Sulfur Status and Plant Health
Although the significance of individual nutrients for maintaining or promoting plant health saw some interest in the 1960s and 1970s (143), research in the field of nutrient-induced resistance mechanisms has been scarce because of its complexity, and because of its limited practical significance due to the availability of effective pesticides.Since the beginning of the 1980s, atmospheric sulfur depositions have been declining drastically after Clean Air Acts came into force, and severe sulfur deficiency advanced to a major nutritional disorder in Western Europe (114,310,311). Increased infections of agricultural crops with fungal pathogens were observed, and diseases spread throughout the regions that were never infected before (312). Sulfur fertilization, applied to the soil as sulfate, proved to have a significant effect on the infection rate and infection severity of different crops by fungal diseases (148). Sulfur fertilization increased the resistance against various fungal diseases in different crops under greenhouse (313,314) and field conditions (315–317). Based on these findings, the concept of sulfurinduced resistance (SIR) was developed; research in this field has strengthened since then, and the advances made are discussed comprehensively by Bloem et al. (318) and Haneklaus et al. (148).
The term SIR stands for the reinforcement of the natural resistance of plants against fungal pathogens through triggering of the stimulation of metabolic processes involving sulfur by targeted fertilizer application strategies (148). A sufficient sulfur supply and an adequate availability of plantavailable sulfate are presumably a prerequisite for inducing S-dependent resistance mechanisms in the plant so that the required sulfur rates and sulfur status may be higher than the physiological demand.
The mechanisms possibly involved in SIR may be related to processes of induced resistance (319), for example, via the formation of phytoalexins and glutathione, or the requirement of cysteine for the synthesis of salicylic acid by �-oxidation and the cysteine pool itself. Another option is the release of reduced sulfur gases, such as H2S, which is described in the literature as being fungitoxic. The H2S may be produced prior to or after cysteine formation (320). Two enzymes that could be responsible for the H2S release are L-cysteine desulfhydrase (LCD) and O-acetyl-L-serine(thiol)lyase (OAS-TL). The LCD catalyzes the decomposition of cysteine to pyruvate, ammonia, and H2S. The OAS-TL is responsible for the incorporation of inorganic sulfur into the amino acid cysteine, which can be subsequently converted into other sulfur-containing compounds such as methionine or glutathione. The H2S is evolved in a side reaction because of the nature of the pyridoxal 5'-phosphate cofactor and the specific reaction mechanism of the OAS-TL protein (321). There is wide variation with regard to specifications about the release of H2S, ranging from 0.04 ng g-1 s-1 in whole soybean plants on a dry matter basis (322) to 100 pmol min-1 cm-1 in leaf discs of cucumber (323). Thus, H2S emissions of cut plant parts may be 500 times higher than in intact plants (Table 7.7).
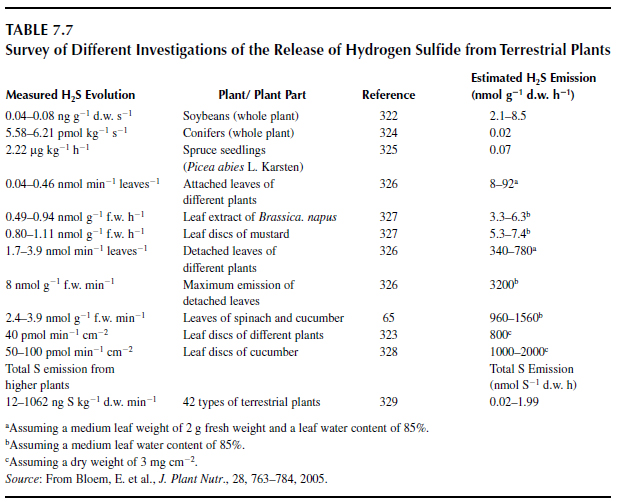 |
The release of H2S by plants is supposedly regulated by interactions in the N and sulfur metabolic pathways. Lakkineni et al. (327) demonstrated a distinct increase in H2S emissions when leaf discs of mustard, wheat, and groundnut (Arachis hypogaea L.) were fed with sulfate or cysteine (Table 7.8). Supply of additional nitrogen with the sulfate did not cause H2S emissions to increase (Table 7.8). Lakkineni et al. (330) suggested a preferable synthesis of nitrogen- or sulfur-containing products at the level of substrate availability.
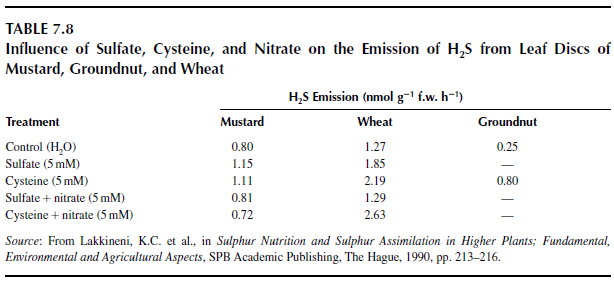 |
H2S and DMS emissions by plants are, however, supposedly not involved in SIR against fungal pathogens belonging to the class Basidiomycetes, as fumigation experiments with fungal mycelium of Rhizoctonia solani revealed that the pathogen metabolized both gases efficiently (331).
The amino acids cysteine and methionine are the major end products of sulfate assimilation in plants and bind up to 90% of the total sulfur (320). Conditions of sulfur deficiency will result in a decrease of sulfur-containing amino acids in proteins (5). As the amino acid composition is genetically determined, this effect is limited, and thereafter the total protein content will be reduced (5). Amino acid type and concentration in plant tissues are related to the susceptibility of plants to pathogens (332). Amino acids occur in the free state in plants, and the amino acids cysteine and methionine are enriched in resistant plant tissues. Soil-applied sulfur significantly increased the free cysteine content in the vegetative tissue from 0.5 to 1.2 �mol g-1 d.w. (63). Bosma et al. (333) reported a two- to five-fold increase in the content of water-soluble nonprotein sulfhydryl compounds in clover (Trifolium spp.) and spinach after fumigation with H2S under field conditions, whereby the cysteine content increased 10-fold. De Kok (18) reported similar results for fumigation experiments with sulfur dioxide.
Glutathione is a major, free, low-molecular, nonprotein, thiol compound and is an important reservoir for nonprotein reduced sulfur in plants (66). A relationship between glutathione content and the extent of protection against fungal diseases exists (72). A low glutathione content in plants does not inevitably imply, however, a higher susceptibility of the plant, as a rapid accumulation of glutathione in response to pathogen attack was noted (334), and this observation proved to be decisive in pathogenesis (72). Sulfur-deficient plants have very low glutathione concentrations, and sulfur fertilization significantly increases the free thiol content (Table 7.3). Basically, sulfur-deficient plants are expected to be more vulnerable to stress factors, which are usually compensated by the glutathione system so that sulfur fertilization should have a positive effect on resistance mechanisms.
Phytoalexins are important for plant defense (335). Phytoalexins are secondary plant metabolites which are synthesized de novo and accumulate in response to diverse forms of stress, including pathogenesis (336). The immunity is generally of short duration and is concentrated around the infected area. According to this definition, the formation of elemental sulfur, the stress-induced formation of pathogenesis-related (PR) proteins, and a novel class of LMW antibiotics, all come under the term phytoalexins. At the moment however, the influence of the sulfur nutritional status on phytoalexin synthesis can only be speculated from the dependency of their precursors on the sulfur supply. The influence of the sulfur nutritional status on the synthesis of PR-12, PR-13, and PR-14 proteins and elemental sulfur depositions in plant tissues remains obscure too (148).




