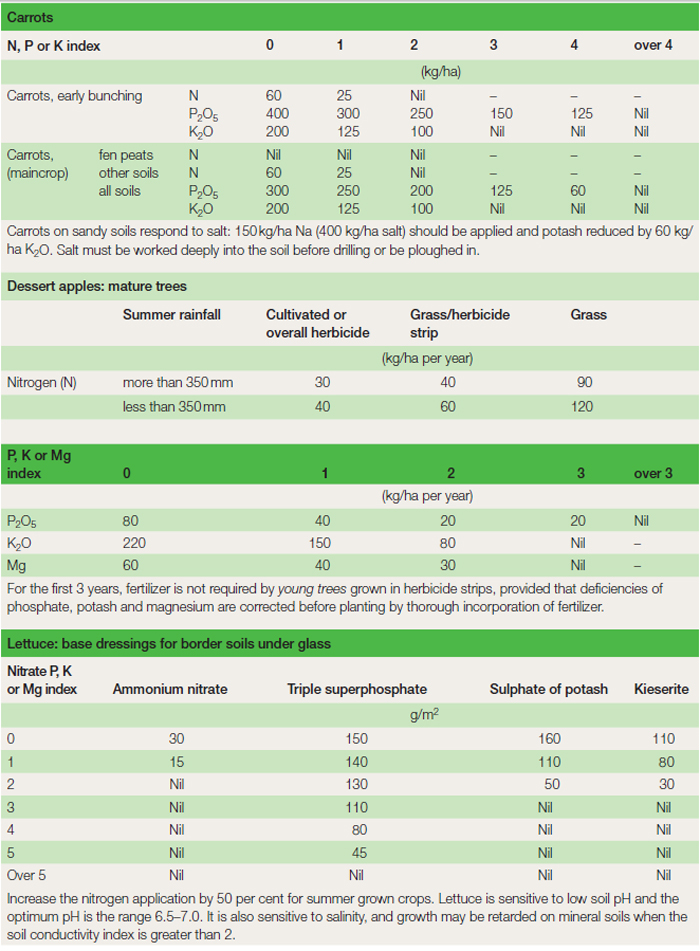
Fertilizer programmes
Content
The fertilizer applications required to produce the desired plant growth vary according to the type of plant, climate, season of growth, other sources of nutrients applied and the nutrient status of the soil. General advice is available in many publications including those of the national advisory services and horticultural industries. Examples are given in Table 21.4. Growing medium analysis The nutrient status of growing media varies greatly between the different materials and within the same materials as time passes. The nutrient levels change because they are being lost by plant uptake, leaching and fixation and gained by the weathering of clay, mineralization of organic matter, and the addition of lime and fertilizers. There are many visual symptoms which indicate a deficiency of one or more essential nutrients (see minerals), but unfortunately by the time they appear the plant has probably already suffered a check in growth or change in the desired type of growth. The concentration of minerals in the plant and the nature of growth are linked so that plant tissue analysis, usually on selected leaves, can provide useful information, particularly in the diagnosis of some nutrient deficiencies; e.g. it is used to identify the calcium levels in apples in order to check their storage qualities. However, nutrient supply is usually assessed by analysis of the growing medium. There is general agreement about the methodology for analyzing soils: However, there needs to be some care where analysis of other growing media is undertaken, because there are considerable differences between the methods particularly with regard to dilution of the nutrient extractant. A representative sample of the growing medium is taken and its nutrient status determined. This involves extracting the available nutrients and measuring the quantities present. The pH level is determined and, where appropriate, the lime requirement: The conductivity of growing media from protected culture is also measured. The nitrogen status of soils is usually determined from previous cropping because, outdoors, nitrates are washed out over winter and their release from organic matter reserves is very variable. In protected planting, nitrate and ammonia levels are usually determined. Results are often given in the form of an index number in order to simplify their presentation and interpretation. The ADAS soil analysis index is based on a ten point scale from 0 (indicating levels which correspond to probable failure of plants if nutrient is not supplied) to 9 (indicating excessively high levels of nutrient present); most outdoor soils normally give levels of 1 to 3.
Fertilizer recommendations The results of the growing medium analysis are interpreted with the appropriate nutrient requirement tables to determine the actual amount of fertilizer to apply. These tables usually have growing medium nutrient status indices to aid interpretation and results are normally given in kg of nutrient per hectare or grams of nutrient per square metre (Table 21.4). In some cases the amount of named fertilizer required is stated; if another fertilizer is to be used to supply the nutrient the quantity needed must be calculated using the nutrient content figures (Table 21.2). It is important that throughout the fertilizer planning process the same units are used, i.e. per cent P2O5 or P per cent; K2O or per cent K. Conversion figures are:
Sampling Normally only a small proportion of the whole growing medium is submitted for analysis and therefore it must be a representative sample of the whole. This is not easy because of the variability of growing media, particularly soils. It is recommended that each sample submitted for testing should be taken from an area no greater than 4 hectares (see Figure 21.8). The material sampled must itself be uniform and so only areas with the same characteristics and past history should be put in the same sample. Irrespective of the area involved, from small plot to 4 hectare field, at least 25 sub-samples should be taken by walking a zigzag path, avoiding the atypical areas such as headlands, wet spots, old paths, hedge lines, old manure heaps, etc. The same amount of soil should be taken from each layer to a depth of 150 mm. This is most easily achieved with a soil auger or tubular corer. Peat bags should be sampled with a cheese-type corer by taking a core at an angle through the planting hole on the opposite side of the plant to the drip nozzle from each of 30 bags chosen from an area up to a maximum of 0.5 hectares. Discard the top 20 mm of each core and if necessary take more than 30 cores to make up a one litre sample for analysis. Samples should be submitted to analytical laboratories in clean containers capable of completely retaining the contents. They should be accompanied by name and address of supplier, the date of sampling and any useful background information. All samples must be clearly identified. Further details of sampling methods in greenhouses or orchards, bags, pots, straw bales, water, etc., are obtainable from the advisory services used. Remember, the result of the analysis can be no better than the extent to which the sample is representative of the whole. |
||||||||||||||||||||||||||||||||||||||||