Osteichthyes: Bony Fishes
Osteichthyes:
Bony Fishes
Origin, Evolution, and Diversity
In the early to middle Silurian, a lineage of fishes with bony endoskeletons gave rise to a clade of vertebrates that contains 96% of the living fishes and all of the living tetrapods. The fishes of this clade have traditionally been termed “bony fishes” (Osteichthyes), because it was originally believed these were the only fishes with bony skeletons. Although it is now recognized that bone occurs in many other early fishes (ostracoderms, placoderms, and acanthodians), bony fishes and their tetrapod descendants are united by the presence of endochondral bone (bone that replaces cartilage developmentally), the presence of lungs or a swim bladder derived from the gut, and several cranial and dental characters. Because the traditional usage of Osteichthyes does not describe a monophyletic (natural) group (Figure 26-2), most recent classifications, including the one presented at the end of this section, do not recognize this term as a valid taxon. Rather, it is used as a term of convenience to describe a group of vertebrates with endochondral bone that are conventionally termed fishes.
Fossils of the earliest bony fishes show similarities in several craniopharyngeal structures, including a bony operculum and branchiostegal rays, with acanthodians (Figure 25-17), indicating they likely descended from a common ancestor. By the middle of the Devonian the bony fishes already had radiated extensively into two major lin-eages, with adaptations that fitted them for every aquatic habitat except the most inhospitable. One of these lineages, the ray-finned fishes (class Actinopterygii), includes the modern bony fishes (Figure 26-15), the most speciose of living vertebrates. A second lineage, the lobe-finned fishes (class Sarcopterygii), is represented today by only seven fishlike vertebrates, the lungfishes and the coelacanth (Figures 26-22 and 26-23). The evolutionary history of this lineage is of particular interest because it gave rise to the land vertebrates (tetrapods).
Several key adaptations contributed to their radiation. Bony fishes have an operculum over the gill composed of bony plates and attached to a series of muscles. This feature increased respiratory efficiency because the outward rotation of the operculum created a negative pressure so that water would be drawn across the gills, as well as pushed across by the mouth pump. A gas-filled derivative of the esophagus provided an additional means of gas exchange in hypoxic waters and an efficient means for achieving neutral buoyancy. Progressive specialization of jaw musculature and skeletal elements involved in feeding is another key feature in their evolution.
Characteristics of Class Actinopterygii
Class Actinopterygii: Ray-Finned Fishes
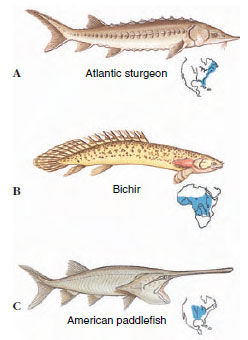
The ray-finned fishes are an enormous assemblage containing all of our familiar bony fishes—more than 23,600 species. The earliest actinopterygians, known as palaeoniscids (pay´lee-onis ´ids), were small fishes, with large eyes, a heterocercal tail (Figure 26-16), and thick, interlocking scales with an outer layer of ganoin (Figure 26-18). These fishes had a single dorsal fin and numerous bony rays derived from scales stacked end to end, distinctively different in appearance from the lobefinned fishes with which they shared the Devonian waters. Palaeoniscids are represented by fossil fragments as early as the late Silurian, and flourished throughout the late Paleozoic era, during the same period that ostracoderms, placoderms, and acanthodians disappeared and sarcopterygians declined in abundance (Figure 26-1). This suggests the morphological specializations evolving in the actinopterygian lineage gave them ecological superiority over most other fishes.
From these earliest ray-finned
fishes, two major groups appeared.
Those with the most primitive characteristics
are the chondrosteans (Gr. chondros, cartilage, + osteon, bone),
represented today by the freshwater
and anadromous sturgeons, paddlefishes,
and bichirs (Figure 26-19). The
chondrosteans show many characters
similar to their paleoniscid ancestors,
including a heterocercal tail and
ganoid scutes or scales. The bichir Polypterus (Gr. poly, many, + pteros,
winged) of African waters is an interesting
relict with lungs and other primitive
characters that make it resemble
palaeoniscids more than any other living
fish.
The second major group of rayfinned fishes to emerge from the palaeoniscid stock were the neopterygians (Gr. neos, new, + pteryx, fin). The neopterygians appeared in the late Permian and radiated extensively during the Mesozoic era (Figure 26-1). During the Mesozoic one lineage gave rise to a secondary radiation that led to the modern bony fishes, the teleosts. There are two surviving genera of early neopterygians, the bowfin Amia (Gr. tunalike fish) of shallow, weedy waters of the Great Lakes and Mississippi Valley, and the gars Lepisosteus (Gr. lepidos, scale, + osteon, bone) of eastern and southern North America (Figure 26-20). The seven species of gars are large, ambush predators with elongate bodies and jaws filled with needlelike teeth. Gars and bowfin may gulp air to surface, filling their vascularized swim bladder with air to supplement oxygen obtained in the gills.
The major lineage of neopterygians are the teleosts (Gr. teleos, perfect, + osteon, bone), the modern bony fishes (Figure 26-15). Teleost diversity is astounding, with about 23,600 described species, representing about 96% of all living fishes or about half of all vertebrates (Figure 26-21). In addition, it has been estimated there are an additional 5,000 to 10,000 undescribed species. Although most of the 200 or so new species of teleosts described each year are from poorly sampled areas such as South America or deep oceanic waters, several new species are described each year from areas as well known as the fresh waters of North America! Teleosts range in size from 10 mm adult gobies to the 17 m oarfish and the 900 kg, 4.5 m blue marlin (Figure 26-21). These fishes occupy almost every conceivable habitat, from elevations up to 5200 m in Tibet to 8000 m below the surface of the ocean. Some species live in hot springs at 44° C, while others live under the Antarctic ice at −2° C. They may live in lakes with salt concentrations three times that of seawater, caves of total darkness, swamps devoid of oxygen, or even make extended excursions onto land, as do the mudskippers (Figure 26-21).
Several morphological trends in the teleost lineage allowed them to diversify into this truly incredible variety of habitats and forms. The heavy dermal armor of primitive ray-finned fishes was replaced by light, thin, flexible cycloid and ctenoid scales (Figure 26-18). Some teleosts, such as most eels and catfishes, completely lack scales. The increased mobility and speed that resulted from the loss of the heavy armor improved predator avoidance and food getting. Changes in the fins of teleosts increased maneuverability and speed and allowed fins to serve a variety of other functions. The symmetrical shape of the homocercal tail (Figure 26-16) of most teleosts focused musculature contractions on the tail, resulting in greater speed. The dorsal fin shifted from a fixed keel that primarily prevented rolling, to a flexible and highly specialized structure in advanced teleosts (Figure 26-15).
These changes in the morphology of the fins were useful for camouflage, braking and other complex movements, streamlining, and social communication. Bizarre modifications of the dorsal fin include the lure of anglerfishes, venom-delivering spines of scorpionfishes, and the suctorial disc of sharksuckers (Figure 26-21). In addition, the swim bladder shifted from primarily respiratory to buoyancy in function. The teleost lineage demonstrated an increasingly fine control of gas resorption and secretion in the swim bladder. Control of buoyancy likely coevolved with fin modifications to improve locomotion. Finally, greater feeding efficiency was brought about by several anatomical modifications. Changes in the jaw suspension enabled the orobranchial cavity to expand rapidly, creating a highly sophisticated suction device. Rapid jaw protrusion was made possible by sliding the upper jaw forward, increasing final attack velocity by 39% to 89%. The gill arches of many teleosts diversified into powerful pharyngeal jaws for chewing, grinding, and crushing. With so many separate innovations to work with, it is not surprising the teleosts have become the most diverse of fishes.
Characteristics of Class Sarcopterygii
Class Sarcopterygii: Lobe- Finned Fishes
The lobe-finned fishes are today represented by only seven species: six species (three genera) of lungfishes and the coelacanth—survivors of a group once abundant during the Devonian period of the Paleozoic (Figures 26-22 and 26-23).
All early sarcopterygians had lungs as well as gills, and a tail of the heterocercal type. However, during the Paleozoic the orientation of the vertebral column changed so that the tail became symmetrical, with the median dorsal and ventral fins displaced posteriorly to form one continuous, flexible fin around the tail. This type of tail is called diphycercal (Figure 26-16). The strong, fleshy, paired lobed fins of the sarcopterygians (pectoral and pelvic) may have been used much like four legs to scuttle along the bottom. They had powerful jaws and their skin was covered with heavy scales that consisted of a dentine-like material called cosmine overlaid by a thin enamel.
Of the three surviving genera of lungfishes, most similar to early forms is Neoceratodus (Gr. neos, new, + keratos, horn, + odes, form), the living Australian lungfish, which may attain a length of 1.5 m (Figure 26-22). This lungfish, unlike its relatives, normally relies on gill respiration, and cannot survive long out of water. The South American lungfish Lepidosiren (L. lepidus, pretty, + siren, mythical mermaid) and the African lungfish Protopterus (Gr. protos, first, + pteron, wing) can live out of water for long periods of time. Protopterus lives in African streams and ponds that may dry during the dry season, with their mud beds baked hard by the hot tropical sun. The fish burrows down at the approach of the dry season and secretes a copious slime that is mixed with mud to form a hard cocoon in which it estivates until the rains return. Surprisingly little is known about the ecology of the South American lungfish Lepidosiren.
Coelacanths and rhipidistians collectively have been termed crossopterygians, but this group is considered polyphytetic and no longer is recognized by most classifications. The rhipidistians flourished in the late Paleozoic era and then became extinct. Rhipidistians are of special importance because they include the ancestors of the tetrapods (and, in cladistic terms, are therefore a paraphyletic group). The coelacanths also arose in the Devonian period, radiated somewhat, and reached their evolutionary peak in the Mesozoic era. At the end of the Mesozoic era they nearly disappeared but left one remarkable surviving species, Latimeria chalumnae (named for M. Courtenay- Latimer, South African museum director) (Figure 26-23). Since the last coelacanths were believed to
have
become extinct 70 million years ago,
the astonishment of the scientific
world may be imagined when the
remains of a coelacanth were found
on a dredge off the coast of South
Africa in 1938. An intensive search to locate more specimens was successful
off the coast of the Comoro Islands.
There fishermen occasionally catch
them at great depths with hand lines,
providing specimens for research.
This was believed to be the only population
of Latimeria until 1998, when
the scientific world was again surprised
by the capture of a coelacanth,
possibly representing a new species,
in Indonesia, 10,000 km from the
Comoros!
The “modern” marine coelacanth is a descendant of the Devonian freshwater stock. The tail is of the diphycercal type (Figure 26-16) but possesses a small lobe between the upper and lower caudal lobes, producing a three-pronged structure (Figure 26-23).
Coelacanths are a deep metallic blue with irregular white or brassy flecks, providing camouflage against the dark lava-cave reefs they inhabit. Young are born fully formed after hatching internally from eggs 9 cm in diameter—the largest among bony fishes.
Origin, Evolution, and Diversity
In the early to middle Silurian, a lineage of fishes with bony endoskeletons gave rise to a clade of vertebrates that contains 96% of the living fishes and all of the living tetrapods. The fishes of this clade have traditionally been termed “bony fishes” (Osteichthyes), because it was originally believed these were the only fishes with bony skeletons. Although it is now recognized that bone occurs in many other early fishes (ostracoderms, placoderms, and acanthodians), bony fishes and their tetrapod descendants are united by the presence of endochondral bone (bone that replaces cartilage developmentally), the presence of lungs or a swim bladder derived from the gut, and several cranial and dental characters. Because the traditional usage of Osteichthyes does not describe a monophyletic (natural) group (Figure 26-2), most recent classifications, including the one presented at the end of this section, do not recognize this term as a valid taxon. Rather, it is used as a term of convenience to describe a group of vertebrates with endochondral bone that are conventionally termed fishes.
Fossils of the earliest bony fishes show similarities in several craniopharyngeal structures, including a bony operculum and branchiostegal rays, with acanthodians (Figure 25-17), indicating they likely descended from a common ancestor. By the middle of the Devonian the bony fishes already had radiated extensively into two major lin-eages, with adaptations that fitted them for every aquatic habitat except the most inhospitable. One of these lineages, the ray-finned fishes (class Actinopterygii), includes the modern bony fishes (Figure 26-15), the most speciose of living vertebrates. A second lineage, the lobe-finned fishes (class Sarcopterygii), is represented today by only seven fishlike vertebrates, the lungfishes and the coelacanth (Figures 26-22 and 26-23). The evolutionary history of this lineage is of particular interest because it gave rise to the land vertebrates (tetrapods).
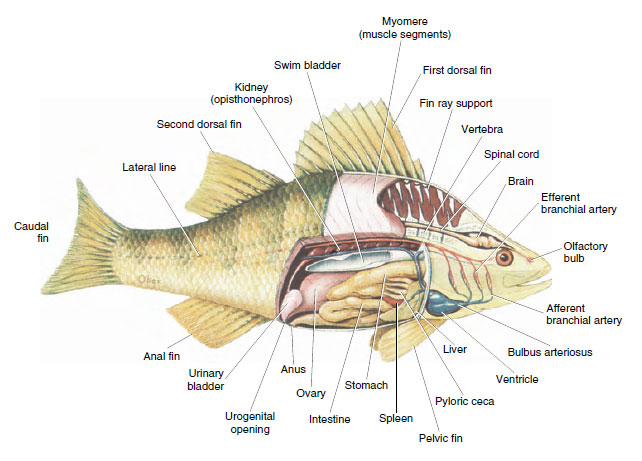 |
| Figure 26-15 Internal anatomy of the yellow perch Perca flavescens, a freshwater teleost fish. |
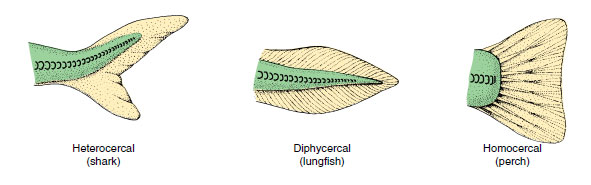 |
| Figure 26-16 Types of caudal fins among fishes. |
Several key adaptations contributed to their radiation. Bony fishes have an operculum over the gill composed of bony plates and attached to a series of muscles. This feature increased respiratory efficiency because the outward rotation of the operculum created a negative pressure so that water would be drawn across the gills, as well as pushed across by the mouth pump. A gas-filled derivative of the esophagus provided an additional means of gas exchange in hypoxic waters and an efficient means for achieving neutral buoyancy. Progressive specialization of jaw musculature and skeletal elements involved in feeding is another key feature in their evolution.
Characteristics of Class Actinopterygii
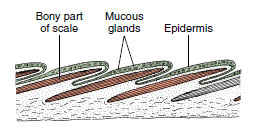 |
| Figure 26-17 Section through the skin of a bony fish, showing the overlapping scales (red). The scales lie in the dermis and are covered by epidermis. |
- Skeleton with bone of endochondral origin; caudal fin heterocercal in ancestral forms, usually homocercal in advanced forms (Figure 26-16); skin with mucous glands and embedded dermal scales (Figure 26-17); scales ganoid in ancestral forms, scales cycloid, ctenoid or absent in advanced forms (Figure 26-18)
- Paired and median fins present, supported by long dermal rays (lepidotrichia); muscles controlling fin movement within body
- Jaws present; teeth usually present with enamaloid covering; olfactory sacs do not open into mouth; spiral valve present in ancestral forms, absent in advanced forms
- Respiration primarily by gills supported by arches and covered with an operculum
- Swim bladder often present with or without a duct connecting to esophagus, usually functioning in buoyancy
- Circulation consisting of a heart with a sinus venosus, an undivided atrium, and an undivided ventricle; single circulation; typically four aortic arches; nucleated erythrocytes
- Excretory system of paired opisthonephric kidneys; sexes usually separate; fertilization usually external; larval forms may differ greatly from adults
- Nervous system of a brain with olfactory lobes, small cerebrum, optic lobes, and cerebellum; 10 pairs of cranial nerves; three pairs of semicircular canals
Class Actinopterygii: Ray-Finned Fishes
The ray-finned fishes are an enormous assemblage containing all of our familiar bony fishes—more than 23,600 species. The earliest actinopterygians, known as palaeoniscids (pay´lee-onis ´ids), were small fishes, with large eyes, a heterocercal tail (Figure 26-16), and thick, interlocking scales with an outer layer of ganoin (Figure 26-18). These fishes had a single dorsal fin and numerous bony rays derived from scales stacked end to end, distinctively different in appearance from the lobefinned fishes with which they shared the Devonian waters. Palaeoniscids are represented by fossil fragments as early as the late Silurian, and flourished throughout the late Paleozoic era, during the same period that ostracoderms, placoderms, and acanthodians disappeared and sarcopterygians declined in abundance (Figure 26-1). This suggests the morphological specializations evolving in the actinopterygian lineage gave them ecological superiority over most other fishes.
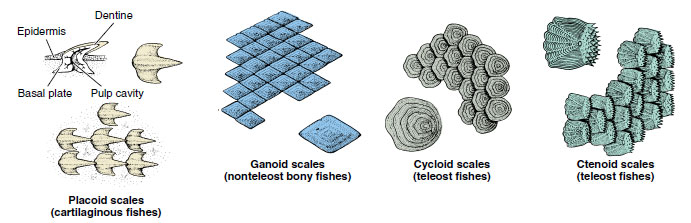 |
| Figure 26-18 Types of fish scales. Placoid scales are small, conical toothlike structures characteristic of Chondrichthyes. Diamond-shaped ganoid scales, present in early bony fishes such as the gar, are composed of layers of silvery enamel (ganoin) on the upper surface and bone on the lower. Teleosts have either cycloid or ctenoid scales. These are thin and flexible and are arranged in overlapping rows. |

| Figure 26-19 Chondrostean ray-finned fishes of the class Actinopterygii. A, Atlantic sturgeon, Acipenser oxyrhynchus (now uncommon), of Atlantic coastal rivers. B, Bichir Polypterus bichir of equatorial west Africa. It is a nocturnal predator. C, Paddlefish Polyodon spathula of the Mississippi River reaches a length of 2 m and a weight of 90 kg. |
The second major group of rayfinned fishes to emerge from the palaeoniscid stock were the neopterygians (Gr. neos, new, + pteryx, fin). The neopterygians appeared in the late Permian and radiated extensively during the Mesozoic era (Figure 26-1). During the Mesozoic one lineage gave rise to a secondary radiation that led to the modern bony fishes, the teleosts. There are two surviving genera of early neopterygians, the bowfin Amia (Gr. tunalike fish) of shallow, weedy waters of the Great Lakes and Mississippi Valley, and the gars Lepisosteus (Gr. lepidos, scale, + osteon, bone) of eastern and southern North America (Figure 26-20). The seven species of gars are large, ambush predators with elongate bodies and jaws filled with needlelike teeth. Gars and bowfin may gulp air to surface, filling their vascularized swim bladder with air to supplement oxygen obtained in the gills.
The major lineage of neopterygians are the teleosts (Gr. teleos, perfect, + osteon, bone), the modern bony fishes (Figure 26-15). Teleost diversity is astounding, with about 23,600 described species, representing about 96% of all living fishes or about half of all vertebrates (Figure 26-21). In addition, it has been estimated there are an additional 5,000 to 10,000 undescribed species. Although most of the 200 or so new species of teleosts described each year are from poorly sampled areas such as South America or deep oceanic waters, several new species are described each year from areas as well known as the fresh waters of North America! Teleosts range in size from 10 mm adult gobies to the 17 m oarfish and the 900 kg, 4.5 m blue marlin (Figure 26-21). These fishes occupy almost every conceivable habitat, from elevations up to 5200 m in Tibet to 8000 m below the surface of the ocean. Some species live in hot springs at 44° C, while others live under the Antarctic ice at −2° C. They may live in lakes with salt concentrations three times that of seawater, caves of total darkness, swamps devoid of oxygen, or even make extended excursions onto land, as do the mudskippers (Figure 26-21).
 |
| Figure 26-20 Nonteleost neopterygian fishes. A, Bowfin Amia calva. B, Longnose gar Lepisosteus osseus. The bowfin lives in the Great Lakes region and Mississippi basin. Gars are common fishes of eastern and southern North America. They frequent slow-moving streams where they may hang motionless in the water, ready to snatch passing fish. |
Several morphological trends in the teleost lineage allowed them to diversify into this truly incredible variety of habitats and forms. The heavy dermal armor of primitive ray-finned fishes was replaced by light, thin, flexible cycloid and ctenoid scales (Figure 26-18). Some teleosts, such as most eels and catfishes, completely lack scales. The increased mobility and speed that resulted from the loss of the heavy armor improved predator avoidance and food getting. Changes in the fins of teleosts increased maneuverability and speed and allowed fins to serve a variety of other functions. The symmetrical shape of the homocercal tail (Figure 26-16) of most teleosts focused musculature contractions on the tail, resulting in greater speed. The dorsal fin shifted from a fixed keel that primarily prevented rolling, to a flexible and highly specialized structure in advanced teleosts (Figure 26-15).
These changes in the morphology of the fins were useful for camouflage, braking and other complex movements, streamlining, and social communication. Bizarre modifications of the dorsal fin include the lure of anglerfishes, venom-delivering spines of scorpionfishes, and the suctorial disc of sharksuckers (Figure 26-21). In addition, the swim bladder shifted from primarily respiratory to buoyancy in function. The teleost lineage demonstrated an increasingly fine control of gas resorption and secretion in the swim bladder. Control of buoyancy likely coevolved with fin modifications to improve locomotion. Finally, greater feeding efficiency was brought about by several anatomical modifications. Changes in the jaw suspension enabled the orobranchial cavity to expand rapidly, creating a highly sophisticated suction device. Rapid jaw protrusion was made possible by sliding the upper jaw forward, increasing final attack velocity by 39% to 89%. The gill arches of many teleosts diversified into powerful pharyngeal jaws for chewing, grinding, and crushing. With so many separate innovations to work with, it is not surprising the teleosts have become the most diverse of fishes.
 |
| Figure 26-21 Diversity among teleosts. A, Blue marlin, Makaira nigricans, one of the largest teleosts. B, Mudskippers, Periophthalmus sp., make extensive excursions on land to graze on algae and capture insects; they build nests in which the young hatch and are guarded by the mother. C, Protective coloration of the flamboyant lionfish, Pterois sp., advises caution; the dorsal spines are poisonous. D, The sucking disk on the sharksucker, Echeneis naucrates, is a modification of the dorsal fin. |
Characteristics of Class Sarcopterygii
- Skeleton with bone of endochondral origin; caudal fin diphycercal in living representatives, heterocercal in ancestral forms; skin with embedded dermal scales (Figure 26-17) with a layer of dentine-like material, cosmine, in ancestral forms
- Paired and median fins present; paired fins with a single basal skeletal element and short dermal rays; muscles that move paired fins located on limb
- Jaws present; teeth are covered with true enamel and typically are crushing plates restricted to palate; olfactory sacs paired, may or may not open into mouth; intestine with spiral valve
- Gills supported by bony arches and covered with an operculum
- Swim bladder vascularized and used for respiration and buoyancy (fat-filled in the coelacanth)
- Circulation consisting of heart with a sinus venosus, two atria, a partly divided ventricle, and a conus arteriosus; double circulation with pulmonary and systemic circuits; characteristically five aortic arches
- Nervous system with olfactory lobes, a cerebrum, a cerebellum, and optic lobes; 10 pairs of cranial nerves; three pairs of semicircular canals
- Sexes separate; fertilization external or internal
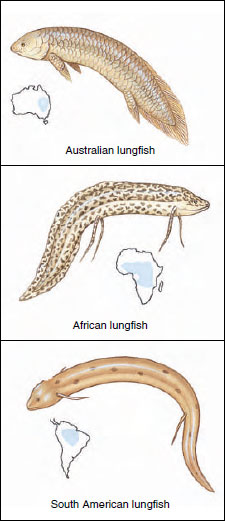 |
| Figure 26-22 Lungfishes are lobe-finned fishes of the class Sarcopterygii. The Australian lungfish Neoceratodus forsteri is the least specialized of three lungfish genera. The African lungfish Protopterus sp. is best adapted of the three for remaining dormant in mucous-lined cocoons breathing air during prolonged periods of drought. |
Class Sarcopterygii: Lobe- Finned Fishes
The lobe-finned fishes are today represented by only seven species: six species (three genera) of lungfishes and the coelacanth—survivors of a group once abundant during the Devonian period of the Paleozoic (Figures 26-22 and 26-23).
All early sarcopterygians had lungs as well as gills, and a tail of the heterocercal type. However, during the Paleozoic the orientation of the vertebral column changed so that the tail became symmetrical, with the median dorsal and ventral fins displaced posteriorly to form one continuous, flexible fin around the tail. This type of tail is called diphycercal (Figure 26-16). The strong, fleshy, paired lobed fins of the sarcopterygians (pectoral and pelvic) may have been used much like four legs to scuttle along the bottom. They had powerful jaws and their skin was covered with heavy scales that consisted of a dentine-like material called cosmine overlaid by a thin enamel.
Of the three surviving genera of lungfishes, most similar to early forms is Neoceratodus (Gr. neos, new, + keratos, horn, + odes, form), the living Australian lungfish, which may attain a length of 1.5 m (Figure 26-22). This lungfish, unlike its relatives, normally relies on gill respiration, and cannot survive long out of water. The South American lungfish Lepidosiren (L. lepidus, pretty, + siren, mythical mermaid) and the African lungfish Protopterus (Gr. protos, first, + pteron, wing) can live out of water for long periods of time. Protopterus lives in African streams and ponds that may dry during the dry season, with their mud beds baked hard by the hot tropical sun. The fish burrows down at the approach of the dry season and secretes a copious slime that is mixed with mud to form a hard cocoon in which it estivates until the rains return. Surprisingly little is known about the ecology of the South American lungfish Lepidosiren.
Coelacanths and rhipidistians collectively have been termed crossopterygians, but this group is considered polyphytetic and no longer is recognized by most classifications. The rhipidistians flourished in the late Paleozoic era and then became extinct. Rhipidistians are of special importance because they include the ancestors of the tetrapods (and, in cladistic terms, are therefore a paraphyletic group). The coelacanths also arose in the Devonian period, radiated somewhat, and reached their evolutionary peak in the Mesozoic era. At the end of the Mesozoic era they nearly disappeared but left one remarkable surviving species, Latimeria chalumnae (named for M. Courtenay- Latimer, South African museum director) (Figure 26-23). Since the last coelacanths were believed to
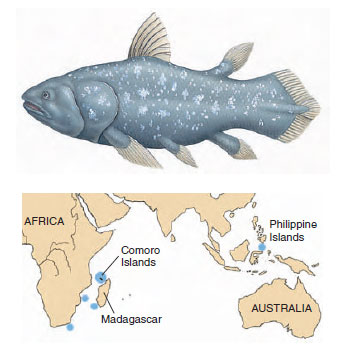 |
| Figure 26-23 The coelacanth Latimeria chalumnae is a surviving marine relict of a group of lobe-finned fishes that flourished some 350 million years ago. |
The “modern” marine coelacanth is a descendant of the Devonian freshwater stock. The tail is of the diphycercal type (Figure 26-16) but possesses a small lobe between the upper and lower caudal lobes, producing a three-pronged structure (Figure 26-23).
Coelacanths are a deep metallic blue with irregular white or brassy flecks, providing camouflage against the dark lava-cave reefs they inhabit. Young are born fully formed after hatching internally from eggs 9 cm in diameter—the largest among bony fishes.




