperture when the body is withdrawn into the shell. Others lack shells altogether. Some are distasteful to other animals, and a few such as Strombus can deal an active blow with the foot, which bears a sharp operculum. Nevertheless, they are eaten by birds, beetles, small mammals, fish, and other predators. Serving as intermediate hosts for many kinds of parasites, especially trematodes, snails are often harmed by larval stages of parasites.
Torsion
Of all molluscs, only gastropods undergo torsion. Torsion is a peculiar phenomenon that moves the mantle cavity, which was originally (primitively) posterior, to the front of the body, thus twisting the visceral organs as well through a 90- to 180-degree rotation. It occurs during the veliger stage, and in some species the first part may take only a few minutes. The second 90 degrees typically takes a longer period. Before torsion occurs, the embryo’s mouth is anterior and the anus and mantle cavity are posterior (Figure 16-13). The change is brought about by an uneven growth of the right and left muscles that attach the shell to the head-foot.
After torsion, the anus and mantle cavity become anterior and open above the mouth and head. The left gill, kidney, and heart auricle are now on the right side, whereas the original right gill, kidney, and heart auricle are now on the left, and the nerve cords have been twisted into a figure eight. Because of the space available in the mantle cavity, the animal’s sensitive head end can now be withdrawn into the protection of the shell, with the tougher foot forming a barrier to the outside.
Varying degrees of detorsion are seen in opisthobranchs and pulmonates, and the anus opens to the right side or even to the posterior. However, both of these groups were derived from torted ancestors.
The curious arrangement that results from torsion poses a serious sanitation problem by creating the possibility of wastes being washed back over the gills (fouling) and causes us to wonder what strong evolutionary pressures selected for such a strange realignment of body structures. Several explanations have been proposed, none entirely satisfying. For example, sense organs of the mantle cavity (osphradia) would better sample water when turned in the direction of travel. Certainly the consequences of torsion and the resulting need to avoid fouling have been very important in the subsequent evolution of gastropods. These consequences cannot be explored, however, until another unusual fea-ture of gastropods— coiling—has been described.
Coiling
Coiling, or spiral winding, of the shell and visceral mass is not the same as torsion. Coiling may occur in the larval stage at the same time as torsion, but the fossil record shows that coiling was a separate evolutionary event and originated in gastropods earlier than torsion did. Nevertheless, all living gastropods have descended from coiled, torted ancestors, whether or not they now show these characteristics.
Early gastropods had a bilaterally symmetrical planospiral shell; that is, all the whorls lay in a single plane (Figure 16-14A). Such a shell was not very compact, since each whorl had to lie completely outside the preceding one. Curiously, a few modern species have secondarily returned to the planospiral form. The compactness problem of a planospiral shell was solved by the conispiral shape, in which each succeeding whorl is at the side of the preceding one (Figure 16-14B). However, this shape was clearly unbalanced, hanging as it was with much weight over to one side. Better weight distribution was achieved by shifting the shell upward and posteriorly, with the shell axis oblique to the longitudinal axis of the foot (Figure 16-14C). The weight and bulk of the main body whorl, the largest whorl of the shell, pressed on the right side of the mantle cavity, however, and apparently interfered with the organs on that side. Accordingly, the gill, auricle, and kidney of the right side have been lost in most living gastropods, leading to a condition of bilateral asymmetry.
Although loss of the right gill was probably an adaptation to the mechanics of carrying a coiled shell, that condition displayed in most modern prosobranchs made possible a way to avoid the problem caused by torsion— fouling. Water is brought into the left side of the mantle cavity and out the right side, carrying with it wastes from the anus and nephridiopore, which lie near the right side. Ways in which fouling is avoided in other gastropods are mentioned later in this section.
Some snails, such as Bullia and Buccinum, are scavengers living on dead and decaying flesh; others are carnivores that tear their prey with radular teeth. Melongena feeds on clams, especially Tagelus, the razor clam, thrusting its proboscis between the gaping shell valves. Fasciolaria and Polinices (Figure 16-15B) feed on a variety of molluscs, preferably bivalves. Urosalpinx cinerea, oyster borers, drill holes through the shell of oysters. Their radula, bearing three longitudinal rows of teeth, is used first to begin the drilling action, then the snails glide forward, evert an accessory boring organ through a pore in the anterior sole of their foot, and hold it against the oyster’s shell, using a chemical agent to soften the shell. Short periods of rasping alternate with long periods of chemical activity until a neat round hole is completed. With its proboscis inserted through the hole, a snail may feed continuously for hours or days, using its radula to tear away the soft flesh. Urosalpinx is attracted to its prey at some distance by sensing some chemical, probably one released in metabolic wastes of the prey.
Cyphoma gibbosum and related species live and feed on gorgonians (phylum Cnidaria, Radiate Animals) in shallow, tropical coral reefs. These snails are commonly known as flamingo tongues. During normal activity their brightly colored mantle entirely envelops the shell, but it can be quickly withdrawn into the shell aperture when the animal is disturbed.
Members of the genus Conus (Figure 16-16) feed on fish, worms, and molluscs. Their radula is highly modified for prey capture. A gland charges the radular teeth with a highly toxic venom. When Conus senses the presence of its prey, a single radular tooth slides into position at the tip of the proboscis. Upon striking the prey, the proboscis expels a tooth like a harpoon, and the poison quiets the prey at once. This is an effective adaptation for a slowly moving predator to prevent escape of a swiftly moving prey. Some species of Conus can deliver very painful stings, and in several species the sting is lethal to humans. The venom consists of a series of toxic peptides, and each Conus species carries peptides (conotoxins) that are specific for the neuroreceptors of its preferred prey. Conotoxins have become valuable tools in research on the various receptors and ion channels of nerve cells.
Some gastropods feed on organic
deposits on the sand or mud. Others
collect the same sort of organic debris
but can digest only the microorganisms
contained in it. Some sessile gastropods,
such as some limpets, are ciliary
feeders that use gill cilia to draw in
particulate matter, roll it into a mucous
ball, and carry it to their mouth. Some
sea butterflies secrete a mucous net to
catch small planktonic forms; then they
draw the web into the mouth.
After maceration by the radula or by some grinding device, such as a gizzard in the sea hare Aplysia, digestion is usually extracellular in the lumen of the stomach or digestive glands. In ciliary feeders the stomachs are sorting regions, and most of the digestion is intracellular in digestive glands.
Internal Form and Function
Respiration in most gastropods is carried out by a ctenidium (two ctenidia is the primitive condition, found in some prosobranchs) located in the mantle cavity, though some aquatic forms, lacking gills, depend on the mantle and skin. After the more derived prosobranchs lost one of the gills, most of them lost half of the remaining one, and the central axis became attached to the wall of the mantle cavity (Figure 16-17). Thus they attained the most efficient gill arrangement for the way the water circulated through the mantle cavity (in one side and out the other).
Pulmonates have a highly vascular area in the mantle that serves as a lung (Figure 16-18). Most of the mantle margin seals to the back of the animal, and the lung opens to the outside by a small opening called a pneumostome. Many aquatic pulmonates must surface to expel a bubble of gas from the lung. To inhale, they curl the edge of the mantle around the pneumostome to form a siphon.
Most gastropods have a single nephridium (kidney). The circulatory and nervous systems are well developed (Figure 16-18). The latter incorporates three pairs of ganglia connected by nerves. Sense organs include eyes or simple photoreceptors, statocysts, tactile organs, and chemoreceptors. The simplest type of gastropod eye is simply a cuplike indentation in the skin lined with pigmented photoreceptor cells. In many gastropods the eyecup contains a lens and is covered with a cornea. A sensory area called an osphradium, located at the base of the incurrent siphon of most gastropods, is chemosensory in some forms, although its function may be mechanoreceptive in some and is still unknown in others.
There are both dioecious and monoecious gastropods. Many gastropods perform courtship ceremonies. During copulation in monoecious species there is an exchange of spermatozoa or spermatophores (bundles of sperm). Many terrestrial pulmonates eject a dart from a dart sac (Figure 16-18) into the partner’s body to heighten excitement before copulation. After copulation each partner deposits its eggs in shallow burrows in the ground. Gastropods with the most primitive characteristics discharge ova and sperm into seawater where fertilization occurs, and embryos soon hatch as freeswimming trochophore larvae. In most gastropods fertilization is internal.
Fertilized eggs encased in transparent shells may be emitted singly to float among the plankton or may be laid in gelatinous layers attached to the substratum. Some marine forms enclose their eggs, either in small groups or in large numbers, in tough egg capsules, or in a wide variety of egg cases (Figure 16-19). The young generally emerge as veliger larvae (Figure 16-7), or they may spend the veliger stage in the case or capsule and emerge as young snails. Some species, including many freshwater snails, are ovoviviparous, brooding their eggs and young in the pallial oviduct.
Major Groups of Gastropods
Traditional classification of class Gastropoda recognized three subclasses: Prosobranchia, much the largest subclass, almost all of which are marine; Opisthobranchia, an assemblage including sea slugs, sea hares, nudibranchs, and canoe shells, all marine; and Pulmonata, containing most freshwater and terrestrial species. Currently, gastropod taxonomy is in flux. Evidence suggests that Prosobranchia is paraphyletic. Opisthobranchia may or may not be paraphyletic, but Opisthobranchia and Pulmonata together apparently form a monophyletic grouping. For convenience and organization, we will continue to use the words “prosobranchs” and “opisthobranchs,” recognizing that they may not represent valid taxa.
Prosobranchs: This group contains most marine snails and some freshwater and terrestrial gastropods. The mantle cavity is anterior as a result of torsion, with the gill or gills lying in front of the heart. Water enters the left side and exits from the right side, and the edge of the mantle often extends into a long siphon to separate incurrent from excurrent flow. In prosobranchs with two gills (for example, the abalone Haliotis and the keyhole limpet Diodora, Figures 16-15A and 16-20A), fouling is avoided by having the excurrent water go up and out through one or more holes in the shell above the mantle cavity. Prosobranchs have one pair of tentacles. Sexes are usually separate. An operculum is often present.
They range in size from periwinkles and small limpets (Patella and Diodora) (Figure 16-20A) to horse conchs (Pleuroploca), the largest gastropods in the Atlantic Ocean. Familiar examples of prosobranchs are abalones (Haliotis), which have an ear-shaped shell; whelks (Busycon), which lay their eggs in double-edged, disc-shaped capsules attached to a cord a meter long; common periwinkles (Littorina); moon snails (Polinices, Figure 16-15B); oyster borers (Urosalpinx), which bore into oysters and suck out their juices; rock shells (Murex), a European species that was used to make the royal purple of the ancient Romans; and some freshwater forms (Goniobasis and Viviparus).
Opisthobranchs: Opisthobranchs are an odd assemblage of molluscs that include sea slugs, sea hares, sea butterflies, and canoe shells. They are nearly all marine; most of them are shallow-water forms, hiding under stones and seaweed; a few are pelagic. Currently nine or more orders of opisthobranchs are recognized. Opisthobranchs show partial or complete detorsion; thus the anus and gill (if present) are displaced to the right side or rear of the body. Clearly, the fouling problem is obviated if the anus is moved away from the head toward the posterior. Two pairs of tentacles are usually found, and the second pair is often further modified (rhinophores, Figure 16-21), with platelike folds that apparently increase the area for chemoreception. Their shell is typically reduced or absent. All are monoecious.
Sea hares (Aplysia Figure 16-21), have large, earlike anterior tentacles and vestigial shells. In pteropods or sea butterflies (Cavolina and Clione) the foot is modified into fins for swimming; thus, they are pelagic and form a part of the plankton fauna.
Nudibranchs are carnivorous and often brightly colored (Figure 16-22). Plumed sea slugs (Aeolidae) which live on sea anemones and hydroids, have elongate papillae (cerata) covering their back. They ingest their prey’s nematocysts and transport the nematocysts undischarged to the tips of their cerata. There the nematocysts are placed in cnidosacs that open to the outside, and the aeolid can use its highjacked nematocysts for its own defense. Hermissenda is one of the more common West Coast nudibranchs.
Pulmonates: Pulmonates show some detorsion and include land and most freshwater snails and slugs (and a few brackish and saltwater forms). They have lost their ancestral ctenidia, but their vascularized mantle wall has become a lung, which fills with air by contraction of the mantle floor (some aquatic species have developed secondary gills in the mantle cavity). The anus and nephridiopore open near the pneumostome, and waste is expelled forcibly with air or water from the lung. They are monoecious. Aquatic species have one pair of nonretractile tentacles, at the base of which are the eyes; land forms have two pairs of tentacles, with the posterior pair bearing the eyes (Figure 16-23). Among the thousands of land species, some of the most familiar American forms are Helix, Polygyra, Succinea, Anguispira, Zonitoides, Limax, and Agriolimax. Aquatic forms are represented by Helisoma, Lymnaea, and Physa. Physa is a left-handed (sinistral) snail.
Class Bivalvia
(Pelecypoda)
Bivalvia are also known as Pelecypoda (pel-e-sip´o-da), or “hatchetfooted” animals, as their name implies (Gr. pelekys, hatchet, + pous, podos, foot). They are bivalved molluscs that include mussels, clams, scallops, oysters, and shipworms (Figures 16-24 to 16-27) and they range in size from tiny seed shells 1 to 2 mm in length to giant South Pacific clams Tridacna, which may reach more than 1 m in length and as much as 225 kg (500 pounds) in weight (see Figure 16-35). Most bivalves are sedentary filter feeders that depend on ciliary currents produced by the gills to bring in food materials. Unlike gastropods, they have no head, no radula, and very little cephalization. Most bivalves are marine, but many live in brackish water and in streams, ponds, and lakes.
Form and Function Shell: Bivalves are laterally compressed, and their two shells (valves) are held together dorsally by a hinge ligament that causes the valves to gape ventrally. The valves are drawn together by adductor muscles that work in opposition to the hinge ligament (Figure 16-26C and D). The umbo is the oldest part of the shell, and growth occurs in concentric lines around it (Figure 16-26A).
Pearl production is a by-product of a protective device used by the animals when a foreign object (grain of sand, parasite, or other) becomes lodged between the shell and mantle. The mantle secretes many layers of nacre around the irritating object (Figure 16-5). Pearls are cultured by inserting particles of nacre, usually taken from the shells of freshwater clams, between the shell and mantle of a certain species of oyster and by keeping the oysters in enclosures for several years. Meleagrina is an oyster used extensively by the Japanese for pearl culture.
Body and Mantle: The visceral mass is suspended from the dorsal midline, and the muscular foot is attached to the visceral mass anteroventrally. The ctenidia hang down on each side, each covered by a fold of the mantle. The posterior edges of the mantle folds are modified to form dorsal excurrent and ventral incurrent openings (Figure 16-28A). In some marine bivalves the mantle is drawn out into long muscular siphons that allow the clam to burrow into the mud or sand and extend the siphons to the water above (Figure 16-28B to D).
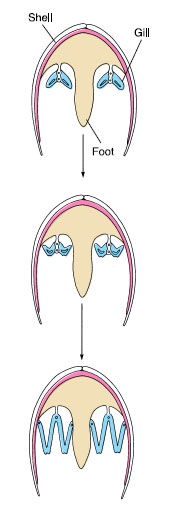
Locomotion: Bivalves initiate movement by extending a slender muscular foot between the valves (Figure 16-28D). Blood is pumped into the foot, causing it to swell and to act as an anchor in the mud or sand, then longitudinal muscles contract to shorten the foot and pull the animal forward.
Scallops and file shells are able to swim with a jerky motion by clapping their valves together to create a sort of jet propulsion. The mantle edges can direct the stream of expelled water, so that the animals can swim in virtually any direction (Figure 16-24).
Gills: Gaseous exchange occurs through both mantle and gills. Gills of most bivalves are highly modified for filter feeding; they are derived from primitive ctenidia by a great lengthening of filaments on each side of the central axis (Figure 16-29). As ends of long filaments became folded back toward the central axis, ctenidial filaments took the shape of a long, slender W. Filaments lying beside each other became joined by ciliary junctions or tissue fusions, forming platelike lamellae with many vertical water tubes inside. Thus water enters the incurrent siphon, propelled by ciliary action, then enters the water tubes through pores between the filaments in the lamellae, proceeds dorsally into a common suprabranchial chamber (Figure 16-30), and then out the excurrent aperture.
Feeding: Most bivalves are filter feeders. Respiratory currents bring both oxygen and organic materials to the gills where ciliary tracts direct them to the tiny pores of the gills. Gland cells on the gills and labial palps secrete copious amounts of mucus, which entangles particles suspended in water going through gill pores. These mucous masses slide down the outside of the gills toward food grooves at the lower edge of the gills (Figure 16-31). Heavier particles of sediment drop off the gills as a result of gravitational pull, but smaller particles travel along the food grooves toward the labial palps. The palps, being also grooved and ciliated, direct the mucous mass into the mouth.
Some bivalves, such as Nucula and Yoldia, are deposit feeders and have long proboscides attached to the labial palps (Figure 16-28C). These can be protruded onto sand or mud to collect food particles, in addition to particles attracted by gill currents.
Shipworms (Figure 16-27) burrow in wood and feed on particles they excavate. Symbiotic bacteria live in a special organ in the bivalve and produce cellulase to digest wood. Other bivalves such as giant clams gain much of their nutrition from the photosynthetic products of symbiotic algae living in their mantle tissue (Figure 16-35).
Septibranchs, another group of bivalves, draw small crustaceans or bits of organic debris into the mantle cavity by sudden inflow of water created by the pumping action of a muscular septum in the mantle cavity.
Internal Structure and Function
The floor of the stomach of filter-feeding bivalves is folded into ciliary tracts for sorting a continuous stream of particles. A cylindrical style sac opening into the stomach secretes a gelatinous rod called a crystalline style, which projects into the stomach and is kept whirling by means of cilia in the style sac (Figure 16-32). Rotation of the style helps to dissolve its surface layers, freeing digestive enzymes (especially amylase) that it contains, and to roll the mucous food mass. Dislodged particles are sorted, and suitable ones are directed to the digestive gland or engulfed by amebocytes. Further digestion is intracellular.
The three-chambered heart, which lies in the pericardial cavity (Figure 16-31), has two auricles and a ventricle and beats slowly, ranging from 0.2 to 30 times per minute. Part of the blood is oxygenated in the mantle and returns to the ventricle through the auricles; the rest circulates through sinuses and passes in a vein to the kidneys, from there to the gills for oxygenation, and back to the auricles.
A pair of U-shaped kidneys (nephridial tubules) lies just ventral and posterior to the heart (Figure 16-31B). The glandular portion of each tubule opens into the pericardium; the bladder portion empties into the suprabranchial chamber.
The nervous system consists of three pairs of widely separated ganglia connected by commissures and a system of nerves. Sense organs are poorly developed. They include a pair of statocysts in the foot, a pair of osphradia of uncertain function in the mantle cavity, tactile cells, and sometimes simple pigment cells on the mantle. Scallops (Pecten, Chlamys) have a row of small blue eyes along each mantle edge (Figure 16-25). Each eye has a cornea, lens, retina, and pigmented layer. Tentacles on the margin of the mantle of Pecten (Figure 16-25) and Lima have tactile and chemoreceptor cells.
Reproduction and Development
Sexes are usually separate. Gametes are discharged into the suprabranchial chamber to be carried out with the excurrent flow. An oyster may produce 50 million eggs in a single season. In most bivalves fertilization is external. The embryo develops into trochophore, veliger, and spat stages (Figure 16-33).
In most freshwater clams fertilization is internal. Eggs drop into the water tubes of the gills where they are fertilized by sperm entering with the incurrent flow. They develop there into a bivalved glochidium larva stage, which is a specialized veliger (Figure 16-34). When discharged, glochidia are carried by water currents, and if they come in contact with a passing fish, they attach to its gills or skin and live as parasites for several weeks. Then they sink to the bottom to begin independent lives. Larval “hitchhiking” helps distribute a form whose locomotion is very limited.
Boring: Many pelecypods can burrow into mud or sand, but some have evolved a mechanism for burrowing into much harder substances, such as wood or stone.
Teredo, Bankia, and some other genera are called shipworms. They can be very destructive to wooden ships and wharves. These strange little clams have a long, wormlike appearance, with a pair of slender posterior siphons that keep water flowing over the gills, and a pair of small globular valves on the anterior end with which they burrow (Figure 16-27). The valves have microscopic teeth that function as very effective wood rasps. The animals extend their burrows with an unceasing rasping motion of the valves. This motion sends a continuous flow of fine wood particles into the digestive tract where they are attacked by cellulase produced by symbiotic bacteria. Interestingly, these bacteria also fix nitrogen, an important property for their hosts, which live on a diet (wood) high in carbon but deficient in nitrogen.
Some clams bore into rock. The piddock (Pholas) bores into limestone, shale, sandstone, and sometimes wood or peat. It has strong valves that bear spines, which it uses to cut away the rock gradually while anchoring itself with its foot. Pholas may grow to 15 cm long and make rock burrows up to 30 cm long.
Class Cephalopoda
Cephalopoda (Gr. kephale, head, + pous, podos, foot) include squids, octopuses, nautiluses, devilfish, and cuttlefish. All are marine, and all are active predators.
The modified foot is concentrated in the head region. It takes the form of a funnel for expelling water from the mantle cavity, and the anterior margin is drawn out into a circle or crown of arms or tentacles.
Cephalopods range upward in size from 2 or 3 cm. The common squid of markets, Loligo, is about 30 cm long. The giant squid Architeuthis is the largest invertebrate known.
Fossil records of cephalopods go back to Cambrian times. The earliest shells were straight cones; others were curved or coiled, culminating in the coiled shell similar to that of the modern Nautilus, the only remaining member of the once flourishing nautiloids (Figure 16-36). Cephalopods without shells or with internal shells (such as octopuses and squids) apparently evolved from some early straightshelled ancestor. Many ammonoids, which are extinct, had quite elaborate shells (Figure 16-36C).
The natural history of some cephalopods is fairly well known. They are marine animals and appear sensitive to the degree of salinity. Few are found in the Baltic Sea, where the water has a low salt content. Cephalopods are found at various depths. Octopuses are often seen in the intertidal zone, lurking among rocks and crevices, but occasionally they are found at great depths. The more active squids are rarely found in very shallow water, and some have been taken at depths of 5000 m. Nautilus is usually found near the bottom in water 50 to 560 m deep, near islands in the southwestern Pacific.
Form and Function
Shell: Although early nautiloid and ammonoid shells were heavy, they were made buoyant by a series of gas chambers, as is that of Nautilus (Figure 16-36B), enabling the animal to swim while carrying its shell. The shell of Nautilus, although coiled, is quite different from that of a gastropod. The shell is divided by transverse septa into internal chambers (Figure 16-36B). The living animal inhabits only the last chamber. As it grows, it moves forward, secreting behind a new septum. The chambers are connected by a cord of living tissue called a siphuncle, which extends from the visceral mass. Cuttlefishes (Figure 16-37) also have a small, curved shell, but it is entirely enclosed by the mantle. In squids most of the shell has disappeared, leaving only a thin, horny strip called a pen, which is enclosed by the mantle. In Octopus (Gr. oktos, eight, + pous, podos, foot) the shell has disappeared entirely.
Locomotion: Cephalopods swim by forcefully expelling water from the mantle cavity through a ventral funnel (or siphon)—a sort of jet propulsion. The funnel is mobile and can be pointed forward or backward to control direction; the force of water expulsion controls speed.
Squids and cuttlefishes are excellent swimmers. The squid body is streamlined and built for speed (Figure 16-38). Cuttlefishes swim more slowly. The lateral fins of squids and cuttlefishes serve as stabilizers, but they are held close to the body for rapid swimming.
Nautilus is active at night; its gasfilled chambers keep the shell upright. Although not as fast as squids, it moves surprisingly well.
Octopus has a rather globular body and no fins (Figure 16-1E). An octopus can swim backward by spurting jets of water from its funnel, but it is better adapted to crawling about over the rocks and coral, using suction discs on its arms to pull or to anchor itself. Some deep-water octopods have the arms webbed like an umbrella and swim in a medusa-like fashion.
Internal Features: The active habits of cephalopods are reflected in their internal anatomy, particularly their respiratory, circulatory, and nervous systems.
Respiration and Circulation: Except for nautiloids, cephalopods have one pair of gills. Because ciliary propulsion would not circulate enough water for their high oxygen requirements, there are no cilia on the gills. Instead, radial muscles in the mantle wall compress the wall and enlarge the mantle cavity, drawing water inside. Strong circular muscles contract and expel water forcibly through the funnel. A system of one-way valves prevents water from being taken in through the funnel and expelled around the mantle margin.
Likewise, the open circulatory system of their ancestral molluscs would be inadequate for cephalopods. Their circulatory system consists of a closed network of vessels, and capillaries conduct blood through the gill filaments. Furthermore, the molluscan plan of circulation places the entire systemic circulation before the blood reaches the gills (in contrast to vertebrates, in which the blood leaves the heart and goes directly to the gills or lungs). This functional problem was solved by the development of accessory or branchial hearts (Figure 16-38B) at the base of each gill to increase the pressure of the blood going through the capillaries there.
Nervous and Sensory Systems: Nervous and sensory systems are more elaborate in cephalopods than in other molluscs. The brain, the largest in any invertebrate, consists of several lobes with millions of nerve cells. Squids have giant nerve fibers (among the largest known in the animal kingdom), which are activated when the animal is alarmed and that initiate maximal contractions of the mantle muscles for a speedy escape.
Sense organs are well developed. Except for Nautilus, which has relatively simple eyes, cephalopods have highly complex eyes with cornea, lens, chambers, and retina (Figure 16-39). Orientation of the eyes is controlled by the statocysts, which are larger and more complex than in other molluscs. The eyes are held in a constant relation to gravity, so that the slit-shaped pupils are always in a horizontal position. Octopods are apparently colorblind but can be taught to discriminate between shapes—for example, a square and a rectangle—and to remember such a discrimination for a considerable time. Experimenters find it easy to modify their behavior patterns by devices of reward and punishment. They are capable of observational learning; when one octopus observes another being rewarded by making a correct choice, the observer learns which choice is rewarded and consistently makes the same selection when given the opportunity.
Octopods use their arms for tactile exploration and can discriminate between textures by feel but apparently not between shapes. Their arms are well supplied with both tactile and chemoreceptor cells. Cephalopods seem to lack a sense of hearing.
Communication: Little is known of social behavior of nautiloids or deepwater cephalopods, but inshore and littoral forms such as Sepia, Sepioteuthis, Loligo, and Octopus have been studied extensively. Although their tactile sense is well developed and they have some chemical sensitivity, visual signals are the predominant means of communication. These signals consist of a host of movements of the arms, fins, and body, as well as many color changes. The movements may range from minor body motions to exaggerated spreading, curling, raising, or lowering of some or all of the arms. Color changes are effected by chromatophores, cells in the skin that contain pigment granules. Tiny muscle cells surround each elastic chromatophore, whose contractions pull the cell boundary of the chromatophore outward, causing it to expand greatly. As the cell expands, the pigment becomes dispersed, changing the color pattern of the animal. When the muscles relax, the chromatophores return to their original size and pigment becomes concentrated again. By means of the chromatophores, which are under nervous and probably hormonal control, an elaborate system of changes in color and pattern is possible, including general darkening or lightening; flushes of pink, yellow, or lavender; and the formation of bars, stripes, spots, or irregular blotches. These colors may be used variously as danger signals, as protective coloring, in courtship rituals, and probably in other ways.
By assuming different color patterns of different parts of the body, a squid can transmit three or four different messages simultaneously to different individuals and in different directions, and it can instantaneously change any or all of the messages. Probably no other system of communication in invertebrates can convey so much information so rapidly.
Deep-water cephalopods may have to depend more on chemical or tactile senses than their littoral or surface cousins, but they also produce their own type of visual signals, for they have evolved many elaborate luminescent organs.
Most cephalopods other than nautiloids have another protective device. An ink sac that empties into the rectum contains an ink gland that secretes sepia, a dark fluid containing the pigment melanin, into the sac. When the animal is alarmed, it releases a cloud of ink, which may hang in the water as a blob or be contorted by water currents. The animal quickly departs from the scene, leaving the ink as a decoy to the predator.
Reproduction: Sexes are separate in cephalopods. In the male seminal vesicle the spermatozoa are encased in spermatophores and stored in a sac that opens into the mantle cavity. One arm of the adult male is modified as an intromittent organ, called a hectocotylus, used to pluck a spermatophore from his own mantle cavity and insert it into the mantle cavity of a female near the oviduct opening (Figure 16-40). Before copulation males often undergo color displays, apparently directed against rival males. Eggs are fertilized as they leave the oviduct and are then usually attached to stones or other objects. Some octopods tend their eggs. Argonauta, the paper nautilus, secretes a fluted “shell,” or capsule, in which she broods her eggs.
The large yolky eggs undergo meroblastic cleavage. During embryonic development, the head and foot become indistinguishable. The ring around the mouth, which bears the arms, or tentacles, may be derived from the anterior part of the foot. A juvenile hatches from the egg; no free-swimming larva exists in cephalopods.
Major Groups of Cephalopods
There are three subclasses of cephalopods: Nautiloidea, which have two pairs of gills; the entirely extinct Ammonoidea; and Coleoidea, which have one pair of gills. Nautiloidea populated the Paleozoic and Mesozoic seas, but there survives only one genus, Nautilus (see Figure 16-36), of which there are five or six species. Nautilus’ head, with its 60 to 90 or more tentacles, can be extended from the opening of the body compartment of the shell. Its tentacles have no suckers but are made adhesive by secretions. They are used in searching for, sensing, and grasping food. Beneath its head is the funnel. Mantle, mantle cavity, and visceral mass are sheltered by the shell.
Ammonoids were widely prevalent in the Mesozoic era but became extinct by the end of the Cretaceous period. They had chambered shells analogous to nautiloids, but the septa were more complex, and the septal sutures (where the septa contact the inside of the shell) were frilled (compare shells in Figure 16-36B and C). The reasons for their extinction remain a mystery. Present evidence suggests that they were gone before the asteroid bombardment at the end of the Cretaceous period, and some nautiloids, which some ammonoids closely resembled, survive to the present.
Subclass Coleoidea includes all living cephalopods except Nautilus. There are four orders of coleoids. Members of order Sepioidea (cuttlefishes and their relatives) have a rounded or compressed, bulky body bearing fins (Figure 16-37). They have eight arms and two tentacles. Both arms and tentacles have suckers, but tentacles bear suckers only at their ends (Figure 16-37). Members of order Teuthoidea (squids, Figure 16-38) have a more cylindrical body but also have eight arms and two tentacles. Order Vampyromorpha (vampire squid) contains only a single, deep-water species. Members of order Octopoda have eight arms and no tentacles (see Figure 16-1E). Their bodies are short and saclike, with no fins. The suckers in squids are stalked (pendunculated), with horny rims bearing teeth; in octopuses the suckers are sessile and have no horny rims.
For more than 50 years five classes of living molluscs were recognized: Amphineura, Gastropoda, Scaphopoda, Bivalvia (also called Pelecypoda), and Cephalopoda. Discovery of Neopilina in the 1950s added another class (Monoplacophora), and Hyman* contended that
 |
| Figure 16-7 Veliger of a snail, Pedicularia, swimming. The adults are parasitic on corals. The ciliated process (velum) develops from the prototroch of the trochophore (Figure 16-6). |
Class Caudofoveata
Members of class Caudofoveata are wormlike, marine organisms ranging from 2 to 140 mm in length (see Figure 16-41). They are mostly burrowers and orient themselves vertically, with the terminal mantle cavity and gills at the entrance of the burrow. They feed on microorganisms and detritus. They have no shell, but their bodies are covered with calcareous scales. There are no spicules or scales on the oral pedal shield, an organ apparently associated with food selection and intake. A radula is present, although reduced in some, and sexes are separate. This little group has fewer than 70 species; however, its features may be closer to those of the common ancestor of molluscs than any other living molluscs.
 |
| Figure 16-8 Neopilina, class Monoplacophora. Living specimens range from 3 mm to about 3 cm in length. A, Ventral view. B, Dorsal view. |
Class Solenogastres
Solenogasters (see Figure 16-41) and caudofoveates were formerly united in class Aplacophora, and some zoologists retain the name Aplacophora for solenogasters, excluding caudofoveates. Both caudofoveates and solenogasters are marine, wormlike, shell-less, with calcareous scales or spicules in their integument, with reduced head, and without nephridia. Solenogasters, however, usually have no radula and no gills (although secondary respiratory structures may be present). Their foot is represented by a midventral, narrow furrow, the pedal groove. They are hermaphroditic. Rather than burrowing, solenogasters live free on the bottom, and they often live and feed on cnidarians. Solenogasters are also a small group, numbering about 250 species.
Class Monoplacophora
Until 1952 it was thought that Monoplacophora were extinct; they were known only from Paleozoic shells. However, in that year living specimens of Neopilina (Gr. neo, new, + pilos, felt cap) were dredged up from the ocean bottom near the west coast of Costa Rica. Nearly a dozen species of monoplacophorans are now known. These molluscs are small and have a low, rounded shell and a creeping foot (Figure 16-8). They have superficial resemblance to limpets, but unlike most other molluscs, a number of organs are serially repeated. Such serial repetition occurs to a more limited extent in chitons. Neopilina has five pairs of gills, two pairs of auricles, six pairs of nephridia, one or two pairs of gonads, and a ladderlike nervous system with 10 pairs of pedal nerves. The mouth bears the characteristic radula.
Class Polyplacophora: Chitons
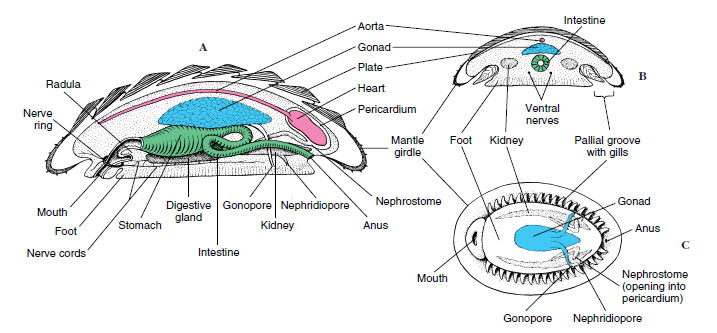
Chitons (Gr. coat of mail, tunic) (Figures 16-9 and 16-10) represent a somewhat more diverse molluscan group. They are rather flattened dorsoventrally and have a convex dorsal surface that bears eight articulating limy plates, or valves, hence their name Polyplacophora (“many plate bearers”). The plates overlap posteriorly and are usually dull colored to match the rocks to which chitons cling. Their head and cephalic sensory organs are reduced, but photosensitive structures (esthetes), which have the form of eyes in some chitons, pierce the plates.
 |
| Figure 16-9 Anatomy of a chiton (class Polyplacophora). A, Longitudinal section. B, Transverse section. C, External ventral view. |
 |
| Figure 16-10 Mossy chiton,Mopalia muscosa. The upper surface of the mantle, or “girdle,” is covered with hairs and bristles, an adaptation for defense. |
The mantle forms a girdle around the margin of the plates, and in some species mantle folds cover part or all of the plates. Compared with the primitive condition, the mantle cavity has been extended along the side of the foot, and the gills have been increased in number. Thus the gills are suspended from the roof of the mantle cavity along each side of the broad ventral foot. With the foot and the mantle margin adhering tightly to the substrate, these grooves become closed chambers, open only at the ends. Water enters the grooves anteriorly, flows across the gills, and leaves posteriorly, bringing a continuous supply of oxygen to the gills. At low tide the margins of the mantle can be tightly pressed to the substratum to diminish water loss, but in some circumstances, the mantle margins can be held open for limited air breathing. A pair of osphradia (sense organs for sampling water) are found in the mantle grooves near the anus of many chitons.
Blood pumped by the threechambered heart reaches the gills by way of an aorta and sinuses. A pair of kidneys (metanephridia) carries waste from the pericardial cavity to the exterior. Two pairs of longitudinal nerve cords are connected in the buccal region.
Sexes are separate in most chitons, and trochophore larvae metamorphose directly into juveniles, without an intervening veliger stage.
Class Scaphopoda
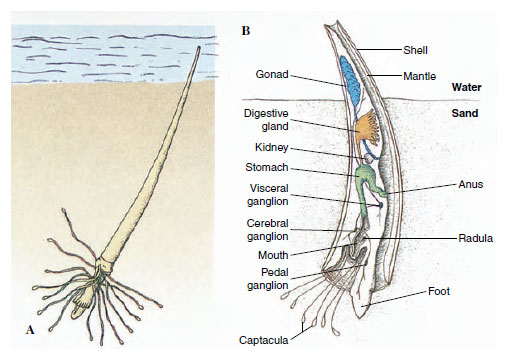
Scaphopoda, commonly called tusk shells or tooth shells, are benthic marine molluscs found from the subtidal zone to over 6000 m depth. They have a slender body covered with a mantle and a tubular shell open at both ends. In scaphopods the molluscan body plan has taken a new direction, with the mantle wrapped around the viscera and fused to form a tube. Most scaphopods are 2.5 to 5 cm long, although they range from 4 mm to 25 cm long. Dentalium (L. dentis, tooth) is a common Atlantic genus.
The foot, which protrudes through the larger end of the shell, is used to burrow into mud or sand, always leaving the small end of the shell exposed to the water above (Figure 16-11). Respiratory water circulates through the mantle cavity both by movements of the foot and ciliary action (Figure 16-11). Gaseous exchange occurs in the mantle, for gills are absent. Most food is detritus and protozoa from the substratum. It is caught on cilia of the foot or on the mucus-covered, ciliated knobs of the long tentacles extending from the head (captacula) and is conveyed to the nearby mouth. A radula carries food to a crushing gizzard. The captacula may serve some sensory function, but eyes, tentacles, and osphradia typical of many other molluscs are lacking. Sexes are separate, and the larva is a trochophore.
 |
| Figure 16-11 The tusk shell, Dentalium (class Scaphopoda). A, It burrows into soft mud or sand and feeds by means of its prehensile tentacles. Respiratory currents of water are drawn in by ciliary action through the small open end of the shell, then expelled through the same opening by muscular action. B, Internal anatomy of Dentalium. |
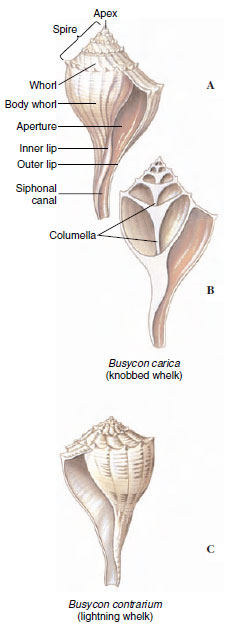 |
| Figure 16-12 Shell of the whelk Busycon. A and B, Busycon carica, a dextral, or right- handed, shell. A dextral shell has the aperture on the right side when the shell is held with the apex up and the aperture facing the observer. C, B. contrarium, a sinistral, or left-handed, shell. |
Class Gastropoda
Among molluscs class Gastropoda is by far the largest and most diverse, containing about 40,000 living and 15,000 fossil species. It contains so much diversity that there is no single general term in our language that can apply to them as a whole. They include snails, limpets, slugs, whelks, conchs, periwinkles, sea slugs, sea hares, and sea butterflies. They range from marine molluscs with many primitive characters to highly evolved terrestrial, air-breathing snails and slugs. These animals are basically bilaterally symmetrical, but because of torsion, a twisting process that occurs in the veliger stage, their visceral mass has become asymmetrical.
The shell, when present, is always of one piece (univalve) and may be coiled or uncoiled. Starting at the apex, which contains the oldest and smallest whorl, the whorls become successively larger and spiral around the central axis, or columella (Figure 16-12). The shell may be right handed (dextral) or left handed (sinistral), depending on the direction of coiling. Dextral shells are far more common. Direction of coiling is genetically controlled.
Gastropods range from microscopic forms to giant marine forms such as Pleuroploca gigantea, a snail with a shell up to 60 cm long, and sea hares Aplysia (see Figure 16-21), some species of which reach 1 m in length. Most of them, however, are between 1 and 8 cm in length. Some fossil gastropods are as much as 2 m long.
The range of gastropod habitats is large. In the sea gastropods are common both in littoral zones and at great depths, and some are even pelagic. Some are adapted to brackish water and others to fresh water. On land they are restricted by such factors as mineral content of the soil and extremes of temperature, dryness, and acidity. Even so, they are widespread, and some have been found at great altitudes and some even in polar regions. Snails occupy all kinds of habitats: in small pools or large bodies of water, in woodlands, in pastures, under rocks, in mosses, on cliffs, in trees, underground, and on the bodies of other animals. They have successfully undertaken every mode of life except aerial locomotion.
Gastropods are usually sluggish, sedentary animals because most of them have heavy shells and slow locomotion. Some are specialized for climbing, swimming, or burrowing. Shells are their chief defense, although they are also protected by coloration and by secretive habits. Many snails have an operculum, a horny plate that covers the shell aperture when the body is withdrawn into the shell. Others lack shells altogether. Some are distasteful to other animals, and a few such as Strombus can deal an active blow with the foot, which bears a sharp operculum. Nevertheless, they are eaten by birds, beetles, small mammals, fish, and other predators. Serving as intermediate hosts for many kinds of parasites, especially trematodes, snails are often harmed by larval stages of parasites.
 |
| Figure 16-13 Torsion in gastropods. A, Ancestral condition before torsion. B, Hypothetical intermediate condition. C, Early gastropod, torsion complete; direction of crawling now tends to carry waste products back into mantle cavity, resulting in fouling. |
Torsion
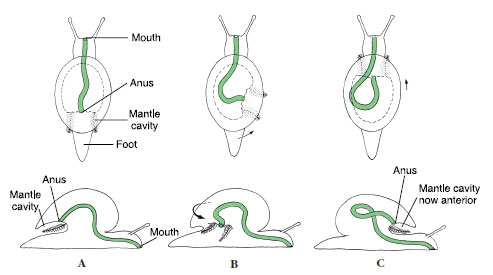
Of all molluscs, only gastropods undergo torsion. Torsion is a peculiar phenomenon that moves the mantle cavity, which was originally (primitively) posterior, to the front of the body, thus twisting the visceral organs as well through a 90- to 180-degree rotation. It occurs during the veliger stage, and in some species the first part may take only a few minutes. The second 90 degrees typically takes a longer period. Before torsion occurs, the embryo’s mouth is anterior and the anus and mantle cavity are posterior (Figure 16-13). The change is brought about by an uneven growth of the right and left muscles that attach the shell to the head-foot.
After torsion, the anus and mantle cavity become anterior and open above the mouth and head. The left gill, kidney, and heart auricle are now on the right side, whereas the original right gill, kidney, and heart auricle are now on the left, and the nerve cords have been twisted into a figure eight. Because of the space available in the mantle cavity, the animal’s sensitive head end can now be withdrawn into the protection of the shell, with the tougher foot forming a barrier to the outside.
Varying degrees of detorsion are seen in opisthobranchs and pulmonates, and the anus opens to the right side or even to the posterior. However, both of these groups were derived from torted ancestors.
The curious arrangement that results from torsion poses a serious sanitation problem by creating the possibility of wastes being washed back over the gills (fouling) and causes us to wonder what strong evolutionary pressures selected for such a strange realignment of body structures. Several explanations have been proposed, none entirely satisfying. For example, sense organs of the mantle cavity (osphradia) would better sample water when turned in the direction of travel. Certainly the consequences of torsion and the resulting need to avoid fouling have been very important in the subsequent evolution of gastropods. These consequences cannot be explored, however, until another unusual fea-ture of gastropods— coiling—has been described.
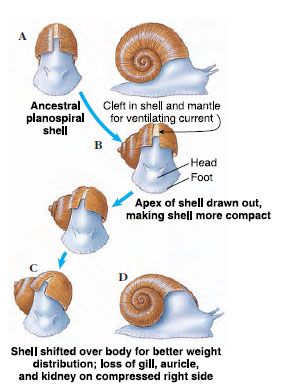 |
| Figure 16-14 Evolution of shell in gastropods. A, Earliest coiled shells were planospiral, each whorl lying completely outside the preceding whorl. B, Better compactness was achieved by snails in which each whorl lay partially to the side of the preceding whorl. C and D, Better weight distribution resulted when shell was moved upward and posteriorly. |
Coiling
Coiling, or spiral winding, of the shell and visceral mass is not the same as torsion. Coiling may occur in the larval stage at the same time as torsion, but the fossil record shows that coiling was a separate evolutionary event and originated in gastropods earlier than torsion did. Nevertheless, all living gastropods have descended from coiled, torted ancestors, whether or not they now show these characteristics.
Early gastropods had a bilaterally symmetrical planospiral shell; that is, all the whorls lay in a single plane (Figure 16-14A). Such a shell was not very compact, since each whorl had to lie completely outside the preceding one. Curiously, a few modern species have secondarily returned to the planospiral form. The compactness problem of a planospiral shell was solved by the conispiral shape, in which each succeeding whorl is at the side of the preceding one (Figure 16-14B). However, this shape was clearly unbalanced, hanging as it was with much weight over to one side. Better weight distribution was achieved by shifting the shell upward and posteriorly, with the shell axis oblique to the longitudinal axis of the foot (Figure 16-14C). The weight and bulk of the main body whorl, the largest whorl of the shell, pressed on the right side of the mantle cavity, however, and apparently interfered with the organs on that side. Accordingly, the gill, auricle, and kidney of the right side have been lost in most living gastropods, leading to a condition of bilateral asymmetry.
Although loss of the right gill was probably an adaptation to the mechanics of carrying a coiled shell, that condition displayed in most modern prosobranchs made possible a way to avoid the problem caused by torsion— fouling. Water is brought into the left side of the mantle cavity and out the right side, carrying with it wastes from the anus and nephridiopore, which lie near the right side. Ways in which fouling is avoided in other gastropods are mentioned later in this section.
Some snails, such as Bullia and Buccinum, are scavengers living on dead and decaying flesh; others are carnivores that tear their prey with radular teeth. Melongena feeds on clams, especially Tagelus, the razor clam, thrusting its proboscis between the gaping shell valves. Fasciolaria and Polinices (Figure 16-15B) feed on a variety of molluscs, preferably bivalves. Urosalpinx cinerea, oyster borers, drill holes through the shell of oysters. Their radula, bearing three longitudinal rows of teeth, is used first to begin the drilling action, then the snails glide forward, evert an accessory boring organ through a pore in the anterior sole of their foot, and hold it against the oyster’s shell, using a chemical agent to soften the shell. Short periods of rasping alternate with long periods of chemical activity until a neat round hole is completed. With its proboscis inserted through the hole, a snail may feed continuously for hours or days, using its radula to tear away the soft flesh. Urosalpinx is attracted to its prey at some distance by sensing some chemical, probably one released in metabolic wastes of the prey.
 |
| Figure 16-15 A, Red abalone, Haliotus rufescens. This huge, limpetlike snail is prized as food and extensively marketed. Abalones are strict vegetarians, feeding especially on sea lettuce and kelp. B, Moon snail, Polinices lewisii. A common inhabitant of West Coast sand flats, the moon snail is a predator of clams and mussels. It uses its radula to drill neat holes through its victim’s shell, through which the proboscis is then extended to eat the bivalve’s fleshy body. |
Cyphoma gibbosum and related species live and feed on gorgonians (phylum Cnidaria, Radiate Animals) in shallow, tropical coral reefs. These snails are commonly known as flamingo tongues. During normal activity their brightly colored mantle entirely envelops the shell, but it can be quickly withdrawn into the shell aperture when the animal is disturbed.
Members of the genus Conus (Figure 16-16) feed on fish, worms, and molluscs. Their radula is highly modified for prey capture. A gland charges the radular teeth with a highly toxic venom. When Conus senses the presence of its prey, a single radular tooth slides into position at the tip of the proboscis. Upon striking the prey, the proboscis expels a tooth like a harpoon, and the poison quiets the prey at once. This is an effective adaptation for a slowly moving predator to prevent escape of a swiftly moving prey. Some species of Conus can deliver very painful stings, and in several species the sting is lethal to humans. The venom consists of a series of toxic peptides, and each Conus species carries peptides (conotoxins) that are specific for the neuroreceptors of its preferred prey. Conotoxins have become valuable tools in research on the various receptors and ion channels of nerve cells.
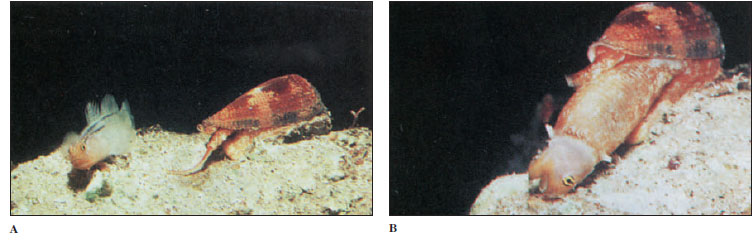 |
| Figure 16-16Conus extends its long, wormlike proboscis (A). When a fish attempts to consume this tasty morsel, the Conus stings it in the mouth and kills it. The snail engulfs the fish with its distensible stomach (B), then regurgitates the scales and bones some hours later. |
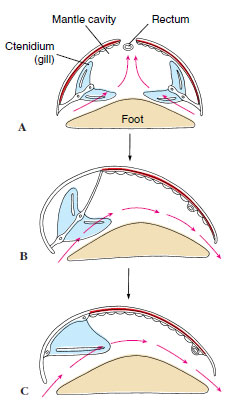 |
| Figure 16-17 Evolution of ctenidia in gastropods, A, Primitive condition with two ctenidia and excurrent water leaving the mantle cavity by a dorsal slit or hole. B, Condition after one ctenidium had been lost. C, Derived condition found in most marine gastropods, in which filaments on one side of remaining gill are lost, and axis is attached to mantle wall. |
After maceration by the radula or by some grinding device, such as a gizzard in the sea hare Aplysia, digestion is usually extracellular in the lumen of the stomach or digestive glands. In ciliary feeders the stomachs are sorting regions, and most of the digestion is intracellular in digestive glands.
Internal Form and Function
Respiration in most gastropods is carried out by a ctenidium (two ctenidia is the primitive condition, found in some prosobranchs) located in the mantle cavity, though some aquatic forms, lacking gills, depend on the mantle and skin. After the more derived prosobranchs lost one of the gills, most of them lost half of the remaining one, and the central axis became attached to the wall of the mantle cavity (Figure 16-17). Thus they attained the most efficient gill arrangement for the way the water circulated through the mantle cavity (in one side and out the other).
Pulmonates have a highly vascular area in the mantle that serves as a lung (Figure 16-18). Most of the mantle margin seals to the back of the animal, and the lung opens to the outside by a small opening called a pneumostome. Many aquatic pulmonates must surface to expel a bubble of gas from the lung. To inhale, they curl the edge of the mantle around the pneumostome to form a siphon.
Most gastropods have a single nephridium (kidney). The circulatory and nervous systems are well developed (Figure 16-18). The latter incorporates three pairs of ganglia connected by nerves. Sense organs include eyes or simple photoreceptors, statocysts, tactile organs, and chemoreceptors. The simplest type of gastropod eye is simply a cuplike indentation in the skin lined with pigmented photoreceptor cells. In many gastropods the eyecup contains a lens and is covered with a cornea. A sensory area called an osphradium, located at the base of the incurrent siphon of most gastropods, is chemosensory in some forms, although its function may be mechanoreceptive in some and is still unknown in others.
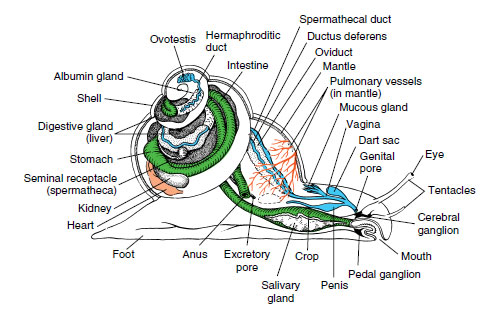 |
| Figure 16-18 Anatomy of a pulmonate snail. |
There are both dioecious and monoecious gastropods. Many gastropods perform courtship ceremonies. During copulation in monoecious species there is an exchange of spermatozoa or spermatophores (bundles of sperm). Many terrestrial pulmonates eject a dart from a dart sac (Figure 16-18) into the partner’s body to heighten excitement before copulation. After copulation each partner deposits its eggs in shallow burrows in the ground. Gastropods with the most primitive characteristics discharge ova and sperm into seawater where fertilization occurs, and embryos soon hatch as freeswimming trochophore larvae. In most gastropods fertilization is internal.
 |
| Figure 16-19 Eggs of marine gastropods. A, The wrinkled whelk, Thais lamelosa, lays egg cases "resembling grains of wheat; each contains hundreds of eggs. B, Egg ribbon of a dorid nudibranch. |
Fertilized eggs encased in transparent shells may be emitted singly to float among the plankton or may be laid in gelatinous layers attached to the substratum. Some marine forms enclose their eggs, either in small groups or in large numbers, in tough egg capsules, or in a wide variety of egg cases (Figure 16-19). The young generally emerge as veliger larvae (Figure 16-7), or they may spend the veliger stage in the case or capsule and emerge as young snails. Some species, including many freshwater snails, are ovoviviparous, brooding their eggs and young in the pallial oviduct.
Major Groups of Gastropods
Traditional classification of class Gastropoda recognized three subclasses: Prosobranchia, much the largest subclass, almost all of which are marine; Opisthobranchia, an assemblage including sea slugs, sea hares, nudibranchs, and canoe shells, all marine; and Pulmonata, containing most freshwater and terrestrial species. Currently, gastropod taxonomy is in flux. Evidence suggests that Prosobranchia is paraphyletic. Opisthobranchia may or may not be paraphyletic, but Opisthobranchia and Pulmonata together apparently form a monophyletic grouping. For convenience and organization, we will continue to use the words “prosobranchs” and “opisthobranchs,” recognizing that they may not represent valid taxa.
 |
| Figure 16-20 A, Diodora aspera, a gastropod with a hole in its apex through which water leaves the mantle cavity. B, Flamingo tongues, Cyphoma gibbosum, are showy inhabitants of Caribbean coral reefs, where they are associated with gorgonians. These snails have a smooth, creamy, orange to pink shell that is normally covered by the brightly marked mantle. |
Prosobranchs: This group contains most marine snails and some freshwater and terrestrial gastropods. The mantle cavity is anterior as a result of torsion, with the gill or gills lying in front of the heart. Water enters the left side and exits from the right side, and the edge of the mantle often extends into a long siphon to separate incurrent from excurrent flow. In prosobranchs with two gills (for example, the abalone Haliotis and the keyhole limpet Diodora, Figures 16-15A and 16-20A), fouling is avoided by having the excurrent water go up and out through one or more holes in the shell above the mantle cavity. Prosobranchs have one pair of tentacles. Sexes are usually separate. An operculum is often present.
They range in size from periwinkles and small limpets (Patella and Diodora) (Figure 16-20A) to horse conchs (Pleuroploca), the largest gastropods in the Atlantic Ocean. Familiar examples of prosobranchs are abalones (Haliotis), which have an ear-shaped shell; whelks (Busycon), which lay their eggs in double-edged, disc-shaped capsules attached to a cord a meter long; common periwinkles (Littorina); moon snails (Polinices, Figure 16-15B); oyster borers (Urosalpinx), which bore into oysters and suck out their juices; rock shells (Murex), a European species that was used to make the royal purple of the ancient Romans; and some freshwater forms (Goniobasis and Viviparus).
Opisthobranchs: Opisthobranchs are an odd assemblage of molluscs that include sea slugs, sea hares, sea butterflies, and canoe shells. They are nearly all marine; most of them are shallow-water forms, hiding under stones and seaweed; a few are pelagic. Currently nine or more orders of opisthobranchs are recognized. Opisthobranchs show partial or complete detorsion; thus the anus and gill (if present) are displaced to the right side or rear of the body. Clearly, the fouling problem is obviated if the anus is moved away from the head toward the posterior. Two pairs of tentacles are usually found, and the second pair is often further modified (rhinophores, Figure 16-21), with platelike folds that apparently increase the area for chemoreception. Their shell is typically reduced or absent. All are monoecious.
 |
| Figure 16-21A, The sea hare, Aplysia dactylomela, crawls and swims across a tropical seagrass bed, assisted by large, winglike arapodia, here curled above the body. B, When attacked, sea hares squirt a copious protective secretion from their “purple gland” in the mantle cavity. |
 |
| Figure 16-22 Phyllidia ocellata, a nudibranch. Like other Phyllidia spp., it has a hard body with dense calcareous spicules and bears its gills along the sides, between its mantle and foot. |
Sea hares (Aplysia Figure 16-21), have large, earlike anterior tentacles and vestigial shells. In pteropods or sea butterflies (Cavolina and Clione) the foot is modified into fins for swimming; thus, they are pelagic and form a part of the plankton fauna.
Nudibranchs are carnivorous and often brightly colored (Figure 16-22). Plumed sea slugs (Aeolidae) which live on sea anemones and hydroids, have elongate papillae (cerata) covering their back. They ingest their prey’s nematocysts and transport the nematocysts undischarged to the tips of their cerata. There the nematocysts are placed in cnidosacs that open to the outside, and the aeolid can use its highjacked nematocysts for its own defense. Hermissenda is one of the more common West Coast nudibranchs.
Pulmonates: Pulmonates show some detorsion and include land and most freshwater snails and slugs (and a few brackish and saltwater forms). They have lost their ancestral ctenidia, but their vascularized mantle wall has become a lung, which fills with air by contraction of the mantle floor (some aquatic species have developed secondary gills in the mantle cavity). The anus and nephridiopore open near the pneumostome, and waste is expelled forcibly with air or water from the lung. They are monoecious. Aquatic species have one pair of nonretractile tentacles, at the base of which are the eyes; land forms have two pairs of tentacles, with the posterior pair bearing the eyes (Figure 16-23). Among the thousands of land species, some of the most familiar American forms are Helix, Polygyra, Succinea, Anguispira, Zonitoides, Limax, and Agriolimax. Aquatic forms are represented by Helisoma, Lymnaea, and Physa. Physa is a left-handed (sinistral) snail.
 |
| Figure 16-23 A, Pulmonate land snail. Note two pairs of tentacles; the second, larger pair bears the eyes. B, Banana slug, Ariolimax columbianus. Note pneumostome. |
Bivalvia are also known as Pelecypoda (pel-e-sip´o-da), or “hatchetfooted” animals, as their name implies (Gr. pelekys, hatchet, + pous, podos, foot). They are bivalved molluscs that include mussels, clams, scallops, oysters, and shipworms (Figures 16-24 to 16-27) and they range in size from tiny seed shells 1 to 2 mm in length to giant South Pacific clams Tridacna, which may reach more than 1 m in length and as much as 225 kg (500 pounds) in weight (see Figure 16-35). Most bivalves are sedentary filter feeders that depend on ciliary currents produced by the gills to bring in food materials. Unlike gastropods, they have no head, no radula, and very little cephalization. Most bivalves are marine, but many live in brackish water and in streams, ponds, and lakes.
Form and Function Shell: Bivalves are laterally compressed, and their two shells (valves) are held together dorsally by a hinge ligament that causes the valves to gape ventrally. The valves are drawn together by adductor muscles that work in opposition to the hinge ligament (Figure 16-26C and D). The umbo is the oldest part of the shell, and growth occurs in concentric lines around it (Figure 16-26A).
 |
 |
|
| Figure 16-24 Bivalve molluscs. A, Mussels, Mytilus edulis, occur in northern oceans around the world; they form dense beds in the ntertidal zone. A host of marine creatures live protected beneath attached mussels. B, Scallops (Chlamys opercularis) swim to escape attack by starfish (Asterias rubens). When alarmed, these most agile of bivalves swim by clapping the two shell valves together |
Figure 16-25 Representing a group that has evolved from burrowing ancestors, the surface-dwelling bivalve Pecten sp. has developed sensory organs along its mantle edges (tentacles and a series of blue eyes). |
Pearl production is a by-product of a protective device used by the animals when a foreign object (grain of sand, parasite, or other) becomes lodged between the shell and mantle. The mantle secretes many layers of nacre around the irritating object (Figure 16-5). Pearls are cultured by inserting particles of nacre, usually taken from the shells of freshwater clams, between the shell and mantle of a certain species of oyster and by keeping the oysters in enclosures for several years. Meleagrina is an oyster used extensively by the Japanese for pearl culture.
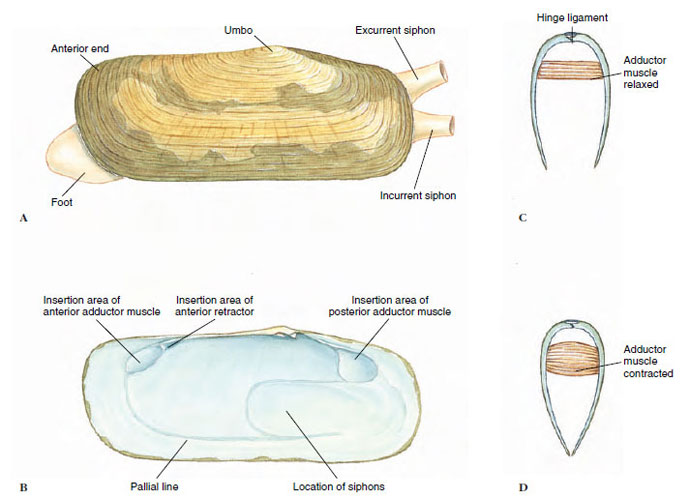 |
| Figure 16-26Tagelus plebius, stubby razor clam (class Bivalvia). A, External view of left valve. B, Inside of right shell showing scars where muscles were attached. The mantle was attached at the pallial line. C and D, Sections showing function of adductor muscles and hinge ligament. In C, the adductor muscle is relaxed, allowing the hinge ligament to pull the valves apart. In D, the adductor muscle is contracted, pulling the valves together. |
 |
| Figure 16-27A, Shipworms are bivalves that burrow in wood, causing great damage to unprotected wooden hulls and piers. B, The two small, anterior valves, seen at left, are used as rasping organs to extend the burrow. |
Body and Mantle: The visceral mass is suspended from the dorsal midline, and the muscular foot is attached to the visceral mass anteroventrally. The ctenidia hang down on each side, each covered by a fold of the mantle. The posterior edges of the mantle folds are modified to form dorsal excurrent and ventral incurrent openings (Figure 16-28A). In some marine bivalves the mantle is drawn out into long muscular siphons that allow the clam to burrow into the mud or sand and extend the siphons to the water above (Figure 16-28B to D).
 |
| Figure 16-28 Adaptations of siphons in bivalves. A, In northwest ugly clams Entodesma saxicola, incurrent and excurrent siphons are clearly visible. B to D, In many marine forms the mantle is drawn out into long siphons. In A, B, and D, the incurrent siphon brings in both food and oxygen. In C, Yoldia, the siphons are respiratory; long ciliated palps feel about over the mud surface and convey food to the mouth. |
 |
| Figure 16-29 Evolution of bivalve ctenidia. By a great lengthening of individual filaments, ctenidia became adapted for filter feeding and separated the incurrent chamber from the excurrent, suprabranchial chamber. |
Locomotion: Bivalves initiate movement by extending a slender muscular foot between the valves (Figure 16-28D). Blood is pumped into the foot, causing it to swell and to act as an anchor in the mud or sand, then longitudinal muscles contract to shorten the foot and pull the animal forward.
Scallops and file shells are able to swim with a jerky motion by clapping their valves together to create a sort of jet propulsion. The mantle edges can direct the stream of expelled water, so that the animals can swim in virtually any direction (Figure 16-24).
Gills: Gaseous exchange occurs through both mantle and gills. Gills of most bivalves are highly modified for filter feeding; they are derived from primitive ctenidia by a great lengthening of filaments on each side of the central axis (Figure 16-29). As ends of long filaments became folded back toward the central axis, ctenidial filaments took the shape of a long, slender W. Filaments lying beside each other became joined by ciliary junctions or tissue fusions, forming platelike lamellae with many vertical water tubes inside. Thus water enters the incurrent siphon, propelled by ciliary action, then enters the water tubes through pores between the filaments in the lamellae, proceeds dorsally into a common suprabranchial chamber (Figure 16-30), and then out the excurrent aperture.
 |
| Figure 16-35 Clam (Tridacna gigas) lies buried in coral rock with greatly enlarged siphonal area visible. These tissues are richly colored and bear enormous numbers of symbiotic single-celled algae (zooxanthellae) that provide much of the clam’s nutriment. |
Feeding: Most bivalves are filter feeders. Respiratory currents bring both oxygen and organic materials to the gills where ciliary tracts direct them to the tiny pores of the gills. Gland cells on the gills and labial palps secrete copious amounts of mucus, which entangles particles suspended in water going through gill pores. These mucous masses slide down the outside of the gills toward food grooves at the lower edge of the gills (Figure 16-31). Heavier particles of sediment drop off the gills as a result of gravitational pull, but smaller particles travel along the food grooves toward the labial palps. The palps, being also grooved and ciliated, direct the mucous mass into the mouth.
Some bivalves, such as Nucula and Yoldia, are deposit feeders and have long proboscides attached to the labial palps (Figure 16-28C). These can be protruded onto sand or mud to collect food particles, in addition to particles attracted by gill currents.
Shipworms (Figure 16-27) burrow in wood and feed on particles they excavate. Symbiotic bacteria live in a special organ in the bivalve and produce cellulase to digest wood. Other bivalves such as giant clams gain much of their nutrition from the photosynthetic products of symbiotic algae living in their mantle tissue (Figure 16-35).
Septibranchs, another group of bivalves, draw small crustaceans or bits of organic debris into the mantle cavity by sudden inflow of water created by the pumping action of a muscular septum in the mantle cavity.
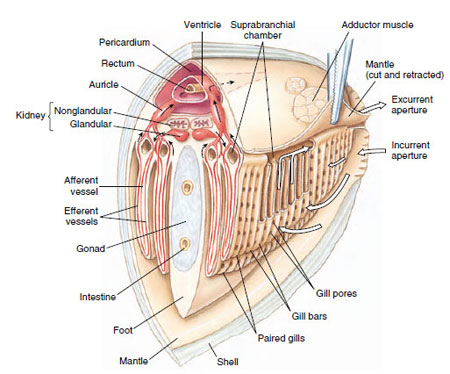 |
| Figure 16-30 Section through heart region of a freshwater clam to show relation of circulatory and respiratory systems. Respiratory water currents: water is drawn in by cilia, enters gill pores, and then passes up water tubes to suprabranchial chambers and out excurrent aperture. Blood in gills exchanges carbon dioxide for oxygen. Blood circulation: ventricle pumps blood forward to sinuses of foot and viscera, and posteriorly to mantle sinuses. Blood returns from mantle to auricles; it returns from viscera to the kidney, and then goes to the gills, and finally to the auricles. |
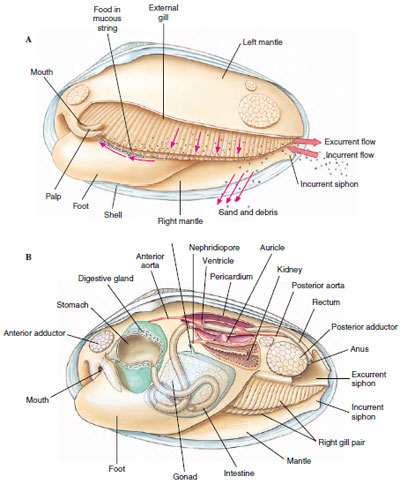 |
| Figure 16-31 A, Feeding mechanism of freshwater clam. Left valve and mantle are removed. Water enters the mantle cavity posteriorly and is drawn forward by ciliary action to the gills and palps. As water enters the tiny openings of the gills, food particles are sieved out and caught in strings of mucus that are carried by cilia to the palps and directed to the mouth. Sand and debris drop into the mantle cavity and are removed by cilia. B, Clam anatomy. |
Internal Structure and Function
The floor of the stomach of filter-feeding bivalves is folded into ciliary tracts for sorting a continuous stream of particles. A cylindrical style sac opening into the stomach secretes a gelatinous rod called a crystalline style, which projects into the stomach and is kept whirling by means of cilia in the style sac (Figure 16-32). Rotation of the style helps to dissolve its surface layers, freeing digestive enzymes (especially amylase) that it contains, and to roll the mucous food mass. Dislodged particles are sorted, and suitable ones are directed to the digestive gland or engulfed by amebocytes. Further digestion is intracellular.
The three-chambered heart, which lies in the pericardial cavity (Figure 16-31), has two auricles and a ventricle and beats slowly, ranging from 0.2 to 30 times per minute. Part of the blood is oxygenated in the mantle and returns to the ventricle through the auricles; the rest circulates through sinuses and passes in a vein to the kidneys, from there to the gills for oxygenation, and back to the auricles.
A pair of U-shaped kidneys (nephridial tubules) lies just ventral and posterior to the heart (Figure 16-31B). The glandular portion of each tubule opens into the pericardium; the bladder portion empties into the suprabranchial chamber.
The nervous system consists of three pairs of widely separated ganglia connected by commissures and a system of nerves. Sense organs are poorly developed. They include a pair of statocysts in the foot, a pair of osphradia of uncertain function in the mantle cavity, tactile cells, and sometimes simple pigment cells on the mantle. Scallops (Pecten, Chlamys) have a row of small blue eyes along each mantle edge (Figure 16-25). Each eye has a cornea, lens, retina, and pigmented layer. Tentacles on the margin of the mantle of Pecten (Figure 16-25) and Lima have tactile and chemoreceptor cells.
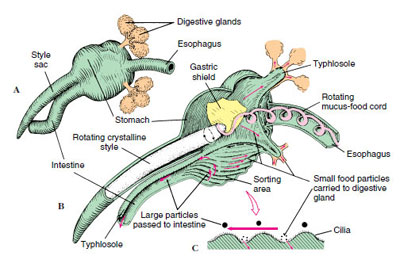 |
| Figure 16-32 Stomach and crystalline style of ciliary-feeding clam. A, External view of stomach and style sac. B, Transverse section showing direction of food movements. Food particles in incoming water are caught in a cord of mucus that is kept rotating by the crystalline style. Ridged sorting areas direct large particles to the intestine and small food particles to digestive glands. C, Sorting action of cilia. |
Reproduction and Development
Sexes are usually separate. Gametes are discharged into the suprabranchial chamber to be carried out with the excurrent flow. An oyster may produce 50 million eggs in a single season. In most bivalves fertilization is external. The embryo develops into trochophore, veliger, and spat stages (Figure 16-33).
In most freshwater clams fertilization is internal. Eggs drop into the water tubes of the gills where they are fertilized by sperm entering with the incurrent flow. They develop there into a bivalved glochidium larva stage, which is a specialized veliger (Figure 16-34). When discharged, glochidia are carried by water currents, and if they come in contact with a passing fish, they attach to its gills or skin and live as parasites for several weeks. Then they sink to the bottom to begin independent lives. Larval “hitchhiking” helps distribute a form whose locomotion is very limited.
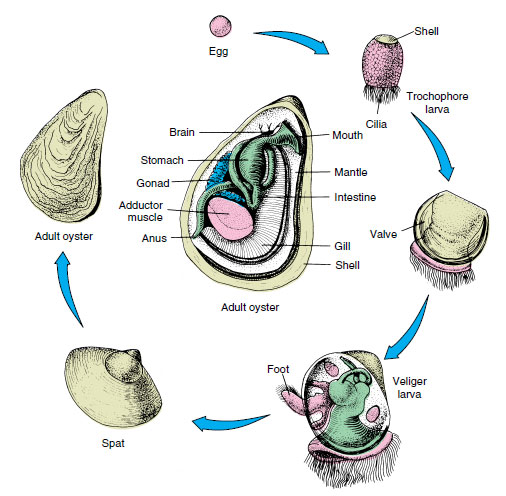 |
| Figure 16-33 Life cycle of oysters. Oyster larvae swim about for approximately 2 weeks before settling down for attachment to become spats. Oysters take about 4 years to grow to commercial size. |
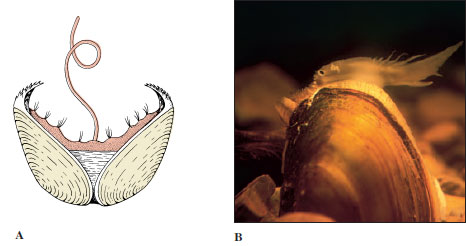 |
| Figure 16-34A, Glochidium, or larval form, for some freshwater clams. When larvae are released from brood pouch of mother, they may become attached to a fish’s gill by clamping their valves closed. They remain as parasites on the fish for several weeks. Their size is approximately 0.3 mm. B, Some clams have adaptations that help their glochidia find a host. The mantle edge of this female pocketbook mussel (Lampsilis ovata) mimics a small minnow, complete with eye. When a smallmouth bass comes to dine, it gets doused with glochidia. |
Boring: Many pelecypods can burrow into mud or sand, but some have evolved a mechanism for burrowing into much harder substances, such as wood or stone.
Teredo, Bankia, and some other genera are called shipworms. They can be very destructive to wooden ships and wharves. These strange little clams have a long, wormlike appearance, with a pair of slender posterior siphons that keep water flowing over the gills, and a pair of small globular valves on the anterior end with which they burrow (Figure 16-27). The valves have microscopic teeth that function as very effective wood rasps. The animals extend their burrows with an unceasing rasping motion of the valves. This motion sends a continuous flow of fine wood particles into the digestive tract where they are attacked by cellulase produced by symbiotic bacteria. Interestingly, these bacteria also fix nitrogen, an important property for their hosts, which live on a diet (wood) high in carbon but deficient in nitrogen.
Some clams bore into rock. The piddock (Pholas) bores into limestone, shale, sandstone, and sometimes wood or peat. It has strong valves that bear spines, which it uses to cut away the rock gradually while anchoring itself with its foot. Pholas may grow to 15 cm long and make rock burrows up to 30 cm long.
Class Cephalopoda
Cephalopoda (Gr. kephale, head, + pous, podos, foot) include squids, octopuses, nautiluses, devilfish, and cuttlefish. All are marine, and all are active predators.
The modified foot is concentrated in the head region. It takes the form of a funnel for expelling water from the mantle cavity, and the anterior margin is drawn out into a circle or crown of arms or tentacles.
Cephalopods range upward in size from 2 or 3 cm. The common squid of markets, Loligo, is about 30 cm long. The giant squid Architeuthis is the largest invertebrate known.
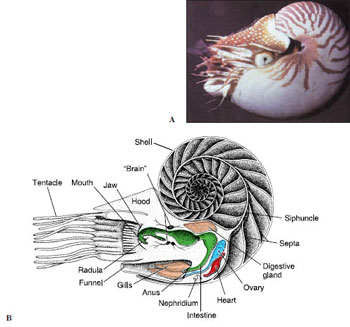 |
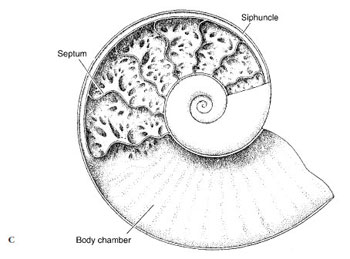 |
| Figure 16-36 Nautilus, a cephalopod. A, Live Nautilus, feeding on a fish. B, Longitudinal section, showing gasfilled chambers of shell, and diagram of body structure. C, Longitudinal section through shell of an ammonoid. |
Fossil records of cephalopods go back to Cambrian times. The earliest shells were straight cones; others were curved or coiled, culminating in the coiled shell similar to that of the modern Nautilus, the only remaining member of the once flourishing nautiloids (Figure 16-36). Cephalopods without shells or with internal shells (such as octopuses and squids) apparently evolved from some early straightshelled ancestor. Many ammonoids, which are extinct, had quite elaborate shells (Figure 16-36C).
The natural history of some cephalopods is fairly well known. They are marine animals and appear sensitive to the degree of salinity. Few are found in the Baltic Sea, where the water has a low salt content. Cephalopods are found at various depths. Octopuses are often seen in the intertidal zone, lurking among rocks and crevices, but occasionally they are found at great depths. The more active squids are rarely found in very shallow water, and some have been taken at depths of 5000 m. Nautilus is usually found near the bottom in water 50 to 560 m deep, near islands in the southwestern Pacific.
Form and Function
Shell: Although early nautiloid and ammonoid shells were heavy, they were made buoyant by a series of gas chambers, as is that of Nautilus (Figure 16-36B), enabling the animal to swim while carrying its shell. The shell of Nautilus, although coiled, is quite different from that of a gastropod. The shell is divided by transverse septa into internal chambers (Figure 16-36B). The living animal inhabits only the last chamber. As it grows, it moves forward, secreting behind a new septum. The chambers are connected by a cord of living tissue called a siphuncle, which extends from the visceral mass. Cuttlefishes (Figure 16-37) also have a small, curved shell, but it is entirely enclosed by the mantle. In squids most of the shell has disappeared, leaving only a thin, horny strip called a pen, which is enclosed by the mantle. In Octopus (Gr. oktos, eight, + pous, podos, foot) the shell has disappeared entirely.
 |
| Figure 16-37 Cuttlefish, Sepia latimanus, has an internal shell familiar to keepers of caged birds as “cuttlebone. |
Locomotion: Cephalopods swim by forcefully expelling water from the mantle cavity through a ventral funnel (or siphon)—a sort of jet propulsion. The funnel is mobile and can be pointed forward or backward to control direction; the force of water expulsion controls speed.
Squids and cuttlefishes are excellent swimmers. The squid body is streamlined and built for speed (Figure 16-38). Cuttlefishes swim more slowly. The lateral fins of squids and cuttlefishes serve as stabilizers, but they are held close to the body for rapid swimming.
Nautilus is active at night; its gasfilled chambers keep the shell upright. Although not as fast as squids, it moves surprisingly well.
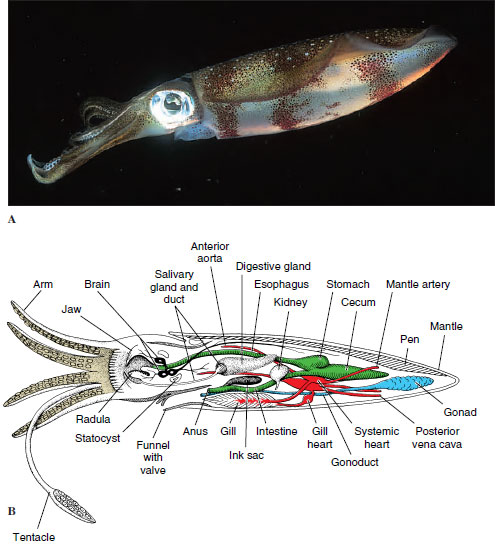 |
| Figure 16-38 A, Reef squid Sepioteuthis lessoniana. B, Lateral view of squid anatomy, with the left half of the mantle removed. |
Octopus has a rather globular body and no fins (Figure 16-1E). An octopus can swim backward by spurting jets of water from its funnel, but it is better adapted to crawling about over the rocks and coral, using suction discs on its arms to pull or to anchor itself. Some deep-water octopods have the arms webbed like an umbrella and swim in a medusa-like fashion.
Internal Features: The active habits of cephalopods are reflected in their internal anatomy, particularly their respiratory, circulatory, and nervous systems.
Respiration and Circulation: Except for nautiloids, cephalopods have one pair of gills. Because ciliary propulsion would not circulate enough water for their high oxygen requirements, there are no cilia on the gills. Instead, radial muscles in the mantle wall compress the wall and enlarge the mantle cavity, drawing water inside. Strong circular muscles contract and expel water forcibly through the funnel. A system of one-way valves prevents water from being taken in through the funnel and expelled around the mantle margin.
Likewise, the open circulatory system of their ancestral molluscs would be inadequate for cephalopods. Their circulatory system consists of a closed network of vessels, and capillaries conduct blood through the gill filaments. Furthermore, the molluscan plan of circulation places the entire systemic circulation before the blood reaches the gills (in contrast to vertebrates, in which the blood leaves the heart and goes directly to the gills or lungs). This functional problem was solved by the development of accessory or branchial hearts (Figure 16-38B) at the base of each gill to increase the pressure of the blood going through the capillaries there.
Nervous and Sensory Systems: Nervous and sensory systems are more elaborate in cephalopods than in other molluscs. The brain, the largest in any invertebrate, consists of several lobes with millions of nerve cells. Squids have giant nerve fibers (among the largest known in the animal kingdom), which are activated when the animal is alarmed and that initiate maximal contractions of the mantle muscles for a speedy escape.
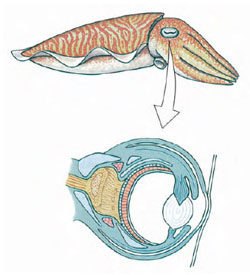 |
| Figure 16-39 Eye of a cuttlefish (Sepia). The structure of cephalopod eyes shows a high degree of convergent evolution with eyes of vertebrates. |
Sense organs are well developed. Except for Nautilus, which has relatively simple eyes, cephalopods have highly complex eyes with cornea, lens, chambers, and retina (Figure 16-39). Orientation of the eyes is controlled by the statocysts, which are larger and more complex than in other molluscs. The eyes are held in a constant relation to gravity, so that the slit-shaped pupils are always in a horizontal position. Octopods are apparently colorblind but can be taught to discriminate between shapes—for example, a square and a rectangle—and to remember such a discrimination for a considerable time. Experimenters find it easy to modify their behavior patterns by devices of reward and punishment. They are capable of observational learning; when one octopus observes another being rewarded by making a correct choice, the observer learns which choice is rewarded and consistently makes the same selection when given the opportunity.
Octopods use their arms for tactile exploration and can discriminate between textures by feel but apparently not between shapes. Their arms are well supplied with both tactile and chemoreceptor cells. Cephalopods seem to lack a sense of hearing.
Communication: Little is known of social behavior of nautiloids or deepwater cephalopods, but inshore and littoral forms such as Sepia, Sepioteuthis, Loligo, and Octopus have been studied extensively. Although their tactile sense is well developed and they have some chemical sensitivity, visual signals are the predominant means of communication. These signals consist of a host of movements of the arms, fins, and body, as well as many color changes. The movements may range from minor body motions to exaggerated spreading, curling, raising, or lowering of some or all of the arms. Color changes are effected by chromatophores, cells in the skin that contain pigment granules. Tiny muscle cells surround each elastic chromatophore, whose contractions pull the cell boundary of the chromatophore outward, causing it to expand greatly. As the cell expands, the pigment becomes dispersed, changing the color pattern of the animal. When the muscles relax, the chromatophores return to their original size and pigment becomes concentrated again. By means of the chromatophores, which are under nervous and probably hormonal control, an elaborate system of changes in color and pattern is possible, including general darkening or lightening; flushes of pink, yellow, or lavender; and the formation of bars, stripes, spots, or irregular blotches. These colors may be used variously as danger signals, as protective coloring, in courtship rituals, and probably in other ways.
By assuming different color patterns of different parts of the body, a squid can transmit three or four different messages simultaneously to different individuals and in different directions, and it can instantaneously change any or all of the messages. Probably no other system of communication in invertebrates can convey so much information so rapidly.
Deep-water cephalopods may have to depend more on chemical or tactile senses than their littoral or surface cousins, but they also produce their own type of visual signals, for they have evolved many elaborate luminescent organs.
Most cephalopods other than nautiloids have another protective device. An ink sac that empties into the rectum contains an ink gland that secretes sepia, a dark fluid containing the pigment melanin, into the sac. When the animal is alarmed, it releases a cloud of ink, which may hang in the water as a blob or be contorted by water currents. The animal quickly departs from the scene, leaving the ink as a decoy to the predator.
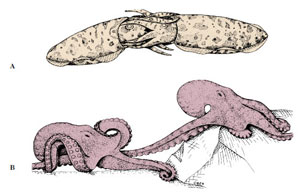 |
| Figure 16-40 Copulation in cephalopods. A, Mating cuttlefishes. B, Male octopus uses modified arm to deposit spermatophores in female mantle cavity to fertilize her eggs. Octopuses often tend their eggs during development. |
Reproduction: Sexes are separate in cephalopods. In the male seminal vesicle the spermatozoa are encased in spermatophores and stored in a sac that opens into the mantle cavity. One arm of the adult male is modified as an intromittent organ, called a hectocotylus, used to pluck a spermatophore from his own mantle cavity and insert it into the mantle cavity of a female near the oviduct opening (Figure 16-40). Before copulation males often undergo color displays, apparently directed against rival males. Eggs are fertilized as they leave the oviduct and are then usually attached to stones or other objects. Some octopods tend their eggs. Argonauta, the paper nautilus, secretes a fluted “shell,” or capsule, in which she broods her eggs.
The large yolky eggs undergo meroblastic cleavage. During embryonic development, the head and foot become indistinguishable. The ring around the mouth, which bears the arms, or tentacles, may be derived from the anterior part of the foot. A juvenile hatches from the egg; no free-swimming larva exists in cephalopods.
Major Groups of Cephalopods
There are three subclasses of cephalopods: Nautiloidea, which have two pairs of gills; the entirely extinct Ammonoidea; and Coleoidea, which have one pair of gills. Nautiloidea populated the Paleozoic and Mesozoic seas, but there survives only one genus, Nautilus (see Figure 16-36), of which there are five or six species. Nautilus’ head, with its 60 to 90 or more tentacles, can be extended from the opening of the body compartment of the shell. Its tentacles have no suckers but are made adhesive by secretions. They are used in searching for, sensing, and grasping food. Beneath its head is the funnel. Mantle, mantle cavity, and visceral mass are sheltered by the shell.
Ammonoids were widely prevalent in the Mesozoic era but became extinct by the end of the Cretaceous period. They had chambered shells analogous to nautiloids, but the septa were more complex, and the septal sutures (where the septa contact the inside of the shell) were frilled (compare shells in Figure 16-36B and C). The reasons for their extinction remain a mystery. Present evidence suggests that they were gone before the asteroid bombardment at the end of the Cretaceous period, and some nautiloids, which some ammonoids closely resembled, survive to the present.
Subclass Coleoidea includes all living cephalopods except Nautilus. There are four orders of coleoids. Members of order Sepioidea (cuttlefishes and their relatives) have a rounded or compressed, bulky body bearing fins (Figure 16-37). They have eight arms and two tentacles. Both arms and tentacles have suckers, but tentacles bear suckers only at their ends (Figure 16-37). Members of order Teuthoidea (squids, Figure 16-38) have a more cylindrical body but also have eight arms and two tentacles. Order Vampyromorpha (vampire squid) contains only a single, deep-water species. Members of order Octopoda have eight arms and no tentacles (see Figure 16-1E). Their bodies are short and saclike, with no fins. The suckers in squids are stalked (pendunculated), with horny rims bearing teeth; in octopuses the suckers are sessile and have no horny rims.




